Chemistry:Ammonium bifluoride
|
| |||

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Ammonium bifluoride
| |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| UN number | 1727 | ||
| |||
| |||
| Properties | |||
| [NH 4][HF 2] | |||
| Molar mass | 57.044 g·mol−1 | ||
| Appearance | Colourless crystals | ||
| Density | 1.50 g cm−3 | ||
| Melting point | 126 °C (259 °F; 399 K) | ||
| Boiling point | 240 °C (464 °F; 513 K)(decomposes) | ||
| 63g/(100 ml) (20 °C) | |||
| Solubility in alcohol | slightly soluble | ||
Refractive index (nD)
|
1.390 | ||
| Structure | |||
| Cubic, related to the CsCl structure | |||
| [NH 4]+ cation: tetrahedral [HF 2]− anion: linear | |||
| Hazards | |||
| GHS pictograms |   [1] [1]
| ||
| H301, H314[1] | |||
| P280, P301+310, P305+351+338, P310[1] | |||
| NFPA 704 (fire diamond) | |||
| Related compounds | |||
Other cations
|
potassium bifluoride | ||
Related compounds
|
ammonium fluoride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Ammonium bifluoride is the inorganic compound with the formula [NH
4][HF
2] or [NH
4]F · HF. It is produced from ammonia and hydrogen fluoride. This colourless salt is a glass-etchant and an intermediate in a once-contemplated route to hydrofluoric acid.
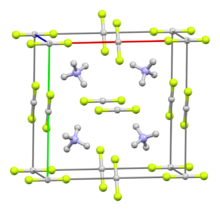
Structure
Ammonium bifluoride, as its name indicates, contains an ammonium cation ([NH
4]+
), and a bifluoride or hydrogen(difluoride) anion ([HF
2]−
). The centrosymmetric triatomic bifluoride anion features the strongest known hydrogen bond, with a F−H length of 114 pm.[2] and a bond energy greater than 155 kJ/mol.[3]
In solid [NH
4][HF
2], each ammonium cation is surrounded by four fluoride centers in a tetrahedron, with hydrogen-fluorine hydrogen bonds present between the hydrogen atoms of the ammonium ion and the fluorine atoms.[4][5] Solutions contain tetrahedral [NH
4]+
cations and linear [HF
2]−
anions.
Production and applications
Ammonium bifluoride is a component of some etchants. It attacks silica component of glass:
Potassium bifluoride is a related more commonly used etchant.
Ammonium bifluoride has been considered as an intermediate in the production of hydrofluoric acid from hexafluorosilicic acid. Thus, hexafluorosilicic acid is hydrolyzed to give ammonium fluoride, which thermally decomposes to give the bifluoride:
- H
2[SiF
6] + 6 NH
3 + 2 H
2O → SiO
2 + 6 [NH
4]F - 2 [NH
4]F → NH
3 + [NH
4][HF
2]
The resulting ammonium bifluoride is converted to sodium bifluoride, which thermally decomposes to release HF.[6]
Ammonium bifluoride is also used as an additive in tin-nickel plating processes as the fluoride ion acts as a complexing agent with the tin, allowing for greater control over the resulting composition and finish.
Toxicity
Ammonium bifluoride is toxic to consume and a skin corrosion agent. Upon exposure to skin, rinsing with water followed by a treatment of calcium gluconate is required.[1] In water, ammonium bifluoride exists in chemical equilibrium with hydrofluoric acid and heating releases hydrogen fluoride gas.[7] Consequently, there is an equivalent toxicological risk as is present with hydrofluoric acid, and the same safety precautions apply.[8][7]
Ammonium bifluoride is used in some automotive wheel cleaning products. Many injuries have resulted in users not being aware of the risks posed by the products.[9] Ammonium bifluoride based products are often considered a safer alternative to hydrofluoric acid, yet still pose clear risks to the handler.[8] Ammonium bifluoride, ammonium fluoride, and hydrofluoric acid have been described as "too dangerous for any use in a car wash environment" by Professional Car Washing and Detailing magazine,[10] advice that accords with a 2015 report from the U.S. Centers for Disease Control and Prevention.[11]
References
- ↑ 1.0 1.1 1.2 1.3 Sigma-Aldrich Co., Ammonium bifluoride. Retrieved on 2013-07-20.
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 978-0-08-037941-8.
- ↑ Emsley, J. (1980) Very Strong Hydrogen Bonds, Chemical Society Reviews, 9, 91–124. doi:10.1039/CS9800900091
- ↑ McDonald, T. R. R. (1960). "The electron-density distribution in ammonium bifluoride". Acta Crystallogr. 13 (2): 113–124. doi:10.1107/S0365110X60000261. Bibcode: 1960AcCry..13..113M.
- ↑ ICSD Entry: 14140. Cambridge Crystallographic Data Centre. https://www.ccdc.cam.ac.uk/structures/Search?Ccdcid=1595541&DatabaseToSearch=Published. Retrieved 2022-06-25.
- ↑ Aigueperse, Jean; Mollard, Paul; Devilliers, Didier; Chemla, Marius; Faron, Robert; Romano, René; Cuer, Jean Pierre (2000). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a11_307.
- ↑ 7.0 7.1 National Industrial Chemicals Notification and Assessment Scheme (April 17, 2020). "Bifluorides: Human health tier II assessment". Department of Health (Australia). https://www.industrialchemicals.gov.au/sites/default/files/Bifluorides_Human%20health%20tier%20II%20assessment.pdf#page=2.
- ↑ 8.0 8.1 Genuino, Homer C.; Opembe, Naftali N.; Njagi, Eric C.; McClain, Skye; Suib, Steven L. (2012). "A review of hydrofluoric acid and its use in the car wash industry" (in en). Journal of Industrial and Engineering Chemistry 18 (5): 1529–1539. doi:10.1016/j.jiec.2012.03.001. ISSN 1226-086X. https://www.sciencedirect.com/science/article/pii/S1226086X12001189.
- ↑ Gormley, James (May 29, 2001). "The truth about ammonium bifluoride". Professional Car Washing and Detailing. http://www.carwash.com/article.asp?IndexID=6631269.
- ↑ Cook, Ryan (October 9, 2013). "The Five Factors of Clean: Chemistry, Part 1". Professional Car Washing and Detailing. https://www.carwash.com/the-five-factors-of-clean-chemistry-part-1/. Retrieved February 8, 2023.
- ↑ Woodie, Maria (December 1, 2015). "OSHA gives employees the right to know". Professional Car Washing and Detailing. https://www.carwash.com/osha-gives-emp-right-to-know/. Retrieved February 8, 2023.
 |