Chemistry:Bevantolol
From HandWiki
Short description: Chemical compound
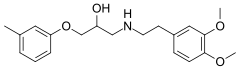 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C20H27NO4 |
| Molar mass | 345.439 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| (verify) | |
Bevantolol (INN) was a drug candidate for angina and hypertension that acted as both a beta blocker and a calcium channel blocker.[1][2] It was discovered and developed by Warner-Lambert[3] but in January 1989 the company announced that it had withdrawn the New Drug Application; the company's chairman said: "Who needs the 30th beta blocker?"[4] As of 2016[update] it wasn't marketed in the US, UK, or Europe and the authors of a Cochrane review could find no product monograph for it.[5]
References
- ↑ "Bevantolol. A preliminary review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy in hypertension and angina pectoris". Drugs 35 (1): 1–21. January 1988. doi:10.2165/00003495-198835010-00001. PMID 2894292.
- ↑ "Bevantolol: a beta-1 adrenoceptor antagonist with unique additional actions". Journal of Clinical Pharmacology 27 (7): 450–60. July 1987. doi:10.1002/j.1552-4604.1987.tb03049.x. PMID 2888789.
- ↑ McPherson, Edwin M. (2007). Pharmaceutical Manufacturing Encyclopedia. (3rd ed.). Burlington: Elsevier. pp. 618–619. ISBN 9780815518563. https://books.google.com/books?id=_J2ti4EkYpkC&pg=PA618.
- ↑ "Warner-Lambert Pipeline Narrowed to 40 Active Research Compounds". Pink Sheet. 30 January 1989. https://pink.pharmamedtechbi.com/PS015015/WARNERLAMBERT-PIPELINE-NARROWED-TO-40-ACTIVE-RESEARCH-COMPOUNDS-ACCUPRIL-QUINAPRIL-NDA-SUBMITTED-JAN-25-80-MIL-BUDGETTED-FOR-CV-WORK-IN-1989.
- ↑ "Blood pressure lowering efficacy of beta-1 selective beta blockers for primary hypertension". The Cochrane Database of Systematic Reviews 3 (4): CD007451. March 2016. doi:10.1002/14651858.CD007451.pub2. PMID 26961574.
 |
