Chemistry:Hydrastine
Hydrastine is an isoquinoline alkaloid which was discovered in 1851 by Alfred P. Durand.[1] Nitric acid induced hydrolysis of hydrastine yields hydrastinine, which was patented by Bayer as a haemostatic drug in the early 1900s.[2] It is present in Hydrastis canadensis (thus the name) and other plants of the family Ranunculaceae.
Total synthesis
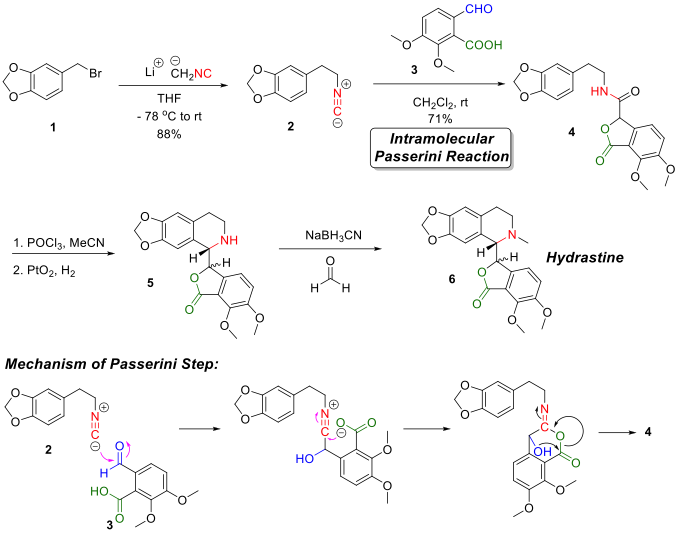
The first attempt for the total synthesis of hydrastine was reported by Sir Robert Robinson and co-workers[3] in 1931. Following studies[4][5] where the synthesis of the key lactonic amide intermediate (structure 4 in figure) was the most troublesome, the major breakthrough was achieved in 1981 when J. R. Falck and co-workers[6] reported a four-step total synthesis of hydrastine from simple starting materials. The key step in the Falck synthesis was using a Passerini reaction to construct the lactonic amide intermediate 4.
Starting from a simple phenylbromide variant 1, alkylation reaction with lithium methylisocyanide gives the isocyanide intermediate 2. Reacting isocyanide intermediate 2 with opianic acid 3 initiated the intramolecular Passerini reaction to give the key lactonic amide intermediate 4. The tetrahydro-isoquinolin ring was formed by first a ring-closure reaction under dehydration conditions using POCl3 and then a catalyzed hydrogenation using PtO2 as the catalyst. Finally, hydrastine was synthesized by installing the N-methyl group via reductive amination reaction with formaldehyde.
Biological action
Hydrastine acts as a convulsant in mice. It appears to do this by binding to bicuculline-sensitive GABAA receptors as a potent competitive antagonist. The action appears to be largely mediated by the (+) enantiomer (IC50 of 0.4µM), as (-)-hydrastine is 180 times less potent in regards to this effect.[7]
See also
- Bicuculline (very similar in structure)
References
- ↑ "On Hydrastine, an Alkaloid Occurring in Hydrastis Canadensis.". Pharmaceutical Journal: A Weekly Record of Pharmacy and Allied Sciences (J. Churchill): 547–. July 1862. https://books.google.com/books?id=IusKAAAAYAAJ&pg=PA547.
- ↑ Römpp CD, Georg Thieme Verlag, 2006
- ↑ "XXXI.—A synthesis of hydrastine. Part I". J. Chem. Soc.: 236–247. 1931. doi:10.1039/JR9310000236. ISSN 0368-1769.
- ↑ "Synthesis of Hydrastine". Nature 165 (4196): 529. 1950. doi:10.1038/165529a0. ISSN 0028-0836. Bibcode: 1950Natur.165..529H.
- ↑ "360. A new route to the phthalide-isoquinoline bases, and a synthesis of (–)-hydrastine". J. Chem. Soc.: 1776–1780. 1950. doi:10.1039/JR9500001776. ISSN 0368-1769.
- ↑ "An intramolecular passerini reaction: Synthesis of hydrastine.". Tetrahedron Letters 22 (7): 619–620. 1981. doi:10.1016/S0040-4039(01)92504-3. ISSN 0040-4039.
- ↑ "(+)-Hydrastine, a potent competitive antagonist at mammalian GABAA receptors". British Journal of Pharmacology 99 (4): 727–730. April 1990. doi:10.1111/j.1476-5381.1990.tb12997.x. PMID 2163278.
External links
{{Navbox | name = GABA receptor modulators | title = GABA receptor modulators | state = collapsed | bodyclass = hlist | groupstyle = text-align:center;
| group1 = Ionotropic | list1 = {{Navbox|subgroup | groupstyle = text-align:center | groupwidth = 5em
| group1 = GABAA | list1 =
- Agonists: (+)-Catechin
- Bamaluzole
- Barbiturates (e.g., phenobarbital)
- BL-1020
- DAVA
- Dihydromuscimol
- GABA
- Gabamide
- GABOB
- Gaboxadol (THIP)
- Homotaurine (tramiprosate, 3-APS)
- Ibotenic acid
- iso-THAZ
- iso-THIP
- Isoguvacine
- Isomuscimol
- Isonipecotic acid
- Kojic amine
- Lignans (e.g., honokiol)
- Methylglyoxal
- Monastrol
- Muscimol
- Nefiracetam
- Neuroactive steroids (e.g., allopregnanolone)
- Org 20599
- PF-6372865
- Phenibut
- Picamilon
- P4S
- Progabide
- Propofol
- Quisqualamine
- SL-75102
- TACA
- TAMP
- Terpenoids (e.g., borneol)
- Thiomuscimol
- Tolgabide
- ZAPA
- Positive modulators (abridged; see here for a full list): α-EMTBL
- Alcohols (e.g., ethanol)
- Anabolic steroids
- Avermectins (e.g., ivermectin)
- Barbiturates (e.g., phenobarbital)
- Benzodiazepines (e.g., diazepam)
- Bromide compounds (e.g., potassium bromide)
- Carbamates (e.g., meprobamate)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Dihydroergolines (e.g., ergoloid (dihydroergotoxine))
- Etazepine
- Etifoxine
- Fenamates (e.g., mefenamic acid)
- Flavonoids (e.g., apigenin, hispidulin)
- Fluoxetine
- Flupirtine
- Imidazoles (e.g., etomidate)
- Kava constituents (e.g., kavain)<!--PMID: 9776662-->
- Lanthanum
- Loreclezole
- Monastrol
- Neuroactive steroids (e.g., allopregnanolone, [[Chemistry:Cholecholesterol]], THDOC)
- Niacin
- Nicotinamide (niacinamide)
- Nonbenzodiazepines (e.g., β-carbolines (e.g., [[abecarnil), cyclopyrrolones (e.g., zopiclone), imidazopyridines (e.g., zolpidem), pyrazolopyrimidines (e.g., zaleplon))
- Norfluoxetine
- Petrichloral
- Phenols (e.g., propofol)
- Phenytoin
- Piperidinediones (e.g., glutethimide)
- Propanidid
- Pyrazolopyridines (e.g., etazolate)
- Quinazolinones (e.g., methaqualone)
- Retigabine (ezogabine)
- ROD-188
- Skullcap constituents (e.g., baicalin)
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal))
- Topiramate
- Valerian constituents (e.g., valerenic acid)
- Volatiles/gases (e.g., chloral hydrate, chloroform, [[Chemistry:Diethyl diethyl ether, Parparaldehyde]], sevoflurane)
- Antagonists: Bicuculline
- Coriamyrtin
- Dihydrosecurinine
- Gabazine (SR-95531)
- Hydrastine
- Hyenachin (mellitoxin)
- PHP-501
- Pitrazepin
- Securinine
- Sinomenine
- SR-42641
- SR-95103
- Thiocolchicoside
- Tutin
- Negative modulators: 1,3M1B
- 3M2B
- 11-Ketoprogesterone
- 17-Phenylandrostenol
- α5IA (LS-193,268)
- β-CCB
- β-CCE
- β-CCM
- β-CCP
- β-EMGBL
- Anabolic steroids
- Amiloride
- Anisatin
- β-Lactams (e.g., penicillins, cephalosporins, carbapenems)
- Basmisanil
- Bemegride
- Bicyclic phosphates (TBPS, TBPO, IPTBO)
- BIDN
- Bilobalide
- Bupropion
- CHEB
- Chlorophenylsilatrane
- Cicutoxin
- Cloflubicyne
- Cyclothiazide
- DHEA
- DHEA-S
- Dieldrin
- (+)-DMBB
- DMCM
- DMPC
- EBOB
- Etbicyphat
- FG-7142 (ZK-31906)
- Fiproles (e.g., fipronil)
- Flavonoids (e.g., amentoflavone, oroxylin A)
- Flumazenil
- Fluoroquinolones (e.g., ciprofloxacin)
- Flurothyl
- Furosemide
- Golexanolone
- Iomazenil (123I)
- IPTBO
- Isopregnanolone (sepranolone)
- L-655,708
- Laudanosine
- Leptazol
- Lindane
- MaxiPost
- Morphine
- Morphine-3-glucuronide
- MRK-016
- Naloxone
- Naltrexone
- Nicardipine
- Nonsteroidal antiandrogens (e.g., [[apalutamide, [[Chemistry:Bicalutbicalutamide, Enzalutenzalutamide, Chemistry:Flutamide|flut]]amide]], nilutamide)
- Oenanthotoxin
- Pentylenetetrazol (pentetrazol)
- Phenylsilatrane
- Picrotoxin (i.e., picrotin, picrotoxinin and dihydropicrotoxinin)
- Pregnenolone sulfate
- Propybicyphat
- PWZ-029
- Radequinil
- Ro 15-4513
- Ro 19-4603
- RO4882224
- RO4938581
- Sarmazenil
- SCS
- Suritozole
- TB-21007
- TBOB
- TBPS
- TCS-1105
- Terbequinil
- TETS
- Thujone
- U-93631
- Zinc
- ZK-93426
| group2 = GABAA-ρ | list2 =
- Agonists: BL-1020
- CACA
- CAMP
- Homohypotaurine
- GABA
- GABOB
- Ibotenic acid
- Isoguvacine
- Muscimol
- N4-Chloroacetylcytosine arabinoside
- Picamilon
- Progabide
- TACA
- TAMP
- Thiomuscimol
- Tolgabide
- Positive modulators: Allopregnanolone
- Alphaxolone
- ATHDOC
- Lanthanides
- Antagonists: (S)-2-MeGABA
- (S)-4-ACPBPA
- (S)-4-ACPCA
- 2-MeTACA
- 3-APMPA
- 4-ACPAM
- 4-GBA
- cis-3-ACPBPA
- CGP-36742 (SGS-742)
- DAVA
- Gabazine (SR-95531)
- Gaboxadol (THIP)
- I4AA
- Isonipecotic acid
- Loreclezole
- P4MPA
- P4S
- SKF-97541
- SR-95318
- SR-95813
- TPMPA
- trans-3-ACPBPA
- ZAPA
- Negative modulators: 5α-Dihydroprogesterone
- Bilobalide
- Loreclezole
- Picrotoxin (picrotin, picrotoxinin)
- Pregnanolone
- ROD-188
- THDOC
- Zinc
}}
| group2 = Metabotropic
| list2 =
| below =
- See also
- Receptor/signaling modulators
- GABAA receptor positive modulators
- GABA metabolism/transport modulators
}}
 |