Chemistry:Curium(III) fluoride
From HandWiki
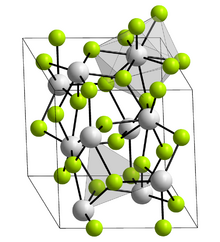 Crystal structure
| |
| Names | |
|---|---|
| IUPAC name
Curium(III) fluoride
| |
| Other names
Curium trifluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| CmF3 | |
| Appearance | Colorless solid[1] |
| Melting point | 1406 ± 20 °C; [1] |
| ~10 mg/L | |
| Structure | |
| Rhombohedral, hR24 | |
| P3c1, No. 165[2] | |
a = 0.7012 nm, c = 0.7198 nm
| |
Lattice volume (V)
|
0.30650 |
Formula units (Z)
|
6 |
| Thermochemistry | |
Std molar
entropy (S |
121 J/mol·K[1] |
Std enthalpy of
formation (ΔfH⦵298) |
−1660 kJ/mol[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Curium(III) fluoride or curium trifluoride is the chemical compound composed of curium and fluorine with the formula CmF3.[3] It is a white, nearly insoluble salt that has the same crystal structure as LaF3. It precipitates as a hydrate when fluoride ions are added to a weakly acidic Cm(III) solution; alternatively it can be synthesized by reacting hydrofluoric acid with Cm(OH)3. The anhydrous form is then obtained by desiccation or by treatment with hydrogen fluoride gas.[1]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Haire, Richard G. (2006). "Curium (9.7.3 Halides)". in Morss; Edelstein, Norman M.; Fuger, Jean. The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. p. 1417. doi:10.1007/1-4020-3598-5_9. ISBN 1-4020-3555-1. https://archive.org/details/chemistryactinid00katz.
- ↑ Nave, S. E.; Haire, R. G.; Huray, Paul G. (1983). "Magnetic properties of actinide elements having the 5f6 and 5f7 electronic configurations". Physical Review B 28 (5): 2317. doi:10.1103/PhysRevB.28.2317.
- ↑ Macintyre, Jane E. (23 July 1992) (in en). Dictionary of Inorganic Compounds. CRC Press. p. 3046. ISBN 978-0-412-30120-9. https://books.google.com/books?id=9eJvoNCSCRMC&dq=Curicium+tetrafluoride&pg=PA3046. Retrieved 27 June 2023.
 |
