Chemistry:Manganese(III) fluoride
| |

| |
| Names | |
|---|---|
| IUPAC name
Manganese(III) fluoride
| |
| Other names
Manganese trifluoride, manganic fluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| MnF3 | |
| Molar mass | 111.938 g/mol |
| Appearance | purple-pink powder hygroscopic |
| Density | 3.54 g/cm3 |
| Melting point | > 600 °C (1,112 °F; 873 K) (decomposes) |
| hydrolysis | |
| +10,500·10−6 cm3/mol | |
| Structure | |
| Monoclinic, mS48 | |
| C2/c, No. 15 | |
| distorted octahedral | |
| Hazards | |
| Main hazards | toxic fumes |
| GHS pictograms |  
|
| GHS Signal word | danger |
| Related compounds | |
Other anions
|
manganese(III) oxide, manganese(III) acetate |
Other cations
|
chromium(III) fluoride, iron(III) fluoride. cobalt(III) fluoride |
Related compounds
|
manganese(II) fluoride, manganese(IV) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Manganese(III) fluoride (also known as Manganese trifluoride) is the inorganic compound with the formula MnF3. This red/purplish solid is useful for converting hydrocarbons into fluorocarbons, i.e., it is a fluorination agent.[2] It forms a hydrate and many derivatives.
Synthesis, structure and reactions
MnF3 can be prepared by treating a solution of MnF2 in hydrogen fluoride with fluorine:[3]
- MnF2 + 0.5 F2 → MnF3
It can also be prepared by the reaction of elemental fluorine with a manganese(II) halide at ~250 °C.[4]
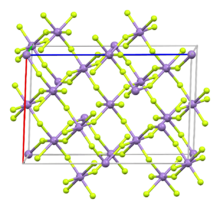
Structure
Like vanadium(III) fluoride, MnF3 features octahedral metal centers with the same average M-F bond distances. In the Mn compound, however, is distorted (and hence a monoclinic unit cell vs. a higher symmetry one) due to the Jahn-Teller effect, with pairs of Mn-F distances of 1.79, 1.91, 2.09 Å.[5][6][7]
The hydrate MnF3.3H2O is obtained by crystallisation of MnF3 from hydrofluoric acid. The hydrate exists as two polymorphs, with space groups P21/c and P21/a. Each consists of the salt [Mn(H2O)4F2]+[Mn(H2O)2F4]− ).[8]
Reactions
MnF3 is Lewis acidic and forms a variety of derivatives. One example is K2MnF3(SO4).[9] MnF3 reacts with sodium fluoride to give the octahedral hexafluoride:[4]
- 3NaF + MnF3 → Na3MnF6
Related reactions salts of the anions MnF52− or MnF4−. These anions adopt chain and layer structures respectively, with bridging fluoride. Manganese remains 6 coordinate, octahedral, and trivalent in all of these materials.[4]
Manganese(III) fluoride fluorinates organic compounds including aromatic hydrocarbons,[10] cyclobutenes,[11] and fullerenes.[12]
On heating, MnF3 decomposes to manganese(II) fluoride.[13][14]
MnF3 is a source of MnCl3 complexes by reaction with bismuth trichloride.[15]
See also
- CoF3, another fluorinating agent based on a transition metal in an oxidising +3 state.
References
- ↑ GHS: sigma-aldrich 339296 [|permanent dead link|dead link}}]
- ↑ Burley, G. A.; Taylor, R. (2004). "Manganese(III) Fluoride". Encyclopedia of Reagents for Organic Synthesis. J. Wiley & Sons. doi:10.1002/047084289X.rn00411.
- ↑ Z. Mazej (2002). "Room temperature syntheses of MnF3, MnF4 and hexafluoromanganete(IV) salts of alkali cations". Journal of Fluorine Chemistry 114 (1): 75–80. doi:10.1016/S0022-1139(01)00566-8.
- ↑ 4.0 4.1 4.2 Inorganic chemistry, Catherine E. Housecroft, A.G. Sharpe, pp.711-712, section Manganese (III) , googlebooks link
- ↑ Wells, A.F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press. ISBN:0-19-855370-6.
- ↑ Hepworth, M. A.; Jack, K. H.; Nyholm, R. S. (1957). "Interatomic Bonding in Manganese Trifluoride". Nature 179 (4552): 211–212. doi:10.1038/179211b0. Bibcode: 1957Natur.179..211H.
- ↑ M. A. Hepworth; K. H. Jack (1957). "The Crystal Structure of Manganese Trifluoride, MnF3". Acta Crystallographica 10 (5): 345–351. doi:10.1107/S0365110X57001024.
- ↑ Molinier Michel; Massa Werner (1992). "Structures of two polymorphs of MnF3·3H2O". Journal of Fluorine Chemistry 57 (1–3): 139–146. doi:10.1016/S0022-1139(00)82825-0.
- ↑ Bhattacharjee, M. N; Chaudhuri, M. K. (2007). "Dipotassium Trifluorosulfato‐Manganate(III)". Inorganic Syntheses. 27. 312–313. doi:10.1002/9780470132586.ch61. ISBN 978-0-470-13258-6.
- ↑ Fluorination of p-chlorobenzotrifluoride by manganese trifluoride A. Kachanov, V. Kornilov, V.Belogay , Fluorine Notes :Vol. 1 (1) November–December 1998 , via notes.fluorine1.ru
- ↑ Junji Mizukado; Yasuhisa Matsukawa; Heng-dao Quan; Masanori Tamura; Akira Sekiya (2006). "Fluorination of Fluoro-Cyclobutene with High-Valency Metal Fluoride". Journal of Fluorine Chemistry 127: 79–84. doi:10.1016/j.jfluchem.2005.10.007.
- ↑ V. É. Aleshina; A. Ya. Borshchevskii; E. V. Skokan; I. V. Arkhangel'skii; A.V. Astakhov; N.B. Shustova (2002). "Fluorination of the Cubic and Hexagonal C60 Modifications by Crystalline Manganese Trifluoride". Physics of the Solid State 44 (4): 629–630. doi:10.1134/1.1470543. Bibcode: 2002PhSS...44..629A.
- ↑ Chisholm, Hugh, ed (1911). "Manganese § Manganic Salts". Encyclopædia Britannica. 17 (11th ed.). Cambridge University Press. p. 570.
- ↑ In situ time-resolved X-ray diffraction study of manganese trifluoride thermal decomposition , J.V. Raua, V. Rossi Albertinib, N.S. Chilingarova, S. Colonnab, U. Anselmi Tamburini, Journal of Fluorine Chemistry 4506 (2001) 1–4 , online version
- ↑ Nachtigall, Olaf; Pataki, Astrid; Molski, Matthias; Lentz, Dieter; Spandl, Johann (2015). "Solvates of Manganese Trichloride Revisited - Synthesis, Isolation, and Crystal Structure of MnCl3(THF)3". Zeitschrift für Anorganische und Allgemeine Chemie 641 (6): 1164–1168. doi:10.1002/zaac.201500106.
Further reading
- Novel syntheses of some binary fluorides: the role of anhydrous hydrogen fluoride Acta Chim. Slov. 1999, 46(2), pp. 229–238, Zoran Mazej, Karel Lutar and Boris Žemva
- Knudsen Cell mass spectrometry study of Manganese Trifluoride vaporisation, High temperature corrosion and materials chemistry IV: proceedings of the International Symposium, pp. 521–525, google books
External links
- National Pollutant Inventory: Fluoride and compounds fact sheet
- National Pollutant Inventory: Manganese and compounds Fact Sheet
 |

