Chemistry:Valine
| |||
|
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Valine
| |||
| Other names
2-Amino-3-methylbutanoic acid
2-Aminoisovaleric acid Valic acid | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI |
| ||
| ChEMBL |
| ||
| ChemSpider | |||
| DrugBank |
| ||
| EC Number |
| ||
| |||
| KEGG |
| ||
PubChem CID
|
|||
| UNII |
| ||
| |||
| |||
| Properties[3] | |||
| C5H11NO2 | |||
| Molar mass | 117.148 g·mol−1 | ||
| Density | 1.316 g/cm3 | ||
| Melting point | 298 °C (568 °F; 571 K) (decomposition) | ||
| soluble, 85 g/L [1] | |||
| Acidity (pKa) | 2.32 (carboxyl), 9.62 (amino)[2] | ||
| -74.3·10−6 cm3/mol | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
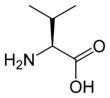
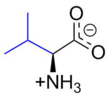
Valine (symbol Val or V)[4] is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −COO− form under biological conditions), and a side chain isopropyl group, making it a non-polar aliphatic amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Human dietary sources are foods that contain protein, such as meats, dairy products, soy products, beans and legumes. It is encoded by all codons starting with GU (GUU, GUC, GUA, and GUG).
History and etymology
Valine was first isolated from casein in 1901 by Hermann Emil Fischer.[5] The name valine comes from valeric acid, which in turn is named after the plant valerian due to the presence of the acid in the roots of the plant.[6][7]
Nomenclature
According to IUPAC, carbon atoms forming valine are numbered sequentially starting from 1 denoting the carboxyl carbon, whereas 4 and 4' denote the two terminal methyl carbons.[8]
Metabolism
Source and biosynthesis
Valine, like other branched-chain amino acids, is synthesized by bacteria and plants, but not by animals.[9] It is therefore an essential amino acid in animals, and needs to be present in the diet. Adult humans require about 24 mg/kg body weight daily.[10] It is synthesized in plants and bacteria via several steps starting from pyruvic acid. The initial part of the pathway also leads to leucine. The intermediate α-ketoisovalerate undergoes reductive amination with glutamate. Enzymes involved in this biosynthesis include:[11]
- Acetolactate synthase (also known as acetohydroxy acid synthase)
- Acetohydroxy acid isomeroreductase
- Dihydroxyacid dehydratase
- Valine aminotransferase
Degradation
Like other branched-chain amino acids, the catabolism of valine starts with the removal of the amino group by transamination, giving alpha-ketoisovalerate, an alpha-keto acid, which is converted to isobutyryl-CoA through oxidative decarboxylation by the branched-chain α-ketoacid dehydrogenase complex.[12] This is further oxidised and rearranged to succinyl-CoA, which can enter the citric acid cycle.
Synthesis
Racemic valine can be synthesized by bromination of isovaleric acid followed by amination of the α-bromo derivative[13]
- HO2CCH2CH(CH3)2 + Br2 → HO2CCHBrCH(CH3)2 + HBr
- HO2CCHBrCH(CH3)2 + 2 NH3 → HO2CCH(NH2)CH(CH3)2 + NH4Br
Medical significance
Metabolic diseases
The degradation of valine is impaired in the following metabolic diseases:
- Combined malonic and methylmalonic aciduria (CMAMMA)
- Maple syrup urine disease (MSUD)
- Methylmalonic acidemia
- Propionic acidemia
Insulin resistance
Lower levels of serum valine, like other branched-chain amino acids, are associated with weight loss and decreased insulin resistance: higher levels of valine are observed in the blood of diabetic mice, rats, and humans.[14] Mice fed a BCAA-deprived diet for one day had improved insulin sensitivity, and feeding of a valine-deprived diet for one week significantly decreases blood glucose levels.[15] In diet-induced obese and insulin resistant mice, a diet with decreased levels of valine and the other branched-chain amino acids resulted in a rapid reversal of the adiposity and an improvement in glucose-level control.[16] The valine catabolite 3-hydroxyisobutyrate promotes insulin resistance in mice by stimulating fatty acid uptake into muscle and lipid accumulation.[17] In mice, a BCAA-restricted diet decreased fasting blood glucose levels and improved body composition.[18]
Hematopoietic stem cells
Dietary valine is essential for hematopoietic stem cell (HSC) self-renewal, as demonstrated by experiments in mice.[19] Dietary valine restriction selectively depletes long-term repopulating HSC in mouse bone marrow. Successful stem cell transplantation was achieved in mice without irradiation after 3 weeks on a valine restricted diet. Long-term survival of the transplanted mice was achieved when valine was returned to the diet gradually over a 2-week period to avoid refeeding syndrome.
See also
References
- ↑ "Physicochemical Information". emdmillipore. 2022. https://www.emdmillipore.com/US/en/product/S-Valine,MDA_CHEM-816006.
- ↑ Data for Biochemical Research. Oxford: Clarendon Press. 1959. OCLC 859821178.
- ↑ Weast, Robert C., ed (1981). CRC Handbook of Chemistry and Physics (62nd ed.). Boca Raton, FL: CRC Press. p. C-569. ISBN 0-8493-0462-8.
- ↑ "Nomenclature and Symbolism for Amino Acids and Peptides". IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. http://www.chem.qmul.ac.uk/iupac/AminoAcid/AA1n2.html.
- ↑ "Valine". Encyclopædia Britannica Online. http://www.britannica.com/EBchecked/topic/622178/valine. Retrieved 2015-12-06.
- ↑ "Valine". Merriam-Webster Online Dictionary. http://www.merriam-webster.com/dictionary/valine.
- ↑ "Valeric acid". Merriam-Webster Online Dictionary. http://www.merriam-webster.com/dictionary/valeric+acid.
- ↑ Amino Acids, Peptides and Proteins. Specialist Periodical Reports. 16. London: Royal Society of Chemistry. 1985. p. 389. ISBN 978-0-85186-144-9.
- ↑ Nitrogen metabolism in rice. Boca Raton, Florida: CRC Press. 2016. pp. 159. ISBN 978-1-4987-4668-7. OCLC 945482059. https://books.google.com/books?id=shDYCwAAQBAJ&pg=PA159.
- ↑ Institute of Medicine (2002). "Protein and Amino Acids". Dietary Reference Intakes for Energy, Carbohydrates, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. Washington, DC: The National Academies Press. pp. 589–768. doi:10.17226/10490. ISBN 978-0-309-08537-3. https://www.nap.edu/read/10490/chapter/12.
- ↑ Lehninger, Albert L.; Nelson, David L.; Cox, Michael M. (2000). Principles of Biochemistry (3rd ed.). New York: W. H. Freeman. ISBN 1-57259-153-6..
- ↑ Biochemistry. Van Holde, K. E., Ahern, Kevin G. (3rd ed.). San Francisco, Calif.: Benjamin Cummings. 2000. pp. 776. ISBN 978-0-8053-3066-3. OCLC 42290721.
- ↑ "dl-Valine". Organic Syntheses 20: 106. 1940. http://www.orgsyn.org/demo.aspx?prep=CV3P0848.; Collective Volume, 3, pp. 848.
- ↑ "Branched-chain amino acids in metabolic signalling and insulin resistance". Nature Reviews. Endocrinology 10 (12): 723–36. December 2014. doi:10.1038/nrendo.2014.171. PMID 25287287.
- ↑ "Effects of individual branched-chain amino acids deprivation on insulin sensitivity and glucose metabolism in mice". Metabolism 63 (6): 841–50. June 2014. doi:10.1016/j.metabol.2014.03.006. PMID 24684822.
- ↑ "Restoration of metabolic health by decreased consumption of branched-chain amino acids". The Journal of Physiology 596 (4): 623–645. February 2018. doi:10.1113/JP275075. PMID 29266268.
- ↑ "A branched-chain amino acid metabolite drives vascular fatty acid transport and causes insulin resistance". Nature Medicine 22 (4): 421–6. April 2016. doi:10.1038/nm.4057. PMID 26950361.
- ↑ "Decreased Consumption of Branched-Chain Amino Acids Improves Metabolic Health". Cell Reports 16 (2): 520–530. July 2016. doi:10.1016/j.celrep.2016.05.092. PMID 27346343.
- ↑ "Depleting dietary valine permits nonmyeloablative mouse hematopoietic stem cell transplantation". Science 354 (6316): 1152–1155. December 2016. doi:10.1126/science.aag3145. PMID 27934766. Bibcode: 2016Sci...354.1152T.
External links
 |