Biology:Dactinomycin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Cosmegen |
| Other names | Actinomycin D 2-Amino- 4,6-dimethyl- 3-oxo- 3H-phenoxazine- 1,9-dicarboxylic acid bis- [(5,12-diisopropyl- 9,13,16-trimethyl- 4,7,11,14,17-pentaoxo- hexadecahydro- 10-oxa- 3a,6,13,16-tetraaza- cyclopentacyclohexadecen- 8-yl)- amide] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682224 |
| Pregnancy category |
|
| Routes of administration | IV |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 5% |
| Metabolism | hepatic |
| Elimination half-life | 36 hours |
| Excretion | Bile[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| Chemical and physical data | |
| Formula | C62H86N12O16 |
| Molar mass | 1255.438 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Dactinomycin, also known as actinomycin D, is a chemotherapy medication used to treat a number of types of cancer.[2] This includes Wilms tumor, rhabdomyosarcoma, Ewing's sarcoma, trophoblastic neoplasm, testicular cancer, and certain types of ovarian cancer.[2] It is given by injection into a vein.[2]
Most people develop side effects.[2] Common side effects include bone marrow suppression, vomiting, mouth ulcers, hair loss, liver problems, infections, and muscle pains.[2] Other serious side effects include future cancers, allergic reactions, and tissue death if extravasation occurs.[2] Use in pregnancy may harm the baby.[2] Dactinomycin is in the cytotoxic antibiotic family of medications.[3] It is believed to work by blocking the creation of RNA.[2]
Dactinomycin was approved for medical use in the United States in 1964.[2] It is on the 2023 World Health Organization's List of Essential Medicines.[4]
Medical use
Actinomycin is a clear, yellowish liquid administered intravenously and most commonly used in treatment of a variety of cancers, including:
- Gestational trophoblastic neoplasia[5]
- Wilms' tumor[6]
- Rhabdomyosarcoma[7]
- Ewing's sarcoma[8]
- Malignant hydatidiform mole[9]
Sometimes it will be combined with other drugs in chemotherapy regimens, like the VAC regimen (with vincristine and cyclophosphamide) for treating rhabdomyosarcoma and Ewing's sarcoma.[10]
It is also used as a radiosensitizer in adjunct to radiotherapies,[11] since it can increase the radiosensitivity of tumor cells by inhibiting repair of sublethal radiation damage and delay the onset of the compensatory hyperplasia that occurs following irradiation.[12]
Side effects
Common adverse drug reaction includes bone marrow suppression, fatigue, hair loss, mouth ulcer, loss of appetite and diarrhea. Actinomycin is a vesicant, if extravasation occurs.
Mechanism
In cell biology, actinomycin D is shown to have the ability to inhibit transcription. Actinomycin D does this by binding DNA at the transcription initiation complex and preventing elongation of RNA chain by RNA polymerase.[13]
History
Actinomycin D was the first antibiotic shown to have anti-cancer activity.[14] It was first isolated by Selman Waksman and his co-worker H. Boyd Woodruff in 1940,[15] using fermentation products from Streptomyces.[16] It was approved by the U.S. Food and Drug Administration (FDA) on December 10, 1964,[17] and launched by Merck Sharp and Dohme under the trade name Cosmegen.
Research use
Because actinomycin can bind DNA duplexes, it can also interfere with DNA replication, although other chemicals such as hydroxyurea are better suited for use in the laboratory as inhibitors of DNA synthesis.
Actinomycin D and its fluorescent derivative, 7-aminoactinomycin D (7-AAD), are used as stains in microscopy and flow cytometry applications. The affinity of these stains/compounds for GC-rich regions of DNA strands makes them excellent markers for DNA. 7-AAD binds to single stranded DNA; therefore it is a useful tool in determining apoptosis and distinguishing between dead cells and live ones.[18]
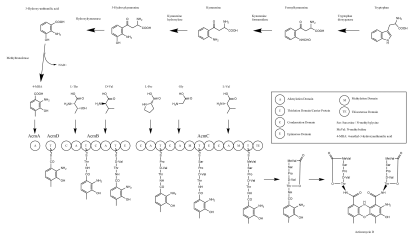
Biosynthesis
Actinomycin D is composed of a central phenoxazinone chromophore tethered to two identical cyclic peptides and was first structurally characterized by Nuclear Magnetic Resonance (NMR) analysis in 1982.[19] The biosynthesis of Actinomycin D has been under investigation since its discovery; early fermentation feeding experiments revealed the roles of both tryptophan and D-glutamate as precursor substrates,[20][19] and strain mutagenesis experiments demonstrated that a phenoxazinone synthase enzyme might be responsible for coupling of two moieties of 4-methyl-3-hydroxyanthranilic acid (4-MHA) into the final phenoxazinone structure.[21] The 4-MHA substrate was shown to be produced from tryptophan through the action of enzymes such as tryptophan dioxygenase, kynurenine formamidase, kynurenine hydroxylase, hydroxykynurenase, and methyltransferase.[22][23]
Early experiments elucidated the presence of non-ribosomal peptide synthetases,[24][25][26][27] and subsequent purification and heterologous expression experiments[24][25][28][29] showed the acmD and acmA genes to be responsible for activation of the 4-MHA, which then undergoes chain elongation through the action of the acmB and acmC genes. In total, the NRPS assembly line is composed of twenty-two modules, including two each of epimerase and methylase domains.[30][23] Recent sequencing of the actinomycin D gene cluster in Streptomyces chrysomallus showed that the four NRPS genes were surrounded on both sides by the two clusters of the genes involved in the well-studied kynurenine pathway and responsible for the production of 4-MHA from tryptophan, with nine paralogs identified between the two clusters.[23]
References
- ↑ Pharmacology and Therapeutics for Dentistry. Mosby. 2017. pp. 530–562. doi:10.1016/B978-0-323-39307-2.00036-9.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 "Dactinomycin". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/dactinomycin.html.
- ↑ British national formulary : BNF 69 (69 ed.). British Medical Association. 2015. p. 582. ISBN 9780857111562.
- ↑ The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. 2023. WHO/MHP/HPS/EML/2023.02.
- ↑ "Results with EMA/CO (etoposide, methotrexate, actinomycin D, cyclophosphamide, vincristine) chemotherapy in gestational trophoblastic neoplasia". International Journal of Gynecological Cancer 16 (3): 1432–1438. 2006. doi:10.1111/j.1525-1438.2006.00606.x. PMID 16803542.
- ↑ "The treatment of Wilms' tumor: results of the Second National Wilms' Tumor Study". Cancer 47 (9): 2302–2311. May 1981. doi:10.1002/1097-0142(19810501)47:9<2302::aid-cncr2820470933>3.0.co;2-k. PMID 6164480.
- ↑ "Immune-mediated thrombocytopenia following dactinomycin therapy in a child with alveolar rhabdomyosarcoma: the unresolved issues". Journal of Pediatric Hematology/Oncology 26 (11): 777–779. November 2004. doi:10.1097/00043426-200411000-00020. PMID 15543019.
- ↑ "Improved outlook for Ewing's sarcoma with combination chemotherapy (vincristine, actinomycin D and cyclophosphamide) and radiation therapy". Cancer 38 (5): 1925–1930. November 1976. doi:10.1002/1097-0142(197611)38:5<1925::AID-CNCR2820380510>3.0.CO;2-J. PMID 991106.
- ↑ "Reproductive outcome after discharge of patients with high-risk hydatidiform mole with or without use of one bolus dose of actinomycin D, as prophylactic chemotherapy, during the uterine evacuation of molar pregnancy". Gynecologic Oncology 115 (3): 476–481. December 2009. doi:10.1016/j.ygyno.2009.09.012. PMID 19818481.
- ↑ "Vincristine, Actinomycin, and Cyclophosphamide Compared With Vincristine, Actinomycin, and Cyclophosphamide Alternating With Vincristine, Topotecan, and Cyclophosphamide for Intermediate-Risk Rhabdomyosarcoma: Children's Oncology Group Study D9803". J Clin Oncol 27 (31): 5182–5188. 2009. doi:10.1200/JCO.2009.22.3768. PMID 19770373.
- ↑ "41 - Dermatologic Toxicities of Anticancer Therapy". Abeloff's Clinical Oncology. Elsevier. 2020. pp. 621–648. doi:10.1016/B978-0-323-47674-4.00041-4. ISBN 9780323476744.
- ↑ "Mechanism of intestinal radiosensitization by actinomycin D". The British Journal of Radiology 46 (544): 302–308. April 1973. doi:10.1259/0007-1285-46-544-302. PMID 4720744.
- ↑ "Actinomycin and DNA transcription". Proceedings of the National Academy of Sciences of the United States of America 82 (16): 5328–5331. August 1985. doi:10.1073/pnas.82.16.5328. PMID 2410919. Bibcode: 1985PNAS...82.5328S.
- ↑ "Actinomycin. Chemistry and mechanism of action". Chemical Reviews 74 (6): 625–652. 1974. doi:10.1021/cr60292a002.
- ↑ "Bacteriostatic and bacteriocidal substances produced by soil actinomycetes". Proceedings of the Society for Experimental Biology and Medicine 45: 609–614. 1940. doi:10.3181/00379727-45-11768.
- ↑ "Dactinomycin". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. National Institute of Diabetes and Digestive and Kidney Diseases. 2012.
- ↑ "Drugs@FDA: Dactinomycin". https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&varApplNo=050682.
- ↑ "Cell kinetic study of normal human bone marrow hematopoiesis and acute leukemia using 7AAD/PY". European Journal of Haematology 64 (1): 10–21. January 2000. doi:10.1034/j.1600-0609.2000.09005.x. PMID 10680701.
- ↑ 19.0 19.1 "Biosynthesis and characterization of [15N]actinomycin D and conformational analysis by nitrogen-15 nuclear magnetic resonance". Biochemistry 21 (25): 6496–6503. December 1982. doi:10.1021/bi00268a027. PMID 6129895.
- ↑ "Biosynthesis of the actinomycin chromophore. Influence of alpha-, 4-, 5-, and 6-methyl-DL-tryptophan on actinomycin synthesis". Biochimica et Biophysica Acta 62 (1): 80–90. July 1962. doi:10.1016/0006-3002(62)90493-6. PMID 13913519.
- ↑ "Phenoxazinone biosynthesis: accumulation of a precursor, 4-methyl-3-hydroxyanthranilic acid, by mutants of Streptomyces parvulus". Journal of General Microbiology 111 (1): 121–132. March 1979. doi:10.1099/00221287-111-1-121. PMID 458423.
- ↑ "Actinomycin synthesis in Streptomyces antibioticus: enzymatic conversion of 3-hydroxyanthranilic acid to 4-methyl-3-hydroxyanthranilic acid". Journal of Bacteriology 169 (12): 5575–5578. December 1987. doi:10.1128/jb.169.12.5575-5578.1987. PMID 2445729.
- ↑ 23.0 23.1 23.2 23.3 "The actinomycin biosynthetic gene cluster of Streptomyces chrysomallus: a genetic hall of mirrors for synthesis of a molecule with mirror symmetry". Journal of Bacteriology 192 (10): 2583–2595. May 2010. doi:10.1128/JB.01526-09. PMID 20304989.
- ↑ 24.0 24.1 "Construction and in vitro analysis of a new bi-modular polypeptide synthetase for synthesis of N-methylated acyl peptides". Chemistry & Biology 7 (4): 287–297. April 2000. doi:10.1016/s1074-5521(00)00103-4. PMID 10780924.
- ↑ 25.0 25.1 "Molecular cloning of the actinomycin synthetase gene cluster from Streptomyces chrysomallus and functional heterologous expression of the gene encoding actinomycin synthetase II". Journal of Bacteriology 180 (9): 2468–2474. May 1998. doi:10.1128/jb.180.9.2468-2474.1998. PMID 9573200.
- ↑ "The initiation of peptide formation in the biosynthesis of actinomycin". The Journal of Biological Chemistry 268 (14): 10612–10620. May 1993. doi:10.1016/S0021-9258(18)82242-6. PMID 7683683.
- ↑ "Epimerization of the D-valine portion in the biosynthesis of actinomycin D". Biochemistry 33 (31): 9358–9364. August 1994. doi:10.1021/bi00197a041. PMID 8049237.
- ↑ "Actinomycin synthetases. Multifunctional enzymes responsible for the synthesis of the peptide chains of actinomycin". The Journal of Biological Chemistry 262 (12): 5852–5856. April 1987. doi:10.1016/s0021-9258(18)45652-9. PMID 3571237.
- ↑ "Acyl pentapeptide lactone synthesis in actinomycin-producing streptomycetes by feeding with structural analogs of 4-methyl-3-hydroxyanthranilic acid". The Journal of Biological Chemistry 259 (13): 8226–8231. July 1984. doi:10.1016/s0021-9258(17)39717-x. PMID 6203903.
- ↑ "Molecular characterization of the genes of actinomycin synthetase I and of a 4-methyl-3-hydroxyanthranilic acid carrier protein involved in the assembly of the acylpeptide chain of actinomycin in Streptomyces". The Journal of Biological Chemistry 274 (18): 12508–12516. April 1999. doi:10.1074/jbc.274.18.12508. PMID 10212227.
External links
- "Dactinomycin". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/dactinomycin.
