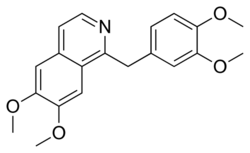
Chemistry:Papaverine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /pəˈpævəriːn/ |
| Trade names | Pavabid, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682707 |
| Pregnancy category |
|
| Routes of administration | Oral, intravenous, intramuscular, rectal, intracavernosal |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 80% |
| Protein binding | ~90% |
| Metabolism | Hepatic |
| Elimination half-life | 1.5–2 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C20H21NO4 |
| Molar mass | 339.391 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Papaverine (Latin papaver, "poppy") is an opium alkaloid antispasmodic drug, used primarily in the treatment of visceral spasms and vasospasms (especially those involving the intestines, heart, or brain), occasionally in the treatment of erectile dysfunction and acute mesenteric ischemia. While it is found in the opium poppy, papaverine differs in both structure and pharmacological action from the analgesic morphine and its derivatives (such as codeine).
In addition to opium, papaverine is purported to be present in high concentrations in star gooseberry.[1]
History

Papaverine was discovered in 1848 by Georg Merck (1825–1873).[2] Merck was a student of the German chemists Justus von Liebig and August Hofmann, and he was the son of Emanuel Merck (1794–1855), founder of the Merck corporation, a major German chemical and pharmaceutical company.[3]
Uses
Papaverine is approved to treat spasms of the gastrointestinal tract, bile ducts and ureter and for use as a cerebral and coronary vasodilator in subarachnoid hemorrhage (combined with balloon angioplasty)[4] and coronary artery bypass surgery.[5] Papaverine may also be used as a smooth muscle relaxant in microsurgery where it is applied directly to blood vessels.
Papaverine is used as an erectile dysfunction drug, alone or sometimes in combination.[6][7] Papaverine, when injected in penile tissue, causes direct smooth muscle relaxation and consequent filling of the corpus cavernosum with blood resulting in erection. A topical gel is also available for ED treatment.[8]
It is also commonly used in cryopreservation of blood vessels along with the other glycosaminoglycans and protein suspensions.[9][10] Functions as a vasodilator during cryopreservation when used in conjunction with verapamil, phentolamine, nifedipine, tolazoline or nitroprusside.[11][12]
Papaverine is also being investigated as a topical growth factor in tissue expansion with some success.[13]
Papaverine is used as an off-label prophylaxis (preventative) of migraine headaches.[14][15][16] It is not a first line drug such as a few beta blockers, calcium channel blockers, tricyclic antidepressants, and some anticonvulsants such as divalproex, but rather when these first line drugs and secondary drugs such as SSRIs, angiotensin II receptor antagonists, etc. fail in the prophylaxis of migraines, have intolerable side effects or are contraindicated.
Papaverine is also present in combinations of opium alkaloid salts such as papaveretum (Omnopon, Pantopon) and others, along with morphine, codeine, and in some cases noscapine and others in a percentage similar to that in opium, or modified for a given application.
Papaverine is found as a contaminant in some heroin[17] and can be used by forensic laboratories in heroin profiling to identify its source.[18] The metabolites can also be found in the urine of heroin users, allowing street heroin to be distinguished from pharmaceutical diacetylmorphine.[19]
Mechanism
The in vivo mechanism of action is not entirely clear, but an inhibition of the enzyme phosphodiesterase causing elevation of cyclic AMP and cyclic GMP[clarification needed] levels is significant. It may also alter mitochondrial respiration.
Papaverine has also been demonstrated to be a selective phosphodiesterase inhibitor for the PDE10A subtype found mainly in the striatum of the brain. When administered chronically to mice, it produced motor and cognitive deficits and increased anxiety, but conversely may produce an antipsychotic effect,[20][21] although not all studies support this view.[22]
Side effects
Frequent side effects of papaverine treatment include polymorphic ventricular tachycardia, constipation, interference with sulphobromophthalein[citation needed] retention test (used to determine hepatic function), increased transaminase levels, increased alkaline phosphatase levels, somnolence, and vertigo.
Rare side effects include flushing of the face, hyperhidrosis (excessive sweating), cutaneous eruption, arterial hypotension, tachycardia, loss of appetite, jaundice, eosinophilia, thrombopenia, mixed hepatitis, headache, allergic reaction, chronic active hepatitis, and paradoxical aggravation of cerebral vasospasm.[23]
Papaverine in the plant Sauropus androgynus is linked to bronchiolitis obliterans.[24]
Formulations and trade names
Papaverine is available in its salt form as the hydrochloride, codecarboxylate, adenylate, and teprosylate. It was also once available as a salt of hydrobromide, camsylate, cromesilate, nicotinate, and phenylglycolate. The hydrochloride salt is available for intramuscular, intravenous, rectal and oral administration. The teprosylate is available in intravenous, intramuscular, and orally administered formulations. The codecarboxylate is available in oral form, only, as is the adenylate.
The codecarboxylate is sold under the name Albatran,[citation needed] the adenylate as Dicertan,[citation needed] and the hydrochloride salt is sold variously as Artegodan (Germany), Cardioverina (countries outside Europe and the United States), Dispamil (countries outside Europe and the United States), Opdensit (Germany), Panergon (Germany), Paverina Houde (Italy, Belgium), Pavacap (United States), Pavadyl (United States), Papaverine (Israel), Papaverin-Hamelin (Germany), Paveron (Germany), Spasmo-Nit (Germany), Cardiospan, Papaversan, Cepaverin, Cerespan, Drapavel, Forpaven, Papalease, Pavatest, Paverolan, Therapav (Canada[25]), Vasospan, Cerebid, Delapav, Dilaves, Durapav, Dynovas, Optenyl, Pameion, Papacon, Pavabid, Pavacen, Pavakey, Pavased, Pavnell, Alapav, Myobid, Vasal, Pamelon, Pavadel, Pavagen, Ro-Papav, Vaso-Pav, Papanerin-hcl, Qua bid, Papital T.R., Paptial T.R., Pap-Kaps-150.[citation needed] In Hungary, papaverine and homatropine methylbromide are used in mild drugs that help "flush" the bile.[26]
References
- ↑ Kao CH; Ho YJ; Wu CL; ChangLai SP (1999). "Using 99mTc-DTPA Radioaerosol Inhalation Lung Scintigraphies to Detect the Lung Injury Induced by Consuming Sauropus androgynus Vegetable and Comparison with Conventional Pulmonary Function Tests". Respiration (Karger AG) 66 (1): 46–51. doi:10.1159/000029336. PMID 9973690.
- ↑ "Vorläufige Notiz über eine neue organische Base im Opium". Annalen der Chemie und Pharmacie 66: 125–128. 1848. doi:10.1002/jlac.18480660121. https://books.google.com/books?id=nDY9AAAAcAAJ&pg=PA125.
- ↑ (in en) Justus Von Liebig: The Chemical Gatekeeper. Cambridge University Press. 2002-06-20. p. 120. ISBN 978-0-521-52473-5. https://books.google.com/books?id=VugoemP2th0C&pg=PA120.
- ↑ "Intra-arterial papaverine infusions for the treatment of cerebral vasospasm induced by aneurysmal subarachnoid hemorrhage". Neurocritical Care 2 (2): 124–132. 2005. doi:10.1385/NCC:2:2:124. PMID 16159054.
- ↑ "Reactivity of the human internal thoracic artery to vasodilators in coronary artery bypass grafting". European Journal of Cardio-Thoracic Surgery 26 (5): 956–959. November 2004. doi:10.1016/j.ejcts.2004.07.047. PMID 15519189.
- ↑ "[An overview of the management of erectile disorders]". Presse Médicale 34 (13 Suppl): 5–7. July 2005. PMID 16158020.
- ↑ "Intracavernous pharmacotherapy for erectile dysfunction". Endocrine 23 (2–3): 149–155. 2004. doi:10.1385/ENDO:23:2-3:149. PMID 15146094.
- ↑ "Papaverine topical gel for treatment of erectile dysfunction". The Journal of Urology 153 (2): 361–365. February 1995. doi:10.1097/00005392-199502000-00019. PMID 7815584.
- ↑ "Sucrose promotes the functional activity of blood vessels after cryopreservation in DMSO-containing fetal calf serum". Naunyn-Schmiedeberg's Archives of Pharmacology 345 (5): 594–597. May 1992. doi:10.1007/bf00168954. PMID 1528275.
- ↑ "Cryopreservation of human bronchi". The Journal of Asthma 30 (6): 451–457. 1993. doi:10.3109/02770909309056754. PMID 8244915.
- ↑ "Effects of cryopreservation upon vein function in vivo". Cryobiology 31 (1): 71–81. February 1994. doi:10.1006/cryo.1994.1009. PMID 8156802.
- ↑ "Cryopreservation prevents arterial allograft dilation". Annals of Vascular Surgery 16 (6): 762–767. November 2002. doi:10.1007/s10016-001-0072-1. PMID 12391500.
- ↑ "Accelerating tissue expansion by application of topical papaverine cream". Plastic and Reconstructive Surgery 114 (5): 1166–1169. October 2004. doi:10.1097/01.PRS.0000135854.48570.76. PMID 15457029.
- ↑ "Papaverine in the prophylaxis of migraine and other vascular headache in children". Acta Paediatrica Scandinavica 67 (2): 209–212. March 1978. doi:10.1111/j.1651-2227.1978.tb16304.x. PMID 343489.
- ↑ "Brief therapeutic report: papaverine prophylaxis of complicated migraine". Headache 17 (4): 159–162. September 1977. doi:10.1111/j.1526-4610.1977.hed1704159.x. PMID 893088.
- ↑ "Letter: Papaverine in prophylactic treatment of migraine". Lancet 1 (7869): 1290. June 1974. doi:10.1016/S0140-6736(74)90045-2. PMID 4134173.
- ↑ "Comparison of the various opiate alkaloid contaminants and their metabolites found in illicit heroin with 6-monoacetyl morphine as indicators of heroin ingestion". Journal of Analytical Toxicology 30 (4): 267–273. May 2006. doi:10.1093/jat/30.4.267. PMID 16803666. "In addition to morphine, street heroin contains various alkaloids extracted from the opium poppy, Papaversomniferum, including codeine, thebaine, noscapine, and papaverine".
- ↑ "Biomarkers for illicit heroin: a previously unrecognized origin of papaverine". Journal of Analytical Toxicology 37 (2): 133. March 2013. doi:10.1093/jat/bks099. PMID 23316026.
- ↑ "Supervised injectable heroin or injectable methadone versus optimised oral methadone as treatment for chronic heroin addicts in England after persistent failure in orthodox treatment (RIOTT): a randomised trial". Lancet 375 (9729): 1885–1895. May 2010. doi:10.1016/s0140-6736(10)60349-2. PMID 20511018.
- ↑ "Inhibition of the striatum-enriched phosphodiesterase PDE10A: a novel approach to the treatment of psychosis". Neuropharmacology 51 (2): 386–396. August 2006. doi:10.1016/j.neuropharm.2006.04.013. PMID 16780899.
- ↑ "Phosphodiesterase 10A inhibition is associated with locomotor and cognitive deficits and increased anxiety in mice". European Neuropsychopharmacology 18 (5): 339–363. May 2008. doi:10.1016/j.euroneuro.2007.08.002. PMID 17913473.
- ↑ "Evaluating the antipsychotic profile of the preferential PDE10A inhibitor, papaverine". Psychopharmacology 203 (4): 723–735. May 2009. doi:10.1007/s00213-008-1419-x. PMID 19066855.
- ↑ "Paradoxical aggravation of vasospasm with papaverine infusion following aneurysmal subarachnoid hemorrhage. Case report". Journal of Neurosurgery 84 (4): 690–695. April 1996. doi:10.3171/jns.1996.84.4.0690. PMID 8613866.
- ↑ "Sauropus androgynus (L.) Merr. Induced Bronchiolitis Obliterans: From Botanical Studies to Toxicology". Evidence-Based Complementary and Alternative Medicine 2015: 714158. 2015. doi:10.1155/2015/714158. PMID 26413127.
- ↑ "THERAPAV (PRODUIT PUR) - Détail". http://www.reptox.csst.qc.ca/Produit.asp?no_produit=108688&nom=THERAPAV+(PRODUIT+PUR)&incr=0. CSST - Service du répertoire toxicologique. (French)
- ↑ "Országos Gyógyszerészeti és Élelmezés-egészségügyi Intézet". http://www.ogyi.hu/drug_database/index.php?action=show_details&item=21736.
 |
