Chemistry:Zirconium(III) chloride
| |
| Names | |
|---|---|
| IUPAC name
Zirconium trichloride
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| Cl3Zr | |
| Molar mass | 197.57 g·mol−1 |
| Appearance | Blue-black crystals |
| Density | 3.05 g/cm3[1] |
| Melting point | 627 °C (1,161 °F; 900 K) at 760 mmHg[1] |
| Reacts[1] | |
| Solubility | Soluble in benzene, CS2 |
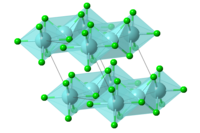
| Structure | |
| Hexagonal, hP6[2] | |
| P63/mcm, No. 193[2] | |
| 6/m 2/m 2/m[2] | |
a = 6.36 Å, c = 6.14 Å[2] α = 90°, β = 90°, γ = 120°
| |
| Thermochemistry | |
Heat capacity (C)
|
96.21 J/mol·K[3] |
Std molar
entropy (S |
145.79 J/mol·K[3] |
Std enthalpy of
formation (ΔfH⦵298) |
−714.21 kJ/mol[3] |
| Related compounds | |
Other anions
|
Zirconium(III) bromide Zirconium(III) iodide |
Other cations
|
Titanium(III) chloride Hafnium(III) chloride |
Related compounds
|
Zirconium(IV) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Zirconium(III) chloride is an inorganic compound with formula ZrCl3. It is a blue-black solid that is highly sensitive to air.
Preparation
The material was first claimed by Ruff and Wallstein who reduced zirconium tetrachloride with aluminium to give impure samples.[4] Subsequently, the problem with aluminium contamination was solved when it was prepared by reduction using zirconium metal:[5]
- Zr + 3 ZrCl4 → 4 ZrCl3
When aluminium is used as the reducing agent with zirconium tetrachloride, a series of choloroaluminates are formed, for example [Zr(AlCl4)2(AlCl4)2] and Zr(AlCl4)3.[6]
Since the trihalides, such as zirconium trichloride, are comparatively nonvolatile, contamination can be avoided by using a gaseous reductant. For example, zirconium trichloride can be prepared by reduction of zirconium tetrachloride with hydrogen.[7]
- ZrCl4 + ½ H2 → ZrCl3 + HCl
Structure
Some zirconium halides (ZrCl3, ZrBr3, and ZrI3) have structures similar to HfI3. They also have similar space group (P63/mcm) and hexagonal structure with 2 molecules in the cell.[2] The magnetic susceptibility of zirconium trichloride suggests metal-metal interactions of the unpaired electron on each Zr(III) center. The magnetic moment of ZrCl3 (0.4 BM) indicates considerable overlap of metal orbitals.[8]
References
- ↑ 1.0 1.1 1.2 Lide, David R., ed (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Press. ISBN 978-1-4200-9084-0.
- ↑ 2.0 2.1 2.2 2.3 2.4 Douglas, Bodie E.; Ho, Shih-Ming (2007). Structure and Chemistry of Crystalline Solids. New York: Springer Science+Business Media, Inc.. p. 101. ISBN 978-0-387-26147-8. https://books.google.com/books?id=hYRCAAAAQBAJ&pg=PA101.
- ↑ 3.0 3.1 3.2 Zirconium chloride in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD), http://webbook.nist.gov (retrieved 2014-06-23)
- ↑ Ruff, Otto; Wallstein, Richard (1923). "Reduktion anorganischer Halogenide III.1) Die Reduktion des Zirkontetrachlorids". Zeitschrift für anorganische und allgemeine Chemie 128: 96–116. doi:10.1002/zaac.19231280110.
- ↑ Hoffman, David M.; Lee, Samkeun (1992). "Synthesis of pyridine complexes of zirconium(III) chloride and the apparent oxidation to zirconium(IV) by a nitrile". Inorganic Chemistry 31 (13): 2675. doi:10.1021/ic00039a002.
- ↑ Larsen, E. M.; Moyer, James W.; Gil-Arnao, Francisco.; Camp, Michael J. (1974). "Synthesis of crystalline zirconium trihalides by reduction of tetrahalides in molten aluminum halides. Nonreduction of hafnium". Inorganic Chemistry 13 (3): 574. doi:10.1021/ic50133a015.
- ↑ Newnham, I. E.; Watts, J. A. (1960). "The Preparation of the Anhydrous Zirconium Trihalides". Journal of the American Chemical Society 82 (9): 2113. doi:10.1021/ja01494a006.
- ↑ Wells, A.F. Structural Inorganic Chemistry. Oxford Science Publications, 1975, 5th ed, 417-420.
 |

