Chemistry:Androsterone glucuronide
| |
| Names | |
|---|---|
| IUPAC name
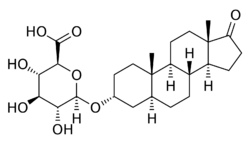
17-Oxo-5α-androstan-3α-yl β-D-glucopyranosiduronic acid
| |
| Systematic IUPAC name
(2S,3S,4S,5R,6R)-6-{[(3aS,3bR,5aS,7R,9aS,9bS,11aS)-9a,11a-Dimethyl-1-oxohexadecahydro-1H-cyclopenta[a]pheananthren-7-yl]oxy}-3,4,5-trihydroxyoxane-2-carboxylic acid | |
| Other names
ADT-G; 5α-Androstan-3α-ol-17-one 3-glucuronide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C25H38O8 | |
| Molar mass | 466.571 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Androsterone glucuronide (ADT-G) is a major circulating and urinary metabolite of testosterone and dihydrotestosterone (DHT).[1] It accounts for 93% of total androgen glucuronides in women.[1] ADT-G is formed from androsterone by UDP-glucuronosyltransferases, with the major enzymes being UGT2B15 and UGT2B17.[1] It is a marker of acne in women while androstanediol glucuronide is a marker of hirsutism (excess hair growth) in women.[2]
Relevance in women's health
Quantification of ADT-G and 3α-diol-G levels in Serum (blood) is an effective means of assessing androgen content in blood and androgenic activity in women.[3] Androsterone glucuronide content can be estimated using Liquid chromatography–mass spectrometry.[3][4] If an unusual level of ADT-G is observed in the blood (either an elevated or decreased amount), proper treatment plans can be developed in order to treat related symptoms.[3]
Elevated levels of androsterone glucuronide in the blood have been observed in adult females who present with inflammatory acne.[5] The introduction of therapeutic antiandrogen treatment and the use of an oral Contraceptive by women who are affected with androsterone related acne can decrease the amount of inflammatory acne present.[5]
See also
References
- ↑ 1.0 1.1 1.2 "Human Metabolome Database: Showing metabocard for Androsterone glucuronide (HMDB0002829)". http://www.hmdb.ca/metabolites/HMDB02829.
- ↑ Jerome F. Strauss, III; Robert L. Barbieri (13 September 2013). Yen and Jaffe's Reproductive Endocrinology. Elsevier Health Sciences. pp. 837–. ISBN 978-1-4557-2758-2. https://books.google.com/books?id=KZ95AAAAQBAJ&pg=PA837.
- ↑ 3.0 3.1 3.2 Labrie, Fernand; Bélanger, Alain; Bélanger, Patrick; Bérubé, René; Martel, Céline; Cusan, Leonello; Gomez, José; Candas, Bernard et al. (2006-06-01). "Androgen glucuronides, instead of testosterone, as the new markers of androgenic activity in women". The Journal of Steroid Biochemistry and Molecular Biology 99 (4): 182–188. doi:10.1016/j.jsbmb.2006.02.004. ISSN 0960-0760. PMID 16621522. https://www.sciencedirect.com/science/article/abs/pii/S0960076006000598.
- ↑ Ke, Yuyong; Gonthier, Renaud; Isabelle, Maxim; Bertin, Jonathan; Simard, Jean-Nicolas; Dury, Alain Y.; Labrie, Fernand (2015-05-01). "A rapid and sensitive UPLC–MS/MS method for the simultaneous quantification of serum androsterone glucuronide, etiocholanolone glucuronide, and androstan-3α, 17β diol 17-glucuronide in postmenopausal women". The Journal of Steroid Biochemistry and Molecular Biology 149: 146–152. doi:10.1016/j.jsbmb.2015.02.009. ISSN 0960-0760. PMID 25701608. https://www.sciencedirect.com/science/article/abs/pii/S0960076015000473.
- ↑ 5.0 5.1 Carmina, E.; Godwin, A. J.; Stanczyk, F. Z.; Lippman, J. S.; Lobo, R. A. (2002-10-01). "The association of serum androsterone glucuronide with inflammatory lesions in women with adult acne1" (in en). Journal of Endocrinological Investigation 25 (9): 765–768. doi:10.1007/BF03345509. ISSN 1720-8386. https://link.springer.com/article/10.1007/BF03345509.
External links
 |
