Chemistry:Fipexide
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
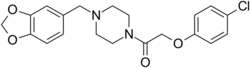
| Formula | C20H21ClN2O4 |
| Molar mass | 388.85 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Fipexide (Attentil, Vigilor) is a psychoactive drug of the piperazine chemical class which was developed in Italy in 1983.[1] It was used as a nootropic drug in Italy and France , mainly for the treatment of senile dementia,[2] but is no longer in common use due to the occurrence of rare adverse drug reactions including fever[3] and hepatitis. Fipexide is similar in action to other nootropic drugs such as piracetam[citation needed] and has a few similarities in chemical structure to centrophenoxine. Chemically, it is an amide union of parachlorophenoxyacetate and methylenedioxybenzylpiperazine (MDBZP), and has been shown to metabolize to the latter, which plays a significant role in its effects[citation needed].
Synthesis

PTC alkylation of piperazine (1) with 2 equivalents of piperonyl chloride [25054-53-9] (2) in the presence of cetrimonium bromide gives 1,4-bis-piperonylpiperazine [55436-41-4] (3). Base catalyzed treatment with 4-Chlorophenoxyacetic acid (4) displaces one of the piperonyl groups to give fipexide (5).
See also
References
- ↑ "[Fipexide: a new drug for the regulation of dopaminergic system at the macromolecular level]" (in it). Bollettino Chimico Farmaceutico 122 (2): 79–85. February 1983. PMID 6871040.
- ↑ "Fipexide, an effective cognition activator in the elderly: a placebo-controlled, double-blind clinical trial". Current Medical Research and Opinion 10 (2): 99–106. 1986. doi:10.1185/03007998609110426. PMID 3519097.
- ↑ "[Fever caused by fipexide. Evaluation of the national pharmacovigilance survey]" (in fr). Therapie 45 (5): 429–31. 1990. PMID 2260037.
- ↑ Gardini, G. P.; Palla, G.; Scapini, G.; Cesaroni, M. R. (2006). "Convenient Synthesis of N-Benzyl-N′-acyl-piperazines". Synthetic Communications. 12 (11): 887–890. ISSN 0039-7911. doi:10.1080/00397918208065967.
- ↑ Anon., FR patent 7524M (1969-12-15).
- ↑ Gian P. Gardini, Giancarlo Scapini, Armando Raimondi, Placido Poidomani, U.S. Patent 4,225,714 (1980 to Farmaceutici Geymonat Sud S.P.A.).
 |
