Chemistry:Etazolate
From HandWiki
Short description: Chemical compound
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
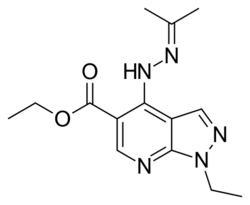
| Formula | C14H19N5O2 |
| Molar mass | 289.339 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Etazolate (SQ-20,009, EHT-0202) is an anxiolytic drug which is a pyrazolopyridine derivative and has unique pharmacological properties.[1][2][3] It acts as a positive allosteric modulator of the GABAA receptor at the barbiturate binding site,[4][5][6][7] as an adenosine antagonist of the A1 and A2 subtypes,[8] and as a phosphodiesterase inhibitor selective for the PDE4 isoform.[9][10][11] It is currently in clinical trials for the treatment of Alzheimer's disease.[12]
See also
References
- ↑ Concise dictionary of pharmacological agents: properties and synonyms. Kluwer Academic. 1999. ISBN 0-7514-0499-3. https://books.google.com/books?id=mqaOMOtk61IC&q=etazolate&pg=PA115.
- ↑ "Anxioselective anxiolytics". Journal of Medicinal Chemistry 26 (5): 619–628. May 1983. doi:10.1021/jm00359a001. PMID 6132997.
- ↑ "Enhancement of the binding of 3H-diazepam to rat brain membranes in vitro by SQ 20009, A novel anxiolytic, gamma-aminobutyric acid (GABA) and muscimol". Life Sciences 24 (9): 833–841. February 1979. doi:10.1016/0024-3205(79)90367-9. PMID 449623.
- ↑ "Interaction of allosteric ligands with GABAA receptors containing one, two, or three different subunits". European Journal of Pharmacology 301 (1–3): 207–214. April 1996. doi:10.1016/0014-2999(96)00066-0. PMID 8773466.
- ↑ "The Pharmacology of the Gamma-Aminobutyric Acid System". Brain mechanisms and psychotropic drugs. Boca Raton: CRC Press. 1996. ISBN 0-8493-8386-2. https://books.google.com/books?id=SPEIn0S6aK8C&q=etazolate&pg=PA109.
- ↑ "GABAA Receptor Chloride Ion Channels". Pharmacology of ionic channel function: activators and inhibitors. Handbook of Experimental Pharmacology. 147. Berlin: Springer. 2000. pp. 499–517. doi:10.1007/978-3-642-57083-4_19. ISBN 3-540-66127-1. https://books.google.com/books?id=t7i4yNiXiFMC&q=etazolate&pg=PA503.
- ↑ "GABA-Drug Interactions". Progress in Drug Research. 31. Boston: Birkhauser. 1987. p. 526. ISBN 3-7643-1837-6. https://books.google.com/books?id=gx6dwxmlQwMC&q=etazolate&pg=PA225.
- ↑ "Adenosine antagonists as potential therapeutic agents". Pharmacology, Biochemistry, and Behavior 29 (2): 433–441. February 1988. doi:10.1016/0091-3057(88)90182-7. PMID 3283781.
- ↑ "1-Ethyl-4-(isopropylidenehydrazino)-1H-pyrazolo-(3,4-b)-pyridine-5-carboxylic acid, ethyl ester, hydrochloride (SQ 20009)--a potent new inhibitor of cyclic 3',5'-nucleotide phosphodiesterases". Biochemical Pharmacology 21 (18): 2443–2450. September 1972. doi:10.1016/0006-2952(72)90414-5. PMID 4345859.
- ↑ "Expression, purification, and characterization of human cAMP-specific phosphodiesterase (PDE4) subtypes A, B, C, and D". Biochemical and Biophysical Research Communications 234 (2): 320–324. May 1997. doi:10.1006/bbrc.1997.6636. PMID 9177268.
- ↑ "Platelet signalling; cAMP and cGMP". Platelets in thrombotic and non-thrombotic disorders: pathophysiology, pharmacology and therapeutics. Cambridge, UK: Cambridge University Press. 2002. ISBN 0-521-80261-X. https://books.google.com/books?id=W1O-rgRYSdEC&q=etazolate+%22PDE4%22&pg=PA294.
- ↑ "EHT 0202". Pipeline. ExonHit. http://www.exonhit.com/therapeutics/pipeline#eht_0202.
 |
