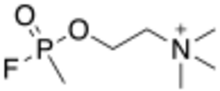
Chemistry:Methylfluorophosphonylcholine
| |
| Names | |
|---|---|
| Preferred IUPAC name
N,N,N-Trimethyl-2-[(methylphosphonofluoridoyl)oxy]ethan-1-aminium | |
| Other names
Methylfluorophosphorylcholine, MFPCh
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C6H16FNO2P | |
| Molar mass | 184.171 g·mol−1 |
| Hazards | |
| Main hazards | Extremely toxic |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
100 μg/kg (mice, intraperitoneal)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Methylfluorophosphonylcholine (MFPCh) is an extremely toxic chemical compound related to the G-series nerve agents. It is an extremely potent acetylcholinesterase inhibitor which is around 100 times more potent than sarin at inhibiting acetylcholinesterase in vitro,[2] and around 10 times more potent in vivo, depending on route of administration and animal species tested.[3] MFPCh is resistant to oxime reactivators, meaning the acetylcholinesterase inhibited by MFPCh can't be reactivated by cholinesterase reactivators.[2][4][5] MFPCh also acts directly on the acetylcholine receptors.[6] However, despite its high toxicity, methylfluorophosphonylcholine is a relatively unstable compound and degrades rapidly in storage, so it was not deemed suitable to be weaponised for military use.[7]
See also
References
- ↑ "ChemIDplus". https://chem.nlm.nih.gov/chemidplus/rn/2797-10-6.
- ↑ 2.0 2.1 Szinicz, L.; Worek, F.; Thiermann, H. (1 April 2004). "Reactivation and aging kinetics of human acetylcholinesterase inhibited by organophosphonylcholines". Archives of Toxicology 78 (4): 212–217. doi:10.1007/s00204-003-0533-0. PMID 14647978.
- ↑ Black RM, Harrison JM. The chemistry of organophosphorus chemical warfare agents. Chapter 10 of The chemistry of organophosphorus compounds. Volume 4, Ter- and quinque-valent phosphorus acids and their derivatives. (1996) ISBN:0-471-95706-2
- ↑ Enander, Inger; Villadsen, John V.; Sillén, Lars Gunnar; Sillén, Lars Gunnar; Zaar, B.; Diczfalusy, E. (1958). "Experiments with Methyl-fluoro-phosphorylcholine-inhibited Cholinesterase.". Acta Chemica Scandinavica 12: 780–781. doi:10.3891/acta.chem.scand.12-0780.
- ↑ Fredriksson, T.; Tibbling, G. (May 1959). "Reversal of effects on the rat nerve-diaphragm preparation produced by methylfluorophosphorylcholines". Biochemical Pharmacology 2 (1): 63–64. doi:10.1016/0006-2952(59)90058-9.
- ↑ Fredriksson, T.; Tibbling, G. (November 1959). "Demonstration of direct cholinergic receptor effects of methylfluorophosphorylcholines". Biochemical Pharmacology 2 (4): 286–289. doi:10.1016/0006-2952(59)90042-5.
- ↑ Black RM, Harrison JM. The chemistry of organophosphorus chemical warfare agents. Chapter 10 of The chemistry of organophosphorus compounds. Volume 4, Ter- and quinque-valent phosphorus acids and their derivatives. (1996) ISBN:0-471-95706-2
 |

