Chemistry:Rosmarinic acid
| |
| Names | |
|---|---|
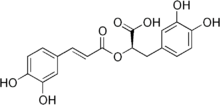
| Preferred IUPAC name
(2R)-3-(3,4-Dihydroxyphenyl)-2-{[(2E)-3-(3,4-dihydroxyphenyl)prop-2-enoyl]oxy}propanoic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C18H16O8 | |
| Molar mass | 360.318 g·mol−1 |
| Appearance | Red-orange powder |
| Melting point | 171 to 175 °C (340 to 347 °F; 444 to 448 K) |
| Slightly soluble | |
| Solubility in other solvents | Well soluble in most organic solvents[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Rosmarinic acid, named after rosemary (Salvia rosmarinus Spenn.), is a polyphenol constituent of many culinary herbs, including rosemary (Salvia rosmarinus L.), perilla (Perilla frutescens L.), sage (Salvia officinalis L.), mint (Mentha arvense L.), and basil (Ocimum basilicum L.).[1]
History
Rosmarinic acid was first isolated and characterized in 1958 by the Italian chemists Scarpatti and Oriente from rosemary (Salvia rosmarinus),[2] after which the acid is named.
Chemistry
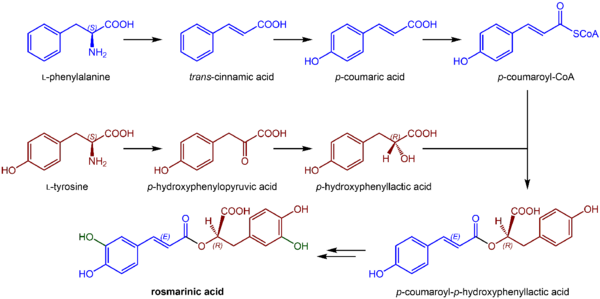
Chemically, rosmarinic acid is a caffeic acid ester, with tyrosine providing another phenolic ring via dihydroxyphenyl-lactic acid.[1] It has a molecular mass of 360 daltons.[1]
Natural occurrences
Rosmarinic acid accumulation is shown in hornworts, in the fern family Blechnaceae, and in species of several orders of mono- and dicotyledonous angiosperms.[3]
It is found most notably in many Lamiaceae (dicotyledons in the order Lamiales), especially in the subfamily Nepetoideae.[1][4] It is found in species used commonly as culinary herbs such as Ocimum basilicum (basil), Ocimum tenuiflorum (holy basil), Melissa officinalis (lemon balm), Salvia rosmarinus (rosemary), Origanum majorana (marjoram), Salvia officinalis (sage), thyme and peppermint.[1][5] It is also found in plants in the family Marantaceae (monocotyledons in the order Zingiberales)[3] such as species in the genera Maranta (Maranta leuconeura, Maranta depressa) and Thalia (Thalia geniculata).[6]
Rosmarinic acid and the derivative rosmarinic acid 3′-O-β-D-glucoside can be found in Anthoceros agrestis, a hornwort (Anthocerotophyta).[7]
Metabolism
The biosynthesis of rosmarinic acid uses 4-coumaroyl-CoA from the general phenylpropanoid pathway as a hydroxycinnamoyl donor.[1] The hydroxycinnamoyl acceptor substrate comes from the shikimate pathway: shikimic acid, quinic acid and 3,4-dihydroxyphenyllactic acid derived from L-tyrosine.[3] Thus, chemically, rosmarinic acid is an ester of caffeic acid with 3,4-dihydroxyphenyllactic acid, but biologically, it is formed from 4-coumaroyl-4′-hydroxyphenyllactate.[8] Rosmarinate synthase is an enzyme that uses caffeoyl-CoA and 3,4-dihydroxyphenyllactic acid to produce CoA and rosmarinate. Hydroxyphenylpyruvate reductase is also an enzyme involved in this biosynthesis.[9]
Uses
When extracted from plant sources or synthesized in manufacturing, rosmarinic acid may be used in foods or beverages as a flavoring, in cosmetics, or as a dietary supplement.[1]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 "Rosmarinic acid". PubChem, US National Library of Medicine. 10 July 2021. https://pubchem.ncbi.nlm.nih.gov/compound/5281792.
- ↑ Scarpati, M. L.; Oriente, G. (1958). "Isolamento costituzione e dell'acido rosmarinico (dal Rosmarinus off.)". Ricerca Scientifica 28: 2329–2333.
- ↑ 3.0 3.1 3.2 Petersen, M.; Abdullah, Y.; Benner, J.; Eberle, D.; Gehlen, K.; Hücherig, S.; Janiak, V.; Kim, K. H. et al. (2009). "Evolution of rosmarinic acid biosynthesis". Phytochemistry 70 (15–16): 1663–1679. doi:10.1016/j.phytochem.2009.05.010. PMID 19560175. Bibcode: 2009PChem..70.1663P.
- ↑ Distribution and taxonomic implications of some phenolics in the family Lamiaceae determined by ESR spectroscopy. J. A. Pedersen, Biochemical Systematics and Ecology, 2000, volume 28, pages 229–253
- ↑ Clifford, M. N. (1999). "Chlorogenic acids and other cinnamates. Nature, occurrence and dietary burden.". Journal of the Science of Food and Agriculture 79 (3): 362–372. doi:10.1002/(SICI)1097-0010(19990301)79:3<362::AID-JSFA256>3.0.CO;2-D.
- ↑ Abdullah, Yana; Schneider, Bernd; Petersen, Maike (12 December 2008). "Occurrence of rosmarinic acid, chlorogenic acid and rutin in Marantaceae species". Phytochemistry Letters 1 (4): 199–203. doi:10.1016/j.phytol.2008.09.010. Bibcode: 2008PChL....1..199A.
- ↑ Vogelsang, Katharina; Schneider, Bernd; Petersen, Maike (2006). "Production of rosmarinic acid and a new rosmarinic acid 3′-O-β-D-glucoside in suspension cultures of the hornwort Anthoceros agrestis Paton.". Planta 223 (2): 369–373. doi:10.1007/s00425-005-0089-8. PMID 16133208.
- ↑ "MetaCyc rosmarinic acid biosynthesis I". biocyc.org. http://www.biocyc.org/META/NEW-IMAGE?type=PATHWAY&object=PWY-5048.
- ↑ Petersen, M.; Alfermann, A. W. (1988). "Two new enzymes of rosmarinic acid biosynthesis from cell cultures of Coleus blumei: hydroxyphenylpyruvate reductase and rosmarinic acid synthase". Zeitschrift für Naturforschung C 43 (7–8): 501–504. doi:10.1515/znc-1988-7-804.
 |