Chemistry:Manganese(II) iodide
| |

| |
| |
| Names | |
|---|---|
| IUPAC name
Manganese(II) iodide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| MnI2 | |
| Molar mass | 308.747 g/mol |
| Appearance | pink crystalline |
| Density | 5.01 g/cm3 |
| Melting point | 701 °C (1,294 °F; 974 K) (anhydrous) 80 °C (tetrahydrate) |
| Boiling point | 1,033 °C (1,891 °F; 1,306 K) |
| soluble | |
| +14,400·10−6 cm3/mol | |
| Structure | |
| Rhombohedral, hP3, SpaceGroup = P-3m1, No. 164 | |
| octahedral | |
| Hazards[1] | |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| H360 | |
| P201, P202, P281, P308+313, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | non-flammable |
| Related compounds | |
Other anions
|
Manganese(II) fluoride Manganese(II) chloride Manganese(II) bromide |
Other cations
|
Iron(II) iodide Cobalt(II) iodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
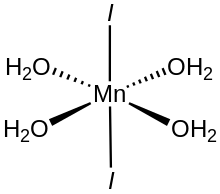
Manganese(II) iodide is the chemical compound composed of manganese and iodide with the formula MnI2(H2O)n. The tetrahydrate is a pink solid while the anhydrous derivative is beige.[2] Both forms feature octahedral Mn centers. Unlike MnCl2(H2O)4 and MnBr2(H2O)4 which are cis, MnI2(H2O)4 is trans.[3]
Preparation
Anhydrous MnI2 is prepared from the elements:[4]
- Mn + I
2 → MnI
2
The tetrahydrate can be prepared by treating manganese(II) carbonate with hydriodic acid. The anhydrous form can be produced from it by dehydration in a vacuum.[2]
Properties
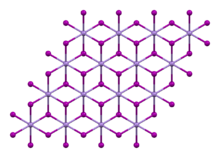
Samples turn brown in air under the influence of light as a result of the oxidation of the iodide ion to iodine.[5] It has a trigonal crystal structure of the cadmium iodide type (polytype 2H)[6][7] with the space group P3m1 (space group no. 164). It dissolves in water and decomposes.[5] The tetrahydrate has a monoclinic crystal structure with the space group P21/c (No. 14).[3]
Applications
It is often used in the lighting industry.[8]
References
- ↑ "223646 Manganese(II) iodide 98%". Sigma-Aldrich. http://www.sigmaaldrich.com/catalog/search/ProductDetail/ALDRICH/223646.
- ↑ 2.0 2.1 Hosseiny, Afshin; MacKie, Anthony G.; McAuliffe, Charles A.; Minten, Karl (1981). "The Coordination Chemistry of Manganese". Inorganica Chimica Acta 49: 99–105. doi:10.1016/S0020-1693(00)90464-X.
- ↑ 3.0 3.1 Moore, J. E.; Abola, J. E.; Butera, R. A. (1985-09-15). "Structure of manganese(II) iodide tetrahydrate, MnI2.4H2O" (in en). Acta Crystallographica Section C: Crystal Structure Communications 41 (9): 1284–1286. doi:10.1107/S0108270185007466. ISSN 0108-2701. https://scripts.iucr.org/cgi-bin/paper?S0108270185007466.
- ↑ Friour, G.; Cahiez, G.; Normant, J. F. (1984). "Organomanganous Reagents; IX. Preparation of Various Halogenated, Alkoxylated, Aryloxylated, and Arylsulfenylated Ketones from Correspondingly Functionalized Carboxylic Acid Chlorides or Anhydrides". Synthesis 1984: 37–40. doi:10.1055/s-1984-30724.
- ↑ 5.0 5.1 Ans, Jan d'; Ans, Jan d' (1998). Elemente, anorganische Verbindungen und Materialien. Taschenbuch für Chemiker und Physiker / D'Ans (4., neubearb. u. rev. Aufl ed.). Ort nicht ermittelbar: Verlag nicht ermittelbar. ISBN 978-3-540-60035-0.
- ↑ Riedel, Erwin, ed (2007). Moderne anorganische Chemie: mit CD-ROM (3. Aufl ed.). Berlin: de Gruyter. ISBN 978-3-11-019060-1.
- ↑ Cable, J. W.; Wilkinson, M. K.; Wollan, E. O.; Koehler, W. C. (1962). "Neutron Diffraction Investigation of the Magnetic Order in MnI2". Phys. Rev. 125 (6): 1860-1864. doi:10.1103/PhysRev.125.1860.
- ↑ Cepanec, Ivica (2004). Synthesis of Biaryls. Elseveir. pp. 104. ISBN 0-08-044412-1. https://books.google.com/books?id=UMLOo1wXWdwC&dq=%22Manganese(II)+bromide+%22&pg=PA104. Retrieved 2008-06-18.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | RaI2 | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | EsI3 | Fm | Md | No | Lr | |||
 |

