Chemistry:Ruthenium hexafluoride
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
ruthenium(VI) fluoride
| |||
| Other names
ruthenium(6+) hexafluoride
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| |||
| |||
| Properties | |||
| RuF6 | |||
| Molar mass | 215.07 g/mol | ||
| Appearance | dark brown crystalline solid[1] | ||
| Density | 3.54 g/cm3 | ||
| Melting point | 54 °C (129 °F; 327 K)[1] | ||
| Boiling point | 200 °C (392 °F, 473.15 K) (decomposes) [2] | ||
| reacts | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Ruthenium hexafluoride, also ruthenium(VI) fluoride (RuF6), is a compound of ruthenium and fluorine and one of the seventeen known binary hexafluorides.
History and synthesis
Ruthenium hexafluoride was discovered by American radiochemists in 1961, soon after the discovery of technetium hexafluoride.[3] It is made by a direct reaction of ruthenium metal in a gas stream of fluorine and argon at 400–450 °C. The yields of this reaction are less than 10%.[4]
- Ru + 3 F2 → RuF6
Description
Ruthenium hexafluoride is a dark brown crystalline solid that melts at 54 °C.[1] The solid structure measured at −140 °C is orthorhombic space group Pnma. Lattice parameters are a = 9.313 Å, b = 8.484 Å, and c = 4.910 Å. There are four formula units (in this case, discrete molecules) per unit cell, giving a density of 3.68 g·cm−3.[4]
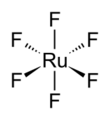
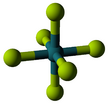
The RuF6 molecule itself (the form important for the liquid or gas phase) has octahedral molecular geometry, which has point group (Oh). The Ru–F bond length is 1.818 Å.[4]
References
- ↑ 1.0 1.1 1.2 CRC Handbook of Chemistry and Physics, 90th Edition, CRC Press, Boca Raton, Florida, 2009, ISBN 978-1-4200-9084-0, Section 4, Physical Constants of Inorganic Compounds, p. 4-85.
- ↑ Haynes, William M (2014-06-04). CRC Handbook of Chemistry and Physics, 95th Edition. CRC Press. ISBN 9781482208689. https://books.google.com/books?id=bNDMBQAAQBAJ.
- ↑ Claassen, Howard H.; Selig, Henry; Malm, John G.; Chernick, Cedric L.; Weinstock, Bernard (1961). "RUTHENIUM HEXAFLUORIDE1". Journal of the American Chemical Society 83 (10): 2390–2391. doi:10.1021/ja01471a039. ISSN 0002-7863. Bibcode: 1961JAChS..83.2390C. https://pubs.acs.org/doi/10.1021/ja01471a039.
- ↑ 4.0 4.1 4.2 Drews, T.; Supeł, J.; Hagenbach, A.; Seppelt, K. (2006). "Solid State Molecular Structures of Transition Metal Hexafluorides". Inorganic Chemistry 45 (9): 3782–3788. doi:10.1021/ic052029f. PMID 16634614.
Further reading
- Gmelins Handbuch der anorganischen Chemie, System Nr. 63, Ruthenium, Supplement, pp. 266–268.
External links
 |