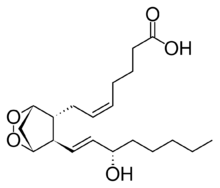
Chemistry:Prostaglandin H2
| |
| Names | |
|---|---|
| Other names
PGH2, Endoperoxide H2, Prostaglandin R2
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| MeSH | Prostaglandin+H2 |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C20H32O5 | |
| Molar mass | 352.465 g/mol |
| Density | 1.129 ± 0.06 g/mL |
| Boiling point | 490 ± 40.0 °C |
| 0.034 g/L | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Prostaglandin H2 (PGH2), or prostaglandin H2 (PGH2), is a type of prostaglandin and a precursor for many other biologically significant molecules. It is synthesized from arachidonic acid in a reaction catalyzed by a cyclooxygenase enzyme.[2] The conversion from arachidonic acid to prostaglandin H2 is a two-step process. First, COX-1 catalyzes the addition of two free oxygens to form the 1,2-dioxane bridge and a peroxide functional group to form prostaglandin G2 (PGG2).[3] Second, COX-2 reduces the peroxide functional group to a secondary alcohol, forming prostaglandin H2. Other peroxidases like hydroquinone have been observed to reduce PGG2 to PGH2.[4] PGH2 is unstable at room temperature, with a half life of 90–100 seconds,[1] so it is often converted into a different prostaglandin. PGH2 is produced by every type of cell except for red blood cells and has a wide range of effects in the body.[5]
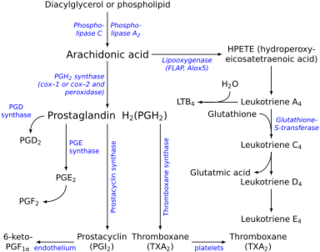
It is acted upon by:
- prostacyclin synthase to create prostacyclin
- thromboxane-A synthase to create thromboxane A2 and 12-(S)-hydroxy-5Z,8E,10E-heptadecatrienoic acid (HHT) (see 12-Hydroxyheptadecatrienoic acid)
- prostaglandin D2 synthase to create prostaglandin D2
- prostaglandin E synthase to create prostaglandin E2
- prostaglandin F synthase to create prostaglandin F2α[6]
It rearranges non-enzymatically to:
- A mixture of 12-(S)-hydroxy-5Z,8E,10E-heptadecatrienoic acid (HHT) and 12-(S)-hydroxy-5Z,8Z,10E-heptadecatrienoic acid (see 12-hydroxyheptadecatrienoic acid)
- These breakdown products are associated with increased aggregation of Amyloid beta peptides and Alzheimer's disease.[7]
Functions of prostaglandin H2:
- regulating the constriction and dilation of blood vessels
- stimulating platelet aggregation
- binds to thromboxane receptor on platelets' cell membranes to trigger platelet migration and adhesion to other platelets[8]
Effects of aspirin on prostaglandin H2:
- Aspirin has been hypothesized to block the conversion of arachidonic acid to prostaglandin

History
Prostaglandin H2 was discovered in 1973 by Diederik H. Nugteren and Elly Christ-Hazelhof while they were researching the formation of prostaglandin E2 from arachidonic acid using enzymes found in vesicular glands.[9]
Synthesis
The original synthesis of prostaglandin H2 by Diederik H. Nugteren and Elly Christ-Hazelhof was performed in 1973.[9] Sheep vesicular glands were homogenized with 1M KH2PO4 and 0.001 M EDTA buffer and then centrifuged to isolate the COX-1 enzymes. Pure arachidonic acid was added to a solution containing the enzymes, and the mixture was shaken. Thin-layer chromatography was used to isolate a band of prostaglandin H2.
In 1986, due to low prostaglandin H2 product purity from thin-layer chromatography and column chromatography, high-performance liquid chromatography with hexane and isopropanol as solvents was developed as an alternative means of isolating the prostaglandin with 98% purity.[10]
References
- ↑ 1.0 1.1 Wishart, David S.; Djombou Feunang, Yannick; Marcu, Ana; Guo, An Chi; Liang, Kevin; Vázquez Fresno, Rosa; Sajed, Tanvir; Johnson, Daniel et al.. "Showing metabocard for Prostaglandin H2 (HMDB0001381)". http://www.hmdb.ca/metabolites/HMDB0001381.
- ↑ "The cyclooxygenase reaction mechanism". Biochemistry 41 (52): 15451–8. December 2002. doi:10.1021/bi026938h. PMID 12501173.
- ↑ Salomon, Robert G.; Miller, Donald B.; Zagorski, Michael G.; Coughlin, Daniel J. (October 1984). "Prostaglandin endoperoxides. 14. Solvent-induced fragmentation of prostaglandin endoperoxides. New aldehyde products from PGH2 and a novel intramolecular 1,2-hydride shift during endoperoxide fragmentation in aqueous solution". Journal of the American Chemical Society 106 (20): 6049–6060. doi:10.1021/ja00332a049. ISSN 0002-7863.
- ↑ "Human cyclooxygenase-2 cDNA". Proceedings of the National Academy of Sciences of the United States of America 89 (16): 7384–8. August 1992. doi:10.1073/pnas.89.16.7384. PMID 1380156. Bibcode: 1992PNAS...89.7384H.
- ↑ Miller, Stephen B. (2006-08-01). "Prostaglandins in Health and Disease: An Overview". Seminars in Arthritis and Rheumatism 36 (1): 37–49. doi:10.1016/j.semarthrit.2006.03.005. ISSN 0049-0172. https://www.sciencedirect.com/science/article/pii/S0049017206000497.
- ↑ Hirata, Takako; Narumiya, Shuh (2011-08-05). "Prostanoid Receptors" (in EN). doi:10.1021/cr200010h. https://pubs.acs.org/doi/pdf/10.1021/cr200010h.
- ↑ Boutaud, Olivier; Ou, Joyce J.; Chaurand, Pierre; Caprioli, Richard M.; Montine, Thomas J.; Oates, John A. (2002). "Prostaglandin H2 (PGH2) accelerates formation of amyloid β1−42 oligomers" (in en). Journal of Neurochemistry 82 (4): 1003–1006. doi:10.1046/j.1471-4159.2002.01064.x. ISSN 1471-4159. https://onlinelibrary.wiley.com/doi/abs/10.1046/j.1471-4159.2002.01064.x.
- ↑ "International Union of Basic and Clinical Pharmacology. LXXXIII: classification of prostanoid receptors, updating 15 years of progress". Pharmacological Reviews 63 (3): 471–538. September 2011. doi:10.1124/pr.110.003517. PMID 21752876.
- ↑ 9.0 9.1 Nugteren, D. H.; Hazelhof, E. (1973-12-20). "Isolation and properties of intermediates in prostaglandin biosynthesis". Biochimica et Biophysica Acta (BBA) - Lipids and Lipid Metabolism 326 (3): 448–461. doi:10.1016/0005-2760(73)90145-8. ISSN 0005-2760. https://www.sciencedirect.com/science/article/pii/0005276073901458.
- ↑ Zulak, I. M.; Puttemans, M. L.; Schilling, A. B.; Hall, E. R.; Venton, D. L. (1986-04-01). "A fast, nondestructive purification scheme for prostaglandin H2 using a nonaqueous, bonded-phase high-performance liquid chromatography system". Analytical Biochemistry 154 (1): 152–161. doi:10.1016/0003-2697(86)90509-9. ISSN 0003-2697. https://www.sciencedirect.com/science/article/pii/0003269786905099.
 |
