Chemistry:Doxofylline
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
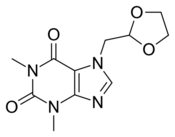
| Formula | C11H14N4O4 |
| Molar mass | 266.257 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Doxofylline (also known as doxophylline) is a phosphodiesterase inhibiting bronchodilator used in the treatment of chronic respiratory diseases such as asthma[1] and COPD.[2] Like theophylline, it is a xanthine derivative.[3][4]
Medical uses
Doxophylline is used to treat chronic respiratory diseases such as asthma[1] and COPD.[2]
In animal and human studies, it has shown similar efficacy to theophylline but with significantly fewer side effects.[5] In February 2014, the US FDA granted an orphan drug designation to doxofylline for the treatment of bronchiectasis following the submission of an application by Alitair Pharmaceuticals, in May 2013.[6][7][8]
Pharmacology
Unlike other xanthines, doxofylline lacks any significant affinity for adenosine receptors and does not produce stimulant effects. This suggests that its antiasthmatic effects are mediated by another mechanism, perhaps its actions on phosphodiesterase.[1] From a pharmacokinetic point of view, doxofylline importantly differs from theophylline also because it lacks the ability to interfere with the cytochrome enzymes CYP1A2, CYP2E1 and CYP3A4, thus preventing significant interaction with other drugs metabolized via these pathways in the liver.[9][10]
Concomitant treatment with certain other medications (including allopurinol, H2 receptor antagonists, lincosamide antibiotics, macrolide antibiotics, and propranolol) can decrease the hepatic clearance of doxofylline, which can result in increased serum levels of doxofylline.
Names
It is marketed under many brand names worldwide, including: Xiva, An Li Nuo Er, An Sai Ma, Ansimar, Asima, Bestofyline, Chuan Ning, D-Fyal, Dilatair, Doxiba, Doxiva, Doxobid, Doxobron, Doxofilina, Doxofillina, Doxofyllin, Doxoll, Doxophylline, Doxovent, Doxyjohn, Fei Te Ai Si,Fixolin,Jian Fang Neng, Lang Ming, Lv Meng, Mai Ping Xi, Maxivent, Mucosma, Na De Lai, Phylex, Phyllin, Puroxan, Rexipin, Shu Zhi, Shuai An, Shuweixin, Suo Di, Suo Ji, Suo Li An, Xi Si Nuo, Xin Qian Ping, Xin Xi Ping, Yi Suo, and Yili.[11]
It is also marketed as a combination drug with terbutaline as Doxoll-TL, Mucosma-T and Phylex-TR.[11] It is also marketed as a combination drug with montelukast as Doxoll-ML, Doxomont, Doxoril-M, Doxovent-M, Lunair-M, and Venidox-M.[11]
References
- ↑ 1.0 1.1 1.2 "Doxofylline, an antiasthmatic drug lacking affinity for adenosine receptors". Archives Internationales de Pharmacodynamie et de Therapie 295: 221–37. 1988. PMID 3245738.
- ↑ 2.0 2.1 "Impact of doxofylline in COPD: A pairwise meta-analysis". Pulmonary Pharmacology & Therapeutics 51: 1–9. August 2018. doi:10.1016/j.pupt.2018.04.010. PMID 29705620. https://kclpure.kcl.ac.uk/portal/files/97650305/Impact_of_doxofylline_in_CAZZOLA_Accepted24April2018_GOLD_VOR_CC_BY_.pdf.
- ↑ "Doxofylline: a new generation xanthine bronchodilator devoid of major cardiovascular adverse effects". Current Medical Research and Opinion 16 (4): 258–68. 2001. doi:10.1185/030079901750120196. PMID 11268710.
- ↑ "Doxofylline and respiratory mechanics. Short-term effects in mechanically ventilated patients with airflow obstruction and respiratory failure". Chest 96 (4): 772–8. October 1989. doi:10.1378/chest.96.4.772. PMID 2791671. http://www.chestjournal.org/cgi/pmidlookup?view=long&pmid=2791671.
- ↑ "Doxofylline: The next generation methylxanthine". Indian Journal of Pediatrics 75 (3): 251–4. March 2008. doi:10.1007/s12098-008-0054-1. PMID 18376093.
- ↑ "Orphan Drug Designations and Approvals List as of 12‐01‐2014". https://www.hrsa.gov/sites/default/files/opa/programrequirements/orphandrugexclusion/janmar2015list.pdf.
- ↑ SMI Support. "News Release 10/26/15" (in en-gb). http://www.alitair.com/index.php/news-release-10-26-15.
- ↑ "Doxofylline - AdisInsight" (in en). https://adisinsight.springer.com/drugs/800003876.
- ↑ "Treatment plan comparison in acute and chronic respiratory tract diseases: an observational study of doxophylline vs. theophylline". Expert Review of Pharmacoeconomics & Outcomes Research 17 (5): 503–510. October 2017. doi:10.1080/14737167.2017.1301815. PMID 28277853. http://eprints.kingston.ac.uk/38771/1/Mennini-F-S-38771-AAM.pdf.
- ↑ "Doxofylline is not just another theophylline!". International Journal of Chronic Obstructive Pulmonary Disease 12: 3487–3493. 2017-12-05. doi:10.2147/COPD.S150887. PMID 29255355.
- ↑ 11.0 11.1 11.2 "Doxofylline - international brand names" (in en). Drugs.com. https://www.drugs.com/international/doxofylline.html.
Further reading
- "Doxofylline: a promising methylxanthine derivative for the treatment of asthma and chronic obstructive pulmonary disease". Expert Opinion on Pharmacotherapy 10 (14): 2343–56. October 2009. doi:10.1517/14656560903200667. PMID 19678793.
 |
