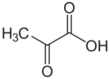
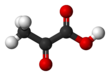
Chemistry:Pyruvic acid
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2-Oxopropanoic acid[1] | |||
| Systematic IUPAC name
2-Oxopropanoic acid | |||
| Other names
2-Oxopropionic acid
Pyruvic acid[1] α-Ketopropionic acid Acetylformic acid Pyroracemic acid Acetylcarboxylic acid | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| Abbreviations | Pyr | ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| KEGG | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C3H4O3 | |||
| Molar mass | 88.06 g/mol | ||
| Density | 1.250 g/cm3 | ||
| Melting point | 11.8 °C (53.2 °F; 284.9 K) | ||
| Boiling point | 165 °C (329 °F; 438 K) | ||
| Acidity (pKa) | 2.50[2] | ||
| Related compounds | |||
Other anions
|
Pyruvate 99px 100px | ||
Related keto-acids, carboxylic acids
|
|||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
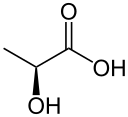
Pyruvic acid (CH3COCOOH) is the simplest of the alpha-keto acids, with a carboxylic acid and a ketone functional group. Pyruvate, the conjugate base, CH3COCOO−, is an intermediate in several metabolic pathways throughout the cell.
Pyruvic acid can be made from glucose through glycolysis, converted back to carbohydrates (such as glucose) via gluconeogenesis, or converted to fatty acids through a reaction with acetyl-CoA.[3] It can also be used to construct the amino acid alanine and can be converted into ethanol or lactic acid via fermentation.
Pyruvic acid supplies energy to cells through the citric acid cycle (also known as the Krebs cycle) when oxygen is present (aerobic respiration), and alternatively ferments to produce lactate when oxygen is lacking.[4]
History
In 1834, Théophile-Jules Pelouze distilled tartaric acid and isolated glutaric acid and another unknown organic acid. Jöns Jacob Berzelius characterized this other acid the following year and named pyruvic acid because it was distilled using heat.[5][6] The correct molecular structure was deduced by the 1870s.[7]
Production
Pyruvic acid is prepared by treating tartaric acid with acid.[8]
- CH
3COCN + 2 H
2O → CH
3COCO
2H + NH
3
Structure
Pyruvic acid crystallizes as the keto acid, not the enol. The six non-hydrogen atoms are nearly coplanar. More relevant to biochemistry is the structure of the pyruvate anion. Several salts of pyruvate have been examined by X-ray crystallography. These tests confirm that pyruvate anion also exists in the keto form.[9][10]
Reactivity
As a simple, abundant and bifunctional compound, pyruvic acid has been shown to participate in many reactions. Pyruvate reacts with amino acids to give alanine by the process called transamination:
- CH
3C(O)CO−
2 + RCH
2NH
2 → CH
3CH(NH
2)CO−
2 + RCHO
Pyruvic acid self-condenses to give zymonic acid, a cyclic dehydrated dimer:
- 2 CH
3C(O)CO
2H → (O=C)(HOC)(HC)C(CH
3)(CO
2H) + H
2O
The dehydration can be induced by distillation of pyruvic acid.[11] Zymonic acid in turn forms a variety of derivatives in aqueous solution.[12]
Pyruvic acid is a precursor to several types of heterocycles. When treated with phenethylamine, it gives tetrahydroisoquinoline by a sequential condensation/acylation process (Bischler–Napieralski reaction). With ortho-phenylenediamine it condenses to give quinoxalines. Condensation with 4,5-diaminopyrimidine give hydroxypteridines.[13]
Biochemistry
Pyruvate is important in biochemistry. It is the output of the metabolism of glucose known as glycolysis.[14] One molecule of glucose breaks down into two molecules of pyruvate,[14] which are then used to provide further energy, in one of two ways. Pyruvate is converted into acetyl-coenzyme A, which is the main input for a series of reactions known as the Krebs cycle (also known as the citric acid cycle or tricarboxylic acid cycle). Pyruvate is also converted to oxaloacetate by an anaplerotic reaction, which replenishes Krebs cycle intermediates; also, the oxaloacetate is used for gluconeogenesis.[15]
If insufficient oxygen is available, the acid is broken down anaerobically, creating lactate in animals and ethanol in plants and microorganisms (and in carp[16]). Pyruvate from glycolysis is converted by fermentation to lactate using the enzyme lactate dehydrogenase and the coenzyme NADH in lactate fermentation, or to acetaldehyde (with the enzyme pyruvate decarboxylase) and then to ethanol in alcoholic fermentation.
Pyruvate is a key intersection in the network of metabolic pathways. Pyruvate can be converted into carbohydrates via gluconeogenesis, to fatty acids or energy through acetyl-CoA, to the amino acid alanine, and to ethanol. Therefore, it unites several key metabolic processes.[17]

Pyruvic acid production by glycolysis
| phosphoenolpyruvate | pyruvate kinase | pyruvic acid | |

|
| ||
| ADP | ATP | ||
| |||
| ADP | ATP | ||
| pyruvate carboxylase and PEP carboxykinase | |||
Compound C00074 at KEGG Pathway Database. Enzyme 2.7.1.40 at KEGG Pathway Database. Compound C00022 at KEGG Pathway Database.
Click on genes, proteins and metabolites below to link to respective articles. [§ 1]
- ↑ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534". http://www.wikipathways.org/index.php/Pathway:WP534.
Decarboxylation to acetyl CoA
Pyruvate decarboxylation by the pyruvate dehydrogenase complex produces acetyl-CoA.
| pyruvate | pyruvate dehydrogenase complex | acetyl-CoA | |
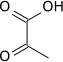
|
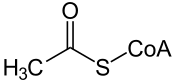
| ||
| CoA + NAD+ | CO2 + NADH + H+ | ||
| |||
Carboxylation to oxaloacetate
Carboxylation by pyruvate carboxylase produces oxaloacetate.
| pyruvate | pyruvate carboxylase | oxaloacetate | |

|
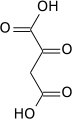
| ||
| ATP + CO2 | ADP + Pi | ||

| |||
Transamination to alanine
Transamination by alanine transaminase produces alanine.
| pyruvate | alanine transaminase | alanine | |

|

| ||
| glutamate | α-ketoglutarate | ||
| |||
| glutamate | α-ketoglutarate | ||
Reduction to lactate
Reduction by lactate dehydrogenase produces lactate.
| pyruvate | lactate dehydrogenase | lactate | |

|
| ||
| NADH | NAD+ | ||
| |||
| NADH | NAD+ | ||
Environmental chemistry
Pyruvic acid is an abundant carboxylic acid in secondary organic aerosols.[18]
Uses
Aside from its major role in the functioning of living organisms, pyruvic acid is of interest as a reagent in the synthesis of specialized organic compounds as discussed above in the reactivity section.[13]
See also
- Pyruvate scale
- Uvitonic acid
Notes
- ↑ 1.0 1.1 Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 748. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ Dawson, R. M. C. (1959). Data for Biochemical Research. Oxford: Clarendon Press.
- ↑ Fox, Stuart Ira (2011). Human Physiology (12th ed.). McGraw=Hill. p. 146.[ISBN missing]
- ↑ Ophardt, Charles E.. "Pyruvic Acid - Cross Roads Compound". Elmhurst College. http://chemistry.elmhurst.edu/vchembook/603pyruvic.html.
- ↑ Thomson, Thomas (1838). "Chapter II. Of fixed acids Section". Chemistry of organic bodies, vegetables. London: J. B. Baillière. p. 65. https://books.google.com/books?id=Wq45AAAAcAAJ&pg=PA65. Retrieved December 1, 2010.
- ↑ Berzelius, J. (1835). "Ueber eine neue, durch Destillation von Wein-und Traubensäure erhaltene Säure". Annalen der Pharmacie 13 (1): 61–63. doi:10.1002/jlac.18350130109. https://zenodo.org/record/1951856.
- ↑ "Pyruvic acid". Journal of the Chemical Society, Abstracts 34: 31. 1878. doi:10.1039/CA8783400019.Berzelius, J. J. (1835). "Ueber die Destillationsproducte der Traubensäure". Annalen der Physik 112 (9): 1–29. doi:10.1002/andp.18351120902.
- ↑ Howard, J. W.; Fraser, W. A.. "Pyruvic Acid". Organic Syntheses 4: 63. http://www.orgsyn.org/demo.aspx?prep=cv1p0475.; Collective Volume, 1, pp. 475
- ↑ Caro Garrido, Camila; Robeyns, Koen; Debecker, Damien P.; Luis, Patricia; Leyssens, Tom (2023). "From Liquid to Solid: Cocrystallization as an Engineering Tool for the Solidification of Pyruvic Acid". Crystals 13 (5): 808. doi:10.3390/cryst13050808.
- ↑ Rach, W.; Kiel, G.; Gattow, G. (1988). "Über Chalkogenolate. 187. Untersuchungen über Salze der Pyruvinsäure 2. Kristallstruktur von Kaliumpyruvat, Neubestimmung der Struktur von Natriumpyruvat". Zeitschrift für Anorganische und Allgemeine Chemie 563: 87–95. doi:10.1002/zaac.19885630113.
- ↑ Heger, Dominik; Eugene, Alexis J.; Parkin, Sean R.; Guzman, Marcelo I. (2019). "Crystal structure of zymonic acid and a redetermination of its precursor, pyruvic acid". Acta Crystallographica Section E 75 (6): 858–862. doi:10.1107/S2056989019007072. PMID 31391982.
- ↑ Perkins, Russell J.; Shoemaker, Richard K.; Carpenter, Barry K.; Vaida, Veronica (2016). "Chemical Equilibria and Kinetics in Aqueous Solutions of Zymonic Acid". The Journal of Physical Chemistry A 120 (51): 10096–10107. doi:10.1021/acs.jpca.6b10526. PMID 27991786.
- ↑ 13.0 13.1 Klingler, Franz Dietrich; Ebertz, Wolfgang (2000). "Oxocarboxylic Acids". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a18_313. ISBN 978-3-527-30385-4.
- ↑ 14.0 14.1 Lehninger, Albert L.; Nelson, David L.; Cox, Michael M. (2008). Principles of Biochemistry (5th ed.). New York, NY: W. H. Freeman and Company. p. 528. ISBN 978-0-7167-7108-1. https://archive.org/details/lehningerprincip00lehn_1/page/528.
- ↑ Nelson, David L.; Cox, Michael M. (2005), Principles of Biochemistry (4th ed.), New York: W. H. Freeman, p. 553, ISBN 0-7167-4339-6
- ↑ Aren van Waarde; G. Van den Thillart; Maria Verhagen (1993). "Ethanol Formation and pH-Regulation in Fish". Surviving Hypoxia. CRC Press. pp. 157–170. ISBN 0-8493-4226-0.
- ↑ Nelson, David L.; Cox, Michael M. (2005), Principles of Biochemistry (4th ed.), New York: W. H. Freeman, p. 553, ISBN 0-7167-4339-6
- ↑ Guzman, Marcelo I.; Eugene, Alexis J. (2021-09-01). "Aqueous Photochemistry of 2-Oxocarboxylic Acids: Evidence, Mechanisms, and Atmospheric Impact" (in en). Molecules 26 (17): 5278. doi:10.3390/molecules26175278. PMID 34500711.
References
- Cody, G. D.; Boctor, N. Z.; Filley, T. R.; Hazen, R. M.; Scott, J. H.; Sharma, A.; Yoder, H. S. Jr (2000). "Primordial Carbonylated Iron-Sulfur Compounds and the Synthesis of Pyruvate". Science 289 (5483): 1337–1340. doi:10.1126/science.289.5483.1337. PMID 10958777. Bibcode: 2000Sci...289.1337C.
External links
| Wikimedia Commons has media related to Pyruvic acid. |
 |