Chemistry:Caffeine citrate
 | |
| Clinical data | |
|---|---|
| Trade names | Cafcit, Gencebok, Cafnea, others |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | By mouth, intravenous (IV) |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
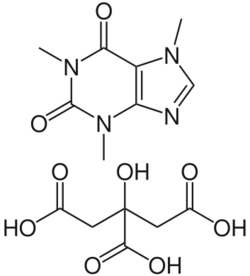
| Formula | C14H18N4O9 |
| Molar mass | 386.317 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Caffeine citrate, sold under the brand name Cafcit among others, is a medication used to treat a lack of breathing in premature babies.[5] Specifically it is given to babies who are born at less than 35 weeks or weigh less than 2 kilograms (4.4 lb) once other causes are ruled out.[6] It is given by mouth or slow injection into a vein.[5]
Side effects can include problems feeding, increased heart rate, low blood sugar, necrotizing enterocolitis, and kidney problems.[5][6] Testing blood caffeine levels is occasionally recommended.[5] Although it is often referred to as a citric acid salt of caffeine,[7] as implied by its name, caffeine citrate in fact consists of cocrystals of the two components.[8] Caffeine citrate is in the xanthine family of medication.[6] It works by stimulating the respiratory centers in the brain.[5]
Although caffeine was isolated in 1819,[9] it was not until 1977 that caffeine citrate was first used for treating apnea of prematurity.[10][11] It is on the World Health Organization's List of Essential Medicines.[12] The intravenous form may also be taken by mouth.[13]
In June 2020, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) recommended the approval of Gencebok.[14] It was approved for use in the European Union in August 2020.[3]
Medical uses
Caffeine citrate is generally the preferred treatment for apnea of prematurity for infants born 28 to 32 weeks or earlier than 28 weeks.[6][15] It has fewer side effects as compared to theophylline.[6]
Caffeine improves airway function in asthma, increasing forced expiratory volume (FEV1) by 5% to 18%, with this effect lasting for up to four hours.[16]
Mechanism
In method of action, the preparation is identical to that of caffeine base as the citrate counter ion dissociates in water. Doses of caffeine citrate, due to the added weight of the citrate moiety, are understandably higher than with caffeine base, i.e., it takes a larger dose to get the same amount of caffeine.[13] The ratio of therapeutic doses of caffeine base to its citrate salt is typically 1:2.[13] Dosing should therefore be clearly distinguished.[13]
Manufacture
The drug is prepared by combining anhydrous caffeine with citric acid monohydrate and sodium citrate dihydrate. Caffeine citrate cocrystals can take on at least two anhydrous polymorphs.[8]
References
- ↑ "Regulatory Decision Summary - Peyona". 23 October 2014. https://hpr-rps.hres.ca/reg-content/regulatory-decision-summary-detail.php?linkID=RDS00634.
- ↑ "Cafcit- caffeine citrate injection". 3 January 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=21489401-f2a7-47d3-897a-4b7efa04dd7a.
- ↑ 3.0 3.1 "Gencebok EPAR". 19 June 2020. https://www.ema.europa.eu/en/medicines/human/EPAR/gencebok.
- ↑ "Gencebok Product information". https://ec.europa.eu/health/documents/community-register/html/h1465.htm.
- ↑ 5.0 5.1 5.2 5.3 5.4 "Caffeine; Caffeine and Sodium Benzoate Injection; Caffeine Citrate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/caffeine-caffeine-and-sodium-benzoate-injection-caffeine-citrate.html.
- ↑ 6.0 6.1 6.2 6.3 6.4 WHO Model Formulary 2008. World Health Organization. 2009. p. 485. ISBN 978-92-4-154765-9.
- ↑ (in en) Manual of Neonatal Respiratory Care. Springer Science & Business Media. 2012. p. 457. ISBN 978-1-4614-2155-9. https://books.google.com/books?id=9rnVSufZFFwC&pg=PA457.
- ↑ 8.0 8.1 "Polymorphism in Caffeine Citric Acid Cocrystals". Journal of Chemical Crystallography 45 (3): 128–133. 1 March 2015. doi:10.1007/s10870-015-0573-3. ISSN 1572-8854. Bibcode: 2015JCCry..45..128S.
- ↑ (in en) In Silico Medicinal Chemistry: Computational Methods to Support Drug Design. Royal Society of Chemistry. 2015. p. 20. ISBN 978-1-78262-163-8. https://books.google.com/books?id=kLfbCgAAQBAJ&pg=PA20.
- ↑ "Caffeine citrate - Is it a silver bullet in neonatology?". Pediatrics and Neonatology 58 (5): 391–397. October 2017. doi:10.1016/j.pedneo.2016.10.003. PMID 28446386.
- ↑ "Efficacy of caffeine in treatment of apnea in the low-birth-weight infant". The Journal of Pediatrics 90 (3): 467–472. March 1977. doi:10.1016/S0022-3476(77)80718-X. PMID 14241.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ 13.0 13.1 13.2 13.3 (in en) Neonatal Formulary: Drug Use in Pregnancy and the First Year of Life (7 ed.). John Wiley & Sons. 2014. p. 120. ISBN 978-1-118-81951-7. https://books.google.com/books?id=EORvBAAAQBAJ&pg=PA120.
- ↑ "Gencebok: Pending EC decision". 25 June 2020. https://www.ema.europa.eu/en/medicines/human/summaries-opinion/gencebok.
- ↑ "NIH-funded research leads to pediatric labeling updates for doxycycline, clindamycin and caffeine citrate" (in EN). 2 April 2020. https://www.nih.gov/news-events/news-releases/nih-funded-research-leads-pediatric-labeling-updates-doxycycline-clindamycin-caffeine-citrate.
- ↑ "Caffeine for asthma". The Cochrane Database of Systematic Reviews 2010 (1). January 2010. doi:10.1002/14651858.CD001112.pub2. PMID 20091514.
External links
- "Caffeine Citrate (CUI C0054436)". https://ncimeta.nci.nih.gov/ncimbrowser/ConceptReport.jsp?dictionary=NCI%20MetaThesaurus&code=C0054436.
 |
