Chemistry:Mephentermine
Mephentermine, sold under the brand name Wyamine among others, is a sympathomimetic medication which was previously used in the treatment of low blood pressure but is mostly no longer marketed.[1][2][3][4][5] It is used by injection into a vein or muscle, by inhalation, and by mouth.[3][2]
Side effects of mephentermine include dry mouth, sedation, reflex bradycardia, arrhythmias, and hypertension.[3] Mephentermine induces the release of norepinephrine and dopamine and is described as an indirectly acting sympathomimetic and psychostimulant.[3] Its sympathomimetic effects are mediated by indirect activation of α- and β-adrenergic receptors.[2] Chemically, it is a substituted phenethylamine and amphetamine and is closely related to phentermine and methamphetamine.[3][6][7]
Mephentermine was first described and introduced for medical use by 1952.[8] It was discontinued in the United States between 2000 and 2004.[9][4] The medication appears to remain available only in India.[3][4][5] Misuse of mephentermine for recreational and performance-enhancing purposes has been increasingly encountered in modern times, especially in India.[10][3]
Medical uses
For maintenance of blood pressure in hypotensive states, the dose for adults is 30 to 45 mg as a single dose, repeated as necessary or followed by intravenous infusion of 0.1% mephentermine in 5% dextrose, with the rate and duration of administration depending on the patient's response. For hypotension secondary to spinal anesthesia in obstetric patients, the dose for adults is 15 mg as a single dose, repeated if needed. The maximum dose 30 mg. Mephentermine has also been used as a decongestant.[1][2]
Available forms
Mephentermine is available in the form of 15 and 30 mg/mL solutions for intravenous infusion or intramuscular injection and in the form of 10 mg oral tablets.[3] It has also been available in the form of inhalers.[2]
Contraindications
Side effects
Interactions
Pharmacology
Pharmacodynamics
Mephentermine is thought to act as a norepinephrine–dopamine releasing agent.[3] It is described as an indirectly acting sympathomimetic, cardiac stimulant, adrenergic, vasoconstrictor, antihypotensive agent, and psychostimulant.[7][9][5][3] Its sympathomimetic effects are mediated by indirect activation of α- and β-adrenergic receptors.[2][1]
Pharmacokinetics
Its onset of action is 5 to 15 minutes with intramuscular injection and is immediate with intravenous administration. Its duration of action is 4 hours with intramuscular injection and 30 minutes with intravenous administration.{{Citation needed|date=July 2024} Mephentermine, along with phentermine, is known to be produced as a metabolite of the orally administered local anesthetic oxetacaine (oxethazaine).[11][12]
Chemistry
Mephentermine, also known as N,α,α-trimethylphenethylamine or N,α-dimethylampetamine, is a phenethylamine and amphetamine derivative.[6][7][3] It is the N-methylated analogue of phentermine (α-methylamphetamine) and is also known as N-methylphentermine.[6][7] In addition, mephentermine is the α-methylated analogue of methamphetamine or the α,α-dimethylated derivative of amphetamine.[6][3] The cathinone (β-keto) derivative of mephentermine is α-methylmethcathinone (βk-mephentermine; RAD-081).[13][14][15]
Synthesis
Mephentermine can by synthesized beginning with a Henry reaction between benzaldehyde (1) and 2-nitropropane (2) to give 2-methyl-2-nitro-1-phenylpropan-1-ol (3).[16] The nitro group is reduced with zinc in sulfuric acid giving 2-phenyl-1,1-dimethylethanolamine (4). Imine formation by dehydration with benzaldehyde gives (5). Alkylation with iodomethane leads to (6). Halogenation with thionyl chloride gives (7). Lastly, a Rosenmund reduction completes the synthesis of mephentermine (8).
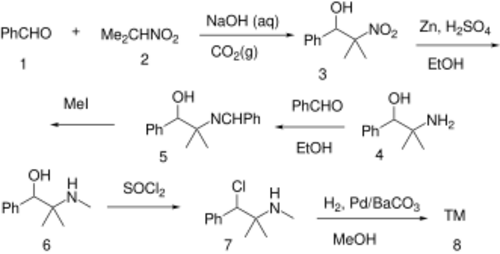
Mephentermine can also be synthesized by condensation of phentermine with benzaldehyde to get a Schiff base which can be alkylated with methyl iodide to give mephentermine.[17]
History
Mephentermine was first described in the literature and was introduced for medical use under the brand name Wyamine by 1952.[8] It was discontinued in the United States between 2000 and 2004.[9][4]
Society and culture
Names
Mephentermine is the generic name of the drug and its INN, BAN, and DCF.[7][9][18] In the case of the sulfate salt, its USAN is mephentermine sulfate and its BANM is mephentermine sulphate.[18][9][4] Synonyms of mephentermine include mephetedrine and mephenterdrine.[9][4][6] Brand names of mephentermine include Wyamine (US), Fentermin (PT), and Mephentine (IN).[7][9][4]
Availability
Mephentermine is no longer available in the United States and remains available in few or no other countries.[4][5] However, it appears to remain available in India.[5][4] It has also remained available in Brazil for use in veterinary medicine.[2]
Recreational use
Misuse of mephentermine for recreational and/or performance-enhancing purposes has been reported along with addiction and dependence and serious health complications.[19][20][21][22][2][23][24][25][10][26][27][28][29][3] It has been especially encountered in India, the only country in which mephentermine appears to remain available for medical use.[3][4][5]
Exercise and sports
Mephentermine has been used as a performance-enhancing drug in exercise and sports.[1][10][3] It is on the World Anti-Doping Agency (WADA) list of prohibited substances.[30][29]
Research
Mephentermine was evaluated in the treatment of congestive heart failure in one small clinical study but was found to be ineffective.[31][32]
Veterinary use
Mephentermine has been used in veterinary medicine in Brazil under the brand names Potenay and Potemax.[2]
References
- ↑ 1.0 1.1 1.2 1.3 "Pharmacology of stimulants prohibited by the World Anti-Doping Agency (WADA)". Br J Pharmacol 154 (3): 606–622. June 2008. doi:10.1038/bjp.2008.124. PMID 18500382.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 2.8 "Mephentermine: rediscovering its biology and use, misuse and their implications". Braz J Psychiatry 33 (1): 98–99. March 2011. doi:10.1590/s1516-44462011000100019. PMID 21537728.
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 3.11 3.12 3.13 3.14 ""Mephentermine Abuse" an Age-old Concern with New Challenges: Review of Literature with Case Series". Annals of Indian Psychiatry 7 (3): 262–266. 2023. doi:10.4103/aip.aip_59_23. ISSN 2588-8366.
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 Schweizerischer Apotheker-Verein (2004). Index Nominum: International Drug Directory. Medpharm Scientific Publishers. p. 757. ISBN 978-3-88763-101-7. https://books.google.com/books?id=EgeuA47Ocm4C&pg=PA757. Retrieved 28 July 2024.
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 "Mephentermine". 5 August 2020. https://drugs.com/international/mephentermine.html.
- ↑ 6.0 6.1 6.2 6.3 6.4 "Mephentermine". https://pubchem.ncbi.nlm.nih.gov/compound/3677.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer US. 2014. p. 968. ISBN 978-1-4757-2085-3. https://books.google.com/books?id=0vXTBwAAQBAJ&pg=PA968. Retrieved 28 July 2024.
- ↑ 8.0 8.1 "Mephentermine: an effective pressor amine; clinical and laboratory observations". Am Heart J 44 (3): 396–406. September 1952. doi:10.1016/0002-8703(52)90261-5. PMID 14952463.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 Schweizerischer Apotheker-Verein (2000). Index Nominum 2000: International Drug Directory. Medpharm Scientific Publishers. p. 647. ISBN 978-3-88763-075-1. https://books.google.com/books?id=5GpcTQD_L2oC&pg=PA647. Retrieved 28 July 2024.
- ↑ 10.0 10.1 10.2 "Mephentermine Dependence in a Young Athlete: Case Report With Review of Literature". J Addict Med 11 (4): 328–330. 2017. doi:10.1097/ADM.0000000000000313. PMID 28574863.
- ↑ "Oxethazaine as the source of mephentermine and phentermine in athlete's urine". Forensic Sci Int 185 (1–3): e1–5. March 2009. doi:10.1016/j.forsciint.2008.12.009. PMID 19157735.
- ↑ "Confirming urinary excretion of mephentermine and phentermine following the ingestion of oxethazaine by gas chromatography-mass spectrometry analysis". J Anal Toxicol 34 (2): 73–77. March 2010. doi:10.1093/jat/34.2.73. PMID 20223098.
- ↑ "Investigation of the Optical Isomers of Methcathinone, and Two Achiral Analogs, at Monoamine Transporters and in Intracranial Self-Stimulation Studies in Rats". ACS Chem Neurosci 11 (12): 1762–1769. June 2020. doi:10.1021/acschemneuro.9b00617. PMID 32356961.
- ↑ Nguyen, Vy (2019). "Analyzing Interactions Between Methcathinone Analogs and the Human Monoamine Transporters". VCU Theses and Dissertations. doi:10.25772/T1DW-MG60.
- ↑ Davies, Rachel A (10 July 2019). Structure-Activity Relationship Studies of Synthetic Cathinones and Related Agents. VCU Scholars Compass (Thesis). doi:10.25772/TZSA-0396. Retrieved 24 November 2024.
- ↑ William F Bruce, Szabo Joseph Lester, Tubis Samuel, U.S. Patent 2,597,445 (1952 to Wyeth Corp)
- ↑ "Preparation of α,α-Dimethyl- and N,α,α-Trimethyl-β-cyclohexylethylamine". Journal of the American Chemical Society 70 (3): 955–957. 1948. doi:10.1021/ja01183a019. Bibcode: 1948JAChS..70..955Z.
- ↑ 18.0 18.1 Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Netherlands. 2012. p. 175. ISBN 978-94-011-4439-1. https://books.google.com/books?id=tsjrCAAAQBAJ&pg=PA175. Retrieved 28 July 2024.
- ↑ "Mephentermine psychosis: misuse of the Wyamine inhaler". Am J Psychiatry 126 (9): 1315–1317. March 1970. doi:10.1176/ajp.126.9.1315. PMID 5413209.
- ↑ "Mephentermine dependence with psychosis. A case report". Br J Psychiatry 152: 129–131. January 1988. doi:10.1192/bjp.152.1.129. PMID 3167321.
- ↑ "Case report of substance dependence with buprenorphine and mephentermine". Indian J Psychiatry 41 (2): 160–162. April 1999. PMID 21455380.
- ↑ "Mephentermine dependence without psychosis: a Brazilian case report". Addiction 105 (6): 1129–1130. June 2010. doi:10.1111/j.1360-0443.2010.02935.x. PMID 20456293.
- ↑ "Mephentermine dependence: an emerging challenge". CNS Neurosci Ther 18 (6): 509–510. June 2012. doi:10.1111/j.1755-5949.2012.00328.x. PMID 22672305.
- ↑ "Mephentermine dependence with psychosis". Gen Hosp Psychiatry 35 (6): 681.e9–10. 2013. doi:10.1016/j.genhosppsych.2013.04.019. PMID 23759255.
- ↑ "Mephentermine dependence syndrome - A new emerging trend of substance use". Asian J Psychiatr 17: 101–102. October 2015. doi:10.1016/j.ajp.2015.07.006. PMID 26236018.
- ↑ "Mephentermine dependence in a young Indian adult without psychosis". BMJ Case Rep 13 (11). November 2020. doi:10.1136/bcr-2020-236924. PMID 33139366.
- ↑ "Mephentermine abuse - A case report". Indian J Psychiatry 63 (4): 400–401. 2021. doi:10.4103/psychiatry.IndianJPsychiatry_934_20. PMID 34456355.
- ↑ "Herculean mistake: mephentermine associated cardiomyopathy". Phys Sportsmed 49 (1): 116–122. February 2021. doi:10.1080/00913847.2020.1763146. PMID 32404042.
- ↑ 29.0 29.1 "Mephentermine Misuse: An Impending Crisis among Sportspersons". J Psychoactive Drugs 54 (2): 196–198. 2022. doi:10.1080/02791072.2021.1936701. PMID 34126873.
- ↑ "The Prohibited List". 1 January 2024. https://www.wada-ama.org/en/prohibited-list.
- ↑ "Use of sympathomimetic amines in heart failure". Am J Cardiol 22 (2): 177–182. August 1968. doi:10.1016/0002-9149(68)90223-3. PMID 4874959.
- ↑ "The ineffectiveness of an inotropic agent, mephentermine (Wyamine), in the treatment of congestive heart failure". Am Heart J 62 (3): 301–303. September 1961. doi:10.1016/0002-8703(61)90395-7. PMID 13702337.
 |
