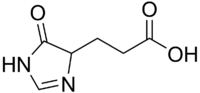
Chemistry:Imidazol-4-one-5-propionic acid
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
3-(5-Oxo-1,4-dihydroimidazol-4-yl)propanoic acid
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| MeSH | Imidazol-4-one-5-propionic+acid |
PubChem CID
|
|
| |
| |
| Properties | |
| C6H8N2O3 | |
| Molar mass | 156.139 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Imidazol-4-one-5-propionic acid is an intermediate in the metabolism of histidine. It is a colorless compound that is sensitive to light in air. The compound features an imidazolone ring.[1]
Occurrence
It arises via the action of urocanase on urocanic acid. Hydrolysis of the heterocycle to the glutamic acid derivative is catalyzed by imidazolonepropionate hydrolase.
Microbial production of imidazol-4-one-5-propionic acid in the human gut has been shown to affect insulin signaling, which is relevant to type II diabetes.[2]
See also
- Formiminoglutamic acid
- Urocanate
- Urocanate hydratase
References
- ↑ Hassall, H.; Greenberg, D. M. (1971). "Preparation and properties of 4(5)-imidazolone-5(4)-propionic acid". Methods Enzymol. 17(Pt. B): 89–91. doi:10.1016/0076-6879(71)17014-0.
- ↑ Koh, Ara; Molinaro, Antonio; Ståhlman, Marcus; Khan, Muhammad Tanweer; Schmidt, Caroline; Mannerås-Holm, Louise; Wu, Hao; Carreras, Alba et al. (2018). "Microbially Produced Imidazole Propionate Impairs Insulin Signaling through mTORC1". Cell 175 (4): 947–961.e17. doi:10.1016/j.cell.2018.09.055. PMID 30401435.
 |

