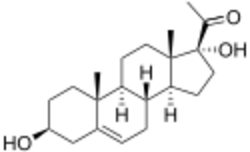
Chemistry:17α-Hydroxypregnenolone
 | |
 | |
| Pharmacokinetic data | |
|---|---|
| Metabolism | Adrenal, Gonads |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| Chemical and physical data | |
| Formula | C21H32O3 |
| Molar mass | 332.484 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 268 °C (514 °F) |
| |
| |
| | |
17α-Hydroxypregnenolone is a pregnane (C21) steroid that is obtained by hydroxylation of pregnenolone at the C17α position. This step is performed by the mitochondrial cytochrome P450 enzyme 17α-hydroxylase (CYP17A1) that is present in the adrenal and gonads. Peak levels are reached in humans at the end of puberty and then decline.[1] High levels are also achieved during pregnancy. It is also a known neuromodulator.
Prohormone
17α-Hydroxypregnenolone is considered a prohormone in the formation of dehydroepiandrosterone (DHEA), itself a prohormone of the sex steroids.
This conversion is mediated by the enzyme 17,20 lyase. As such 17α-hydroxypregenolone represents an intermediary in the Δ5 pathway that leads from pregnenolone to DHEA. 17α-Hydroxypregneolone is also converted to 17α-hydroxyprogesterone, a prohormone for glucocorticosteroids and androstenedione through the activity of 3α-hydroxysteroid dehydrogenase.
Clinical use
Measurements of 17α-hydroxypregnenolone are useful in the diagnosis of certain forms of congenital adrenal hyperplasia.[2] In patients with congenital adrenal hyperplasia due to 3β-hydroxysteroid dehydrogenase deficiency 17α-hydroxypregnenolone is increased, while in patients with congenital adrenal hyperplasia due to 17α-hydroxylase deficiency levels are low to absent.
Neurosteroid
17α-hydroxypregnenolone is a known neuromodulator as its acts in the central nervous system. Specifically, it is known to modulate locomotion.[3]
See also
- Congenital adrenal hyperplasia
- Narave pig, intersex pigs that have low levels of 17α-Hydroxypregnenolone
Additional images
References
- ↑ "Age relationships and sex differences in serum levels of pregnenolone and 17-hydroxypregnenolone in healthy subjects". Clinical Chemistry and Laboratory Medicine 37 (4): 439–47. April 1999. doi:10.1515/CCLM.1999.072. PMID 10369116.
- ↑ "Longitudinal study of plasma pregnenolone and 17-hydroxypregnenolone in full-term and preterm neonates at birth and during the early neonatal period". The Journal of Clinical Endocrinology and Metabolism 87 (9): 4301–6. September 2002. doi:10.1210/jc.2002-020452. PMID 12213889.
- ↑ "7α-Hydroxypregnenolone regulating locomotor behavior identified in the brain and pineal gland across vertebrates". General and Comparative Endocrinology 265: 97–105. September 2018. doi:10.1016/j.ygcen.2017.09.014. PMID 28919448.
 |