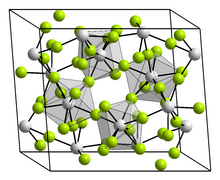
Chemistry:Zirconium tetrafluoride
| |
| Names | |
|---|---|
| IUPAC names
Zirconium(IV) fluoride
Zirconium tetrafluoride | |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| ZrF4 | |
| Molar mass | 167.21 g/mol |
| Appearance | white crystalline powder |
| Density | 4.43 g/cm3 (20 °C) |
| Melting point | 910 °C (1,670 °F; 1,180 K) |
| 1.32 g/100mL (20 °C) 1.388 g/100mL (25 °C) | |
| Structure | |
| Monoclinic, mS60 | |
| C12/c1, No. 15 | |
| Hazards | |
| GHS pictograms | 
|
| GHS Signal word | Danger |
| HH314Script error: No such module "Preview warning".Category:GHS errors | |
| PP260Script error: No such module "Preview warning".Category:GHS errors, PP264Script error: No such module "Preview warning".Category:GHS errors, PP280Script error: No such module "Preview warning".Category:GHS errors, PP301+P330+P331Script error: No such module "Preview warning".Category:GHS errors, PP302+P361+P354Script error: No such module "Preview warning".Category:GHS errors, PP304+P340Script error: No such module "Preview warning".Category:GHS errors, PP305+P354+P338Script error: No such module "Preview warning".Category:GHS errors, PP316Script error: No such module "Preview warning".Category:GHS errors, PP321Script error: No such module "Preview warning".Category:GHS errors, PP363Script error: No such module "Preview warning".Category:GHS errors, PP405Script error: No such module "Preview warning".Category:GHS errors, PP501Script error: No such module "Preview warning".Category:GHS errors | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
98 mg/kg (oral, mouse) 98 mg/kg (oral, rat)[1] |
| Related compounds | |
Other anions
|
Zirconium(IV) chloride Zirconium(IV) bromide Zirconium(IV) iodide |
Other cations
|
Titanium(IV) fluoride Hafnium(IV) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Zirconium(IV) fluoride describes members of a family inorganic compounds with the formula ZrF4(H2O)x. All are colorless, diamagnetic solids. Anhydrous Zirconium(IV) fluoride is a component of ZBLAN fluoride glass.[2]
Structure
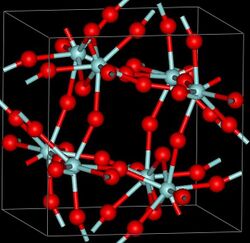
Three crystalline phases of ZrF4 have been reported, the stable β phase (monoclinic, space group I2/c) , α (tetragonal, space group P42/m) which is a high-temperature meta-stable form that can be stabilized by quenching. The γ-form (monoclinic, space group P21/c) is a high-pressure phase, forming at pressures between (4-8 GPa)[3]. α and γ phases are unstable and transform into the β phase at 400[4] and 723°C[3] respectively. There also exists an amorphous phase[3].
Zirconium(IV) fluoride forms several hydrates. The trihydrate has the structure (μ–F)
2[ZrF
3(H
2O)
3]
2.[5]
Preparation and reactions
Zirconium fluoride can be produced by several methods. Zirconium dioxide reacts with hydrogen fluoride and hydrofluoric acid to afford the anhydrous and monohydrates:
- ZrO
2 + 4 HF → ZrF
4 + 2 H
2O
The reaction of Zr metal reacts at high temperatures with HF as well:
- Zr + 4 HF → ZrF
4 + 2 H
2
Zirconium dioxide reacts at 200 °C with solid ammonium bifluoride to give the heptafluorozirconate salt, which can be converted to the tetrafluoride at 500 °C:
- 2ZrO
2 + 7 (NH
4)HF
2 → 2 (NH
4)
3ZrF
7 + 4 H
2O + NH
3 - (NH
4)
3ZrF
7 → ZrF
4 + 3 HF + 3 NH
3
Addition of hydrofluoric acid to solutions of zirconium nitrate precipitates solid monohydrate. Hydrates of zirconium tetrafluoride can be dehydrated by heating under a stream of hydrogen fluoride.
Zirconium fluoride can be purified by distillation or sublimation.[2]
Zirconium fluoride forms double salts with other fluorides. The most prominent is potassium hexafluorozirconate, formed by fusion of potassium fluoride and zirconium tetrafluoride:[6]
- ZrF
4 + 2 KF → K
2ZrF
6
Applications
The major and perhaps only commercial application of zirconium fluoride is as a precursor to ZBLAN glasses.[2]
Mixture of sodium fluoride, zirconium fluoride, and uranium tetrafluoride (53-41-6 mol.%) was used as a coolant in the Aircraft Reactor Experiment. A mixture of lithium fluoride, beryllium fluoride, zirconium fluoride, and uranium-233 tetrafluoride was used in the Molten-Salt Reactor Experiment. (Uranium-233 is used in the thorium fuel cycle reactors.)[citation needed]
References
- ↑ "Zirconium compounds (as Zr)". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH). https://www.cdc.gov/niosh/idlh/7440677.html.
- ↑ 2.0 2.1 2.2 Nielsen, Ralph (2000). "Zirconium and Zirconium Compounds". Ullmann's Encyclopedia of Industrial Chemistry. doi:10.1002/14356007.a28_543. ISBN 3527306730.
- ↑ 3.0 3.1 3.2 Laval, J.-P. (2014-08-15). "Crystal chemistry of anion-excess ReO3-related phases. III." (in en). Acta Crystallographica Section C: Structural Chemistry 70 (8): 742–748. doi:10.1107/S2053229614014338. ISSN 2053-2296. https://journals.iucr.org/c/issues/2014/08/00/ov3051/.
- ↑ Paul L. Brown; Federico J. Mompean; Jane Perrone; Myriam Illemassène (2005). Chemical thermodynamics of zirconium. Gulf Professional Publishing. p. 144. ISBN 0-444-51803-7. https://books.google.com/books?id=DvqwTdVhjMEC&pg=PA144.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. p. 965. ISBN 978-0-08-037941-8.
- ↑ Meshri., Dayal T. (2000), "Fluorine compounds, inorganic, titanium", Kirk-Othmer Encyclopedia of Chemical Technology, New York: John Wiley, doi:10.1002/0471238961.2009200113051908.a01, ISBN 9780471238966, http://onlinelibrary.wiley.com/book/10.1002/0471238961
- ORNL/TM-2006/12 Assessment of Candidate Molten Salt Coolants for the Advanced High-Temperature Reactor (AHTR), March 2006 (Accessed 2008/9/18)
 |
