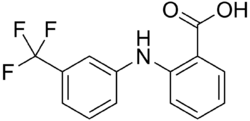
Chemistry:Flufenamic acid
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | extensively |
| Metabolism | Hydroxylation, glucuronidation |
| Elimination half-life | ~3 h |
| Excretion | 50% urine, 36% feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C14H10F3NO2 |
| Molar mass | 281.234 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 124 to 125 °C (255 to 257 °F) resolidification and remelting at 134°C to 136°C |
| Solubility in water | Practically insoluble in water; soluble in ethanol, chloroform and diethyl ether mg/mL (20 °C) |
| |
| |
| (verify) | |
Flufenamic acid (FFA) is a member of the anthranilic acid derivatives (or fenamate) class of nonsteroidal anti-inflammatory drugs (NSAIDs).[1]: 718 Like other members of the class, it is a cyclooxygenase (COX) inhibitor, preventing the formation of prostaglandins.[2] FFA is known to bind to and reduce the activity of prostaglandin F synthase and activate TRPC6.[3]
Scientists led by Claude Winder from Parke-Davis invented FFA in 1963, along with fellow members of the class, mefenamic acid in 1961 and meclofenamic acid in 1964.[1]: 718
Although flufenamic acid was at one time informally referred to as "Fluffy" (see history cache), this pet name could also refer to flufenoxine.
Structure
Flufenamic acid is a highly polymorphic drug molecule with multiple structurally characterized polymorphic modifications.[4] It has a unique chemical structure and stands out among fenamates.[5] Nowadays, eight polymorphic forms are known that are determined by different conformers,[6][7] which makes flufenamic acid unique among other low-molecular medicinal compounds.[8][9] A fundamental feature of the structure of flufenamic acid, which has generated significant interest in the design and development of drugs,[10] is the presence of a trifluoromethyl group. Compounds with fluorine-containing substituents are known to have promising chemical and biological properties,[11][12] since such groups often improve the pharmacokinetics and bioavailability of drugs.[13] Studies have shown the promise of repositioning flufenamic acid and the use of drugs based on it in the treatment of Bartter syndrome.
Medical uses
Until recently, FFA was actively used in medical practice as an analgesic with anti-inflammatory and antipyretic effects.[14] FFA has been proven effective in treating rheumatoid arthritis, osteoarthritis and other inflammation-related diseases.[15] However, despite this, the use of FFA in the United States and other countries [16] is limited since the compound causes frequent side effects. The rate of gastrointestinal side effects can be as high as 60%,[17] manifested as at least one of the following: dyspepsia, nausea, abdominal pain and discomfort, constipation, diarrhoea, flatulence, indigestion, epigastric distress, stomatitits and anorexia.[17] Besides gastrointestinal side effects, the drug can cause headache, dizziness and peripheral oedema.[17]
Side effects
It is not widely used in humans as it has a high rate (30–60%) of gastrointestinal side effects.[18] It is generally not available in the US.[2] It is available in some Asian and European countries as a generic drug.[19]
References
- ↑ 1.0 1.1 "Drugs to treat inflammation: a historical introduction". Current Medicinal Chemistry 12 (25): 2931–2942. 2005. doi:10.2174/092986705774462879. ISBN 978-1-60805-207-3. PMID 16378496.
- ↑ 2.0 2.1 "Mefenamic Acid". LiverTox Database (U.S. National Institutes of Health (NIH)). 23 June 2015. PMID 31643176. http://livertox.nih.gov/MefenamicAcid.htm. Retrieved 3 July 2015. "(fenamates generally not available in the United States, such as tolfenamic acid and flufenamic acid)".
- ↑ "Chemical–Gene Interaction Query: Flufenamic Acid (Homo sapiens)". Comparative Toxicogenomics Database. North Carolina State University. http://ctdbase.org/query.go?type=ixn&chemqt=equals&chem=name%3AFlufenamic+Acid&actionDegreeTypes=increases&actionDegreeTypes=decreases&actionDegreeTypes=affects&actionTypes=activity&actionTypes=binding&geneqt=equals&gene=&pathwayqt=equals&pathway=&taxonqt=equals&taxon=TAXON%3A9606&goqt=equals&go=&sort=chemNmSort&perPage=500&action=Search.
- ↑ "Application of the Method of Molecular Voronoi–Dirichlet Polyhedra for Analysis of Noncovalent Interactions in Crystal Structures of Flufenamic Acid—The Current Record-Holder of the Number of Structurally Studied Polymorphs" (in en). Crystal Growth & Design 15 (6): 2878–2882. June 3, 2015. doi:10.1021/acs.cgd.5b00326. ISSN 1528-7483. Bibcode: 2015CrGrD..15.2878S.
- ↑ "Tailor-Made Amino Acids and Fluorinated Motifs as Prominent Traits in Modern Pharmaceuticals". Chemistry: A European Journal 26 (50): 11349–11390. September 2020. doi:10.1002/chem.202000617. PMID 32359086. Bibcode: 2020ChEuJ..2611349M.
- ↑ "Investigation of the Spatial Structure of Flufenamic Acid in Supercritical Carbon Dioxide Media via 2D NOESY". Materials 16 (4): 1524. February 2023. doi:10.3390/ma16041524. PMID 36837153. Bibcode: 2023Mate...16.1524K.
- ↑ "Conformational State of Fenamates at the Membrane Interface: A MAS NOESY Study". Membranes 13 (6): 607. June 2023. doi:10.3390/membranes13060607. PMID 37367811.
- ↑ "Conformational origins of polymorphism in two forms of flufenamic acid" (in en). Journal of Molecular Structure 1078: 83–89. December 2014. doi:10.1016/j.molstruc.2014.02.001. Bibcode: 2014JMoSt1078...83D.
- ↑ "Nonamorphism in flufenamic acid and a new record for a polymorphic compound with solved structures". Journal of the American Chemical Society 134 (24): 9872–9875. June 2012. doi:10.1021/ja302601f. PMID 22690822. Bibcode: 2012JAChS.134.9872L.
- ↑ "Potent and selective aldo-keto reductase 1C3 (AKR1C3) inhibitors based on the benzoisoxazole moiety: application of a bioisosteric scaffold hopping approach to flufenamic acid". European Journal of Medicinal Chemistry 150: 930–945. April 2018. doi:10.1016/j.ejmech.2018.03.040. PMID 29602039.
- ↑ "Exploration of fluorine chemistry at the multidisciplinary interface of chemistry and biology". The Journal of Organic Chemistry 78 (13): 6358–6383. July 2013. doi:10.1021/jo400301u. PMID 23614876.
- ↑ "Synthetic chemistry and biological activity of pentafluorosulphanyl (SF5) organic molecules" (in en). Journal of Fluorine Chemistry 143: 57–93. November 2012. doi:10.1016/j.jfluchem.2012.06.030.
- ↑ "Pentafluorosulfanyl-containing flufenamic acid analogs: Syntheses, properties and biological activities". Bioorganic & Medicinal Chemistry Letters 25 (20): 4437–4440. October 2015. doi:10.1016/j.bmcl.2015.09.012. PMID 26372652.
- ↑ "Pharmaceutical Co-Crystal of Flufenamic Acid: Synthesis and Characterization of Two Novel Drug-Drug Co-Crystal". Journal of Pharmaceutical Sciences 106 (5): 1384–1390. May 2017. doi:10.1016/j.xphs.2017.01.033. PMID 28185907. Bibcode: 2017JPhmS.106.1384N.
- ↑ "The role of solid state properties on the dissolution performance of flufenamic acid". Journal of Pharmaceutical and Biomedical Analysis 180. February 2020. doi:10.1016/j.jpba.2019.113058. PMID 31881398.
- ↑ "The Latest FDA-Approved Pharmaceuticals Containing Fragments of Tailor-Made Amino Acids and Fluorine". Pharmaceuticals 15 (8): 999. August 2022. doi:10.3390/ph15080999. PMID 36015147.
- ↑ 17.0 17.1 17.2 "Flufenamic acid and meclofenamic acid". Meyler's Side Effects of Drugs (16th ed.). Elsevier. 2016. p. 361. doi:10.1016/B978-0-444-53717-1.00755-1. ISBN 978-0-444-53716-4. https://www.sciencedirect.com/science/article/abs/pii/B9780444537171007551.
- ↑ Aronson, Jeffrey K. (2009). Meyler's Side Effects of Analgesics and Anti-inflammatory Drugs. Elsevier. ISBN 978-0-08-093294-1.
- ↑ "International listings for flufenamic acid". Drugs.com. https://www.drugs.com/international/flufenamic-acid.html.
 |
