Chemistry:Molybdenum(VI) chloride
From HandWiki
| |||
| Names | |||
|---|---|---|---|
| Other names
molybdenum hexachloride
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
| UNII | |||
| |||
| |||
| Properties | |||
| MoCl6 | |||
| Appearance | black solid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tracking categories (test):
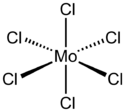
Molybdenum(VI) chloride is the inorganic compound with the formula MoCl6. It is a black diamagnetic solid. The molecules adopt an octahedral structure as seen in β-tungsten(VI) chloride.[1]
Preparation and reactions
Molybdenum(VI) chloride is prepared from the molybdenum hexafluoride with excess boron trichloride:
- MoF6 + 3 BCl3 → MoCl6 + 3 BF2Cl
It is unstable at room temperature with respect to molybdenum(V) chloride and decomposition completes within several days:
- 2 MoCl6 → [MoCl5]2 + Cl2
The treatment of MoF
6 with bismuth trichloride also produces MoCl6.[1]
References
 |