Biology:Vamorolone
 | |
| Clinical data | |
|---|---|
| Trade names | Agamree |
| Other names | VBP; VBP-15; 17α,21-Dihydroxy-16α-methylpregna-1,4,9(11)-triene-3,20-dione |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
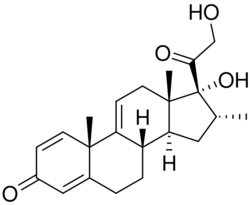
| Formula | C22H28O4 |
| Molar mass | 356.462 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Vamorolone, sold under the brand name Agamree, is a synthetic corticosteroid, which is used for the treatment of Duchenne muscular dystrophy.[4][5][6][7][8] It is taken by mouth.[1]
Vamorolone has anti-inflammatory and immunosuppressive effects as well as other glucocorticoid effects but is thought to lack certain other effects typical of glucocorticoids.[9][1]
Adverse events observed more frequently in the treated cohort in clinical studies included adrenal suppression, cushingoid features, psychiatric disorders, vomiting, weight gain, and vitamin D deficiency, among others.[1] It is a dual atypical glucocorticoid and antimineralocorticoid.[9]
Vamorolone was approved for medical use in the United States in October 2023,[10] and in the European Union in December 2023.[2][3]
Medical uses
Vamorolone is indicated for the treatment of Duchenne muscular dystrophy.[1]
Available forms
Vamorolone is provided in the form of an oral suspension at a concentration of 40 mg/mL.[1]
Side effects
Side effects of vamorolone in clinical trials that occurred at a rate of 10% or greater included development of cushingoid features, psychiatric disorders, vomiting, weight gain, vitamin D deficiency, and cough.[1] The psychiatric disorders that occurred more frequently than with placebo included abnormal behavior, aggression, agitation, anxiety, irritability, altered mood, sleep disorder, and stereotypy.[1] In addition to the preceding side effects, vamorolone shows dose-dependent suppression of the hypothalamic–pituitary–adrenal axis (HPA axis) and hence has potential risks of adrenal suppression and adrenal insufficiency with discontinuation.[1] Vamorolone also shows immunosuppression and is expected to increase the risk of infection, among various other potential adverse effects.[1]
Pharmacology
Pharmacodynamics
Vamorolone is a partial agonist of the glucocorticoid receptor with relative loss of transactivation activities, but retention of transrepression activities, compared to other glucocorticoids. As a result, it is described as possessing "dissociative" glucocorticoid properties.[9] In contrast to other corticosteroids, vamorolone is a potent antagonist of the mineralocorticoid receptor and hence has antimineralocorticoid activity.[9]
Chemistry
Vamorolone is a synthetic corticosteroid and is also known by the chemical name 17α,21-dihydroxy-16α-methylpregna-1,4,9(11)-triene-3,20-dione or as 16α-methyl-9,11-dehydroprednisolone. It is a derivative of cortisol (hydrocortisone) and prednisolone (1,2-dehydrocortisol).
Anti-inflammatory drugs of the corticosteroid class show a carbonyl (=O) or hydroxyl (-OH) group on the C11 carbon of the steroid backbone. In contrast, vamorolone contains a Δ9,11 double bond between the C9 and C11 carbons. This change in structure has been shown to remove a molecular contact site with the glucocorticoid receptor, and leads to dissociative properties.[11]
History
In phase I clinical trials of adult volunteers, vamorolone was shown to be safe and well tolerated, with blood biomarker data suggesting possible loss of safety concerns of the corticosteroid class.[12]
In phase IIa dose-ranging clinical trial of 48 children with Duchenne muscular dystrophy (2 weeks on drug, 2 weeks off drug), vamorolone was shown to be safe and well tolerated, and showed blood biomarker data consistent with a myofiber membrane stabilization and anti-inflammatory effects, and possible loss of safety concerns.[13] These children continued on to a 24-week open-label extension study at the same doses, and this showed dose-dependent improvement of motor outcomes, with 2.0 and 6.0 mg/kg/day suggesting benefit.[14] These same children continued on a long-term extension study with dose escalations, and this suggested continued clinical improvement through 18-months treatment.[15]
Population pharmacokinetics (PK) of vamorolone was shown to fit to a 1-compartment model with zero-order absorption, with both adult men and young boys showing dose-linearity of PK parameters for the doses examined, and no accumulation of the drug during daily dosing. Apparent clearance averaged 2.0 L/h/kg in men and 1.7 L/h/kg in boys. Overall, vamorolone exhibited well-behaved linear PK, with similar profiles in healthy men and boys with DMD, moderate variability in PK parameters, and absorption and disposition profiles similar to those of classical glucocorticoids.[16] Exposure/response analyses have suggested that the motor outcome of time to stand from supine velocity showed the highest sensitivity to vamorolone, with the lowest AUC value providing 50% of maximum effect (E50 = 186 ng·h/mL), followed by time to climb 4 stairs (E50 = 478 ng·h/mL), time to run/walk 10 m (E50 = 1220 ng·h/mL), and 6-minute walk test (E50 = 1770 ng·h/mL). Week 2 changes of proinflammatory PD biomarkers showed exposure-dependent decreases. The E50 was 260 ng·h/mL for insulin-like growth factor-binding protein 2, 1200 ng·h/mL for matrix metalloproteinase 12, 1260 ng·h/mL for lymphotoxin α1/β2, 1340 ng·h/mL for CD23, 1420 ng·h/mL for interleukin-22-binding protein, and 1600 ng·h/mL for macrophage-derived chemokine/C-C motif chemokine 22.[17]
Society and culture
Legal status
In October 2023, the US Food and Drug Administration (FDA) approved vamorolone (Agamree; Santhera Pharmaceuticals) for the treatment of Duchenne muscular dystrophy.[10][18][19]
In October 2023, the Committee for Medicinal Products for Human Use adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Agamree, intended for the treatment of Duchenne muscular dystrophy.[2] The applicant for this medicinal product is Santhera Pharmaceuticals (Deutschland) GmbH.[2]
Vamorolone was approved for medical use in the United States in October 2023,[10] and in the European Union in December 2023.[2][3]
Brand names
Vamorolone is the international nonproprietary name.[20]
Vamorolone is sold under the brand name Agamree.[1][2][3]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 "Agamree- vamorolone kit". 26 October 2023. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=fb376f09-cf9c-42b5-b4dd-02d5bcd73211.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Agamree EPAR". 12 October 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/agamree. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ↑ 3.0 3.1 3.2 3.3 "Agamree Product information". 15 December 2023. https://ec.europa.eu/health/documents/community-register/html/h1776.htm.
- ↑ "Vamorolone - ReveraGen Biopharma". AdisInsight. Springer Nature Switzerland AG. http://adisinsight.springer.com/drugs/800035994.
- ↑ "VBP15: preclinical characterization of a novel anti-inflammatory delta 9,11 steroid". Bioorganic & Medicinal Chemistry 21 (8): 2241–2249. April 2013. doi:10.1016/j.bmc.2013.02.009. PMID 23498916.
- ↑ "VBP15, a novel anti-inflammatory and membrane-stabilizer, improves muscular dystrophy without side effects". EMBO Molecular Medicine 5 (10): 1569–1585. October 2013. doi:10.1002/emmm.201302621. PMID 24014378.
- ↑ "Asynchronous remodeling is a driver of failed regeneration in Duchenne muscular dystrophy". The Journal of Cell Biology 207 (1): 139–158. October 2014. doi:10.1083/jcb.201402079. PMID 25313409.
- ↑ "VBP15, a novel dissociative steroid compound, reduces NFκB-induced expression of inflammatory cytokines in vitro and symptoms of murine trinitrobenzene sulfonic acid-induced colitis". Inflammation Research 65 (9): 737–743. September 2016. doi:10.1007/s00011-016-0956-8. PMID 27261270.
- ↑ 9.0 9.1 9.2 9.3 "Vamorolone targets dual nuclear receptors to treat inflammation and dystrophic cardiomyopathy". Life Sci Alliance 2 (1): e201800186. February 2019. doi:10.26508/lsa.201800186. PMID 30745312.
- ↑ 10.0 10.1 10.2 "Drug Approval Package: Agamree". 7 November 2023. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2023/215239Orig1s000TOC.cfm.
- ↑ "Disruption of a key ligand-H-bond network drives dissociative properties in vamorolone for Duchenne muscular dystrophy treatment". Proceedings of the National Academy of Sciences of the United States of America 117 (39): 24285–24293. September 2020. doi:10.1073/pnas.2006890117. PMID 32917814. Bibcode: 2020PNAS..11724285L.
- ↑ "Phase 1 trial of vamorolone, a first-in-class steroid, shows improvements in side effects via biomarkers bridged to clinical outcomes". Steroids 134: 43–52. June 2018. doi:10.1016/j.steroids.2018.02.010. PMID 29524454.
- ↑ "Phase IIa trial in Duchenne muscular dystrophy shows vamorolone is a first-in-class dissociative steroidal anti-inflammatory drug". Pharmacological Research 136: 140–150. October 2018. doi:10.1016/j.phrs.2018.09.007. PMID 30219580.
- ↑ "Vamorolone trial in Duchenne muscular dystrophy shows dose-related improvement of muscle function". Neurology 93 (13): e1312–e1323. September 2019. doi:10.1212/WNL.0000000000008168. PMID 31451516.
- ↑ "Efficacy and safety of vamorolone in Duchenne muscular dystrophy: An 18-month interim analysis of a non-randomized open-label extension study". PLOS Medicine 17 (9): e1003222. September 2020. doi:10.1371/journal.pmed.1003222. PMID 32956407.
- ↑ "Population Pharmacokinetics of Vamorolone (VBP15) in Healthy Men and Boys With Duchenne Muscular Dystrophy". Journal of Clinical Pharmacology 59 (7): 979–988. July 2019. doi:10.1002/jcph.1388. PMID 30742306.
- ↑ "Exposure-Response Analysis of Vamorolone (VBP15) in Boys With Duchenne Muscular Dystrophy". Journal of Clinical Pharmacology 60 (10): 1385–1396. October 2020. doi:10.1002/jcph.1632. PMID 32434278.
- ↑ "FDA Approves Vamorolone for Treatment of Duchenne Muscular Dystrophy in Patients Aged 2 Years and Older". 26 October 2023. https://www.pharmacytimes.com/view/fda-approves-vamorolone-for-treatment-of-duchenne-muscular-dystrophy-in-patients-aged-2-years-and-older.
- ↑ "Santhera Receives U.S. FDA Approval of Agamree (vamorolone) for the Treatment of Duchenne Muscular Dystrophy" (Press release). Santhera Pharmaceuticals Holding AG. 27 October 2023. Archived from the original on 31 October 2023. Retrieved 13 November 2023 – via GlobeNewswire.
- ↑ "International nonproprietary names for pharmaceutical substances (INN): recommended INN: list 77". WHO Drug Information 31 (1). 2017.
 |
