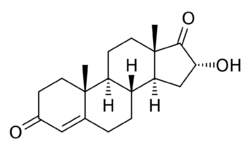
Chemistry:16α-Hydroxyandrostenedione
From HandWiki
| |
| Names | |
|---|---|
| IUPAC name
16α-Hydroxyandrost-4-ene-3,17-dione
| |
| Systematic IUPAC name
(2R,3aS,3bR,9aR,9bS,11aS)-2-Hydroxy-9a,11a-dimethyl-2,3,3a,4,5,8,9,9a,9b,10,11,11a-dodecahydro-1H-cyclopenta[a]phenanthrene-1,7(3bH)-dione | |
| Other names
16α-OH-A4
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C19H26O3 | |
| Molar mass | 302.414 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
16α-Hydroxyandrostenedione (16α-OH-A4), also known as 16α-hydroxyandrost-4-ene-3,17-dione, is an endogenous and naturally occurring steroid and metabolic intermediate in the biosynthesis of estriol during pregnancy.[1][2][3] It is produced from dehydroepiandrosterone (DHEA), which is converted into 16α-hydroxy-DHEA sulfate, then desulfated and aromatized into 16α-hydroxyestrone, and finally converted into estriol by 17β-hydroxysteroid dehydrogenase.[1][2]
See also
- 15α-Hydroxydehydroepiandrosterone
- Androstenedione
- 11β-Hydroxyandrostenedione
References
- ↑ 1.0 1.1 Rodney Rhoades; David R. Bell (2009). Medical Physiology: Principles for Clinical Medicine. Lippincott Williams & Wilkins. pp. 713–714. ISBN 978-0-7817-6852-8. https://books.google.com/books?id=tBeAeYS-vRUC&pg=PA713.
- ↑ 2.0 2.1 Charles Graham (2 December 2012). Reproductive Biology of the Great Apes: Comparative and Biomedical Perspectives. Elsevier. pp. 56–. ISBN 978-0-323-14971-6. https://books.google.com/books?id=iUA0CdGhYksC&pg=PA56.
- ↑ Vitamins and Hormones. Academic Press. 7 September 2005. pp. 282–. ISBN 978-0-08-045978-3. https://books.google.com/books?id=DUst5mwBfN0C&pg=PA282.
 |

