Chemistry:8-Phenyltheophylline
From HandWiki
Short description: Chemical compound
 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
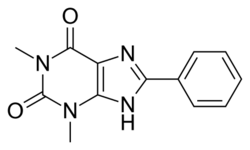
| Formula | C13H12N4O2 |
| Molar mass | 256.265 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
8-Phenyltheophylline (8-phenyl-1,3-dimethylxanthine, 8-PT) is a drug derived from the xanthine family which acts as a potent and selective antagonist for the adenosine receptors A1 and A2A, but unlike other xanthine derivatives has virtually no activity as a phosphodiesterase inhibitor.[1][2][3] It has stimulant effects in animals with similar potency to caffeine.[4] Coincidentally 8-phenyltheophylline has also been found to be a potent and selective inhibitor of the liver enzyme CYP1A2 which makes it likely to cause interactions with other drugs which are normally metabolised by CYP1A2.[5]
See also
- 8-Chlorotheophylline
- 8-Cyclopentyltheophylline
- DPCPX
- DMPX
- Xanthine
References
- ↑ "New derivatives of methyl-xanthines: effect of thiocaffeine thiotheophylline and 8-phenyltheophylline on lipolysis and on phosphodiesterase activities". Pharmacological Research Communications 15 (2): 131–43. February 1983. doi:10.1016/s0031-6989(83)80055-1. PMID 6844374.
- ↑ "Theophylline and selective PDE inhibitors as bronchodilators and smooth muscle relaxants". The European Respiratory Journal 8 (4): 637–42. April 1995. doi:10.1183/09031936.95.08040637. PMID 7664866.
- ↑ "Respiratory effects of xanthines and adenosine analogs in rhesus monkeys". The Journal of Pharmacology and Experimental Therapeutics 254 (3): 786–91. September 1990. PMID 2395111.
- ↑ "Psychomotor stimulant effects of methylxanthines in squirrel monkeys: relation to adenosine antagonism". Psychopharmacology 95 (1): 19–24. 1988. doi:10.1007/bf00212759. PMID 3133696.
- ↑ "Inhibition of human CYP1A2 activity in vitro by methylxanthines: potent competitive inhibition by 8-phenyltheophylline". Xenobiotica; the Fate of Foreign Compounds in Biological Systems 31 (3): 135–51. March 2001. doi:10.1080/00498250110043292. PMID 11465391.
 |
