Chemistry:Praseodymium(IV) fluoride
| |
| Names | |
|---|---|
| Other names
tetrafluoropraseodymium, praseodymium tetrafluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| F4Pr | |
| Molar mass | 216.90127 g·mol−1 |
| Appearance | light-yellow crystals |
| Density | g/cm3 |
| reacts with water | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Warning |
| Related compounds | |
Other cations
|
CeF4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Praseodymium(IV) fluoride (also praseodymium tetrafluoride) is a binary inorganic compound, a highly oxidised metal salt of praseodymium and fluoride[1] with the chemical formula PrF4.
Synthesis
Praseodymium(IV) fluoride can be prepared by the effect of krypton difluoride on praseodymium(IV) oxide:[2]
Praseodymium(IV) fluoride can also be made by the dissolution of sodium hexafluoropraseodymate(IV) in liquid hydrogen fluoride:[3]
Properties
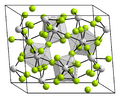
Praseodymium(IV) fluoride forms light yellow crystals. The crystal structure is anticubic and isomorphic to that of uranium tetrafluoride UF4.
Decomposes when heated:
Due to the high normal potential of the tetravalent praseodymium cations (Pr3+ / Pr4+: +3.2 V), praseodymium(IV) fluoride decomposes in water, releasing oxygen, O2.
See also
References
- ↑ Vent-Schmidt, Thomas; Riedel, Sebastian (November 6, 2015). "Investigation of Praseodymium Fluorides: A Combined Matrix-Isolation and Quantum-Chemical Study". Inorganic Chemistry 54 (23): 11114–11120. doi:10.1021/acs.inorgchem.5b01175. PMID 26544761. https://pubs.acs.org/doi/abs/10.1021/acs.inorgchem.5b01175. Retrieved 18 June 2021.
- ↑ Meyer, G.; Morss, Lester R. (1990-12-31) (in en). Synthesis of Lanthanide and Actinide Compounds. Springer Science & Business Media. p. 367. ISBN 978-0-7923-1018-1. https://books.google.com/books?id=bnS5elHL2w8C&q=%22PrF4%22+praseodymium&pg=PA60. Retrieved 18 June 2021.
- ↑ Emeléus, H. J.; Sharpe, A. G. (1977-09-01) (in en). Advances in Inorganic Chemistry and Radiochemistry. Academic Press. p. 368. ISBN 978-0-08-057869-9. https://books.google.com/books?id=es-Pu2hI5swC&q=%22PrF4%22+praseodymium&pg=PA63. Retrieved 18 June 2021.
 |

