Chemistry:Strontium fluoride
| |
| Names | |
|---|---|
| Other names
Strontium difluoride
Strontium(II) fluoride | |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| SrF2 | |
| Molar mass | 125.62 g/mol |
| Density | 4.24 g/cm3 |
| Melting point | 1,473 °C (2,683 °F; 1,746 K) |
| Boiling point | 2,460 °C (4,460 °F; 2,730 K) |
| 0.117 g/100 mL | |
Solubility product (Ksp)
|
4.33×10−9[1] |
| −37.2·10−6 cm3/mol | |
Refractive index (nD)
|
1.439 @0.58 μm |
| Structure | |
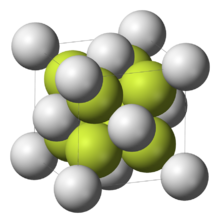
| cubic crystal system, cF12 | |
| Fm3m, #225 | |
a = 5.80 Å, b = 5.80 Å, c = 5.80 Å α = 90°, β = 90°, γ = 90°
| |
| Sr, 8, cubic F, 4, tetrahedral | |
| Hazards | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions
|
Strontium chloride Strontium bromide Strontium iodide |
Other cations
|
beryllium fluoride magnesium fluoride calcium fluoride barium fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Strontium fluoride, SrF2, also called strontium difluoride and strontium(II) fluoride, is a fluoride of strontium. It is a brittle white crystalline solid. In nature, it appears as the very rare mineral strontiofluorite.[2][3]
Preparation
Strontium fluoride is prepared by the action of hydrofluoric acid on strontium carbonate.[4]
Structure
The solid adopts the fluorite structure. In the vapour phase the SrF2 molecule is non-linear with an F−Sr−F angle of approximately 120°.[5] This is an exception to VSEPR theory which would predict a linear structure. Ab initio calculations have been cited to propose that contributions from d orbitals in the shell below the valence shell are responsible.[6] Another proposal is that polarization of the electron core of the strontium atom creates an approximately tetrahedral distribution of charge that interacts with the Sr−F bonds.[7]
Properties
It is almost insoluble in water (its Ksp value is approximately 2.0x10−10 at 25 degrees Celsius).
It irritates eyes and skin, and is harmful when inhaled or ingested.
Similar to CaF2 and BaF2, SrF2 displays superionic conductivity at elevated temperatures.[8]
Strontium fluoride is transparent to light in the wavelengths from vacuum ultraviolet (150 nm) to infrared (11 μm). Its optical properties are intermediate to calcium fluoride and barium fluoride.[9]
Uses
Strontium fluoride is used as an optical material for a small range of special applications, for example, as an optical coating on lenses and also as a thermoluminescent dosimeter crystal.
Another use is as a carrier of strontium-90 radioisotope in radioisotope thermoelectric generators.
References
- ↑ John Rumble (June 18, 2018) (in English). CRC Handbook of Chemistry and Physics (99 ed.). CRC Press. pp. 5–189. ISBN 978-1138561632.
- ↑ "Strontiofluorite". https://www.mindat.org/min-39548.html.
- ↑ "List of Minerals". 21 March 2011. https://www.ima-mineralogy.org/Minlist.htm.
- ↑ W. Kwasnik (1963). "Strontium Fluoride". in G. Brauer. Handbook of Preparative Inorganic Chemistry, 2nd Ed.. 1. NY, NY: Academic Press. pp. 234.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Ab initio model potential study of the equilibrium geometry of alkaline earth dihalides: MX2 (M = Mg, Ca, Sr, Ba; X=F, Cl, Br, I)Seijo L.,Barandiarán Z J. Chem. Phys. 94, 3762 (1991) doi:10.1063/1.459748
- ↑ Core Distortions and Geometries of the Difluorides and Dihydrides of Ca, Sr, and Ba Bytheway I, Gillespie RJ, Tang TH, Bader RF Inorganic Chemistry, Vol.34, No.9, 2407-2414, 1995 doi:10.1021/ic00113a023
- ↑ "Newmet Koch - Strontium". http://www.newmet.co.uk/Products/koch/strontium.php.
- ↑ Mediatopia Ltd. "Strontium Fluoride SrF2". Crystran.com. https://www.crystran.com/optical-materials/strontium-fluoride-srf2.
 |
