Biology:Morphine
 | |
| Error creating thumbnail: Unable to save thumbnail to destination | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈmɔːrfiːn/ |
| Trade names | Statex, MS Contin, MST Continus, Oramorph, Sevredol, and others[1] |
| AHFS/Drugs.com | Monograph |
| Pregnancy category |
|
| Dependence liability | High |
| Addiction liability | High[3] |
| Routes of administration | Inhalation (smoking), insufflation (snorting), by mouth (PO), rectal, subcutaneous (SC), intramuscular (IM), intravenous (IV), epidural, and intrathecal (IT) |
| Drug class | Opioid |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 20–40% (by mouth), 36–71% (rectally),[4] 100% (IV/IM) |
| Protein binding | 30–40% |
| Metabolism | Liver: UGT2B7 |
| Metabolites | • Morphine-3-glucuronide (90%) • Morphine-6-glucuronide (10%) |
| Onset of action | 5 minutes (IV), 15 minutes (IM),[5] 20 minutes (PO)[6] |
| Elimination half-life | 2–3 hours |
| Duration of action | 3–7 hours[7][8] |
| Excretion | Kidney 90%, bile duct 10% |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| Chemical and physical data | |
| Formula | C17H19NO3 |
| Molar mass | 285.343 g·mol−1 |
| 3D model (JSmol) | |
| Solubility in water | HCl & sulf.: 60 mg/mL (20 °C) |
| |
| |
| (verify) | |
Morphine is a strong opiate that is found naturally in opium, a dark brown resin produced by drying the latex of opium poppies (Papaver somniferum). It is mainly used as an analgesic (pain medication). There are numerous methods used to administer morphine: oral; sublingual; via inhalation; injection into a muscle, injection under the skin, or injection into the spinal cord area; transdermal; or via rectal suppository.[7][9] It acts directly on the central nervous system (CNS) to induce analgesia and alter perception and emotional response to pain. Physical and psychological dependence and tolerance may develop with repeated administration.[7] It can be taken for both acute pain and chronic pain and is frequently used for pain from myocardial infarction, kidney stones, and during labor.[7] Its maximum effect is reached after about 20 minutes when administered intravenously and 60 minutes when administered by mouth, while the duration of its effect is 3–7 hours.[7][8] Long-acting formulations of morphine are available as MS-Contin, Kadian, and other brand names as well as generically.[7]
Potentially serious side effects of morphine include decreased respiratory effort, vomiting, nausea, and low blood pressure.[7] Morphine is addictive and prone to abuse.[7] If one's dose is reduced after long-term use, opioid withdrawal symptoms may occur.[7] Common side effects of morphine include drowsiness, vomiting, and constipation.[7] Caution is advised for use of morphine during pregnancy or breast feeding, as it may affect the health of the baby.[7][2]
Morphine was first isolated between 1803 and 1805 by German pharmacist Friedrich Sertürner.[10] This is believed to be the first isolation of a medicinal alkaloid from a plant.[11] Merck began marketing it commercially in 1827.[10] Morphine was more widely used after the invention of the hypodermic syringe in 1853–1855.[10][12] Sertürner originally named the substance morphium, after the Greek god of dreams, Morpheus, as it has a tendency to cause sleep.[12][13]
The primary source of morphine is isolation from poppy straw of the opium poppy.[14] In 2013, approximately 523 tons of morphine were produced.[15] Approximately 45 tons were used directly for pain, an increase of 400% over the last twenty years.[15] Most use for this purpose was in the developed world.[15] About 70 percent of morphine is used to make other opioids such as hydromorphone, oxymorphone, and heroin.[15][16][17] It is a Schedule II drug in the United States,[16] Class A in the United Kingdom,[18] and Schedule I in Canada.[19] It is on the World Health Organization's List of Essential Medicines.[20] Morphine is sold under many brand names.[1] In 2020, it was the 140th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[21][22] It is available as a generic medication.[23]
Medical uses
Pain
Morphine is used primarily to treat both acute and chronic severe pain. Its duration of analgesia is about three to seven hours.[7][8] Side-effects of nausea and constipation are rarely severe enough to warrant stopping treatment.
It is used for pain due to myocardial infarction and for labor pains.[24] However, concerns exist that morphine may increase mortality in the event of non ST elevation myocardial infarction.[25]
Morphine has also traditionally been used in the treatment of acute pulmonary edema.[24] However, a 2006 review found little evidence to support this practice.[26]
A 2016 Cochrane review concluded that morphine is effective in relieving cancer pain.[27]
Shortness of breath
Morphine is beneficial in reducing the symptom of shortness of breath due to both cancer and noncancer causes.[28][29] In the setting of breathlessness at rest or on minimal exertion from conditions such as advanced cancer or end-stage cardiorespiratory diseases, regular, low-dose sustained-release morphine significantly reduces breathlessness safely, with its benefits maintained over time.[30][31]
Opioid use disorder
Morphine is also available as a slow-release formulation for opiate substitution therapy (OST) in Austria, Germany, Bulgaria, Slovenia, and Canada for persons with opioid addiction who cannot tolerate either methadone or buprenorphine.[32]
-
Two capsules (5 mg & 10 mg) of morphine sulfate extended-release
-
1 milliliter ampoule containing 10 mg of morphine
Contraindications
Relative contraindications to morphine include:
- respiratory depression when appropriate equipment is not available.[7]
- Although it has previously been thought that morphine was contraindicated in acute pancreatitis, a review of the literature shows no evidence for this.[33]
Adverse effects
- Common and short term
- Itchiness[34]
- Nausea[34]
- Vomiting[34]
- Constipation[34]
- Drowsiness[34]
- Dry mouth[34]
- Respiratory depression[7]
- Itching
- Other
- Opioid dependence
- Dizziness
- Decreased sex drive
- Loss of appetite
- Impaired sexual function
- Decreased testosterone levels
- Depression
- Immunodeficiency
- Opioid-induced abnormal pain sensitivity
- Irregular menstruation
- Increased risk of falls
- Slowed breathing
- Hallucinations

Constipation
Like loperamide and other opioids, morphine acts on the myenteric plexus in the intestinal tract, reducing gut motility, causing constipation. The gastrointestinal effects of morphine are mediated primarily by μ-opioid receptors in the bowel. By inhibiting gastric emptying and reducing propulsive peristalsis of the intestine, morphine decreases the rate of intestinal transit. Reduction in gut secretion and increased intestinal fluid absorption also contribute to the constipating effect. Opioids also may act on the gut indirectly through tonic gut spasms after inhibition of nitric oxide generation.[35] This effect was shown in animals when a nitric oxide precursor, L-arginine, reversed morphine-induced changes in gut motility.[36]
Hormone imbalance
Clinical studies consistently conclude that morphine, like other opioids, often causes hypogonadism and hormone imbalances in chronic users of both sexes. This side effect is dose-dependent and occurs in both therapeutic and recreational users. Morphine can interfere with menstruation by suppressing levels of luteinizing hormone. Many studies suggest the majority (perhaps as many as 90%) of chronic opioid users have opioid-induced hypogonadism. This effect may cause the increased likelihood of osteoporosis and bone fracture observed in chronic morphine users. Studies suggest the effect is temporary. As of 2013[update], the effect of low-dose or acute use of morphine on the endocrine system is unclear.[37][38]
Effects on human performance
Most reviews conclude that opioids produce minimal impairment of human performance on tests of sensory, motor, or attentional abilities. However, recent studies have been able to show some impairments caused by morphine, which is not surprising, given that morphine is a central nervous system depressant. Morphine has resulted in impaired functioning on critical flicker frequency (a measure of overall CNS arousal) and impaired performance on the Maddox wing test (a measure of the deviation of the visual axes of the eyes). Few studies have investigated the effects of morphine on motor abilities; a high dose of morphine can impair finger tapping and the ability to maintain a low constant level of isometric force (i.e. fine motor control is impaired),[39] though no studies have shown a correlation between morphine and gross motor abilities.
In terms of cognitive abilities, one study has shown that morphine may have a negative impact on anterograde and retrograde memory,[40] but these effects are minimal and transient. Overall, it seems that acute doses of opioids in non-tolerant subjects produce minor effects in some sensory and motor abilities, and perhaps also in attention and cognition. It is likely that the effects of morphine will be more pronounced in opioid-naive subjects than chronic opioid users.
In chronic opioid users, such as those on Chronic Opioid Analgesic Therapy (COAT) for managing severe, chronic pain, behavioural testing has shown normal functioning on perception, cognition, coordination and behaviour in most cases. One 2000 study[41] analysed COAT patients to determine whether they were able to safely operate a motor vehicle. The findings from this study suggest that stable opioid use does not significantly impair abilities inherent in driving (this includes physical, cognitive and perceptual skills). COAT patients showed rapid completion of tasks that require the speed of responding for successful performance (e.g., Rey Complex Figure Test) but made more errors than controls. COAT patients showed no deficits in visual-spatial perception and organization (as shown in the WAIS-R Block Design Test) but did show impaired immediate and short-term visual memory (as shown on the Rey Complex Figure Test – Recall). These patients showed no impairments in higher-order cognitive abilities (i.e., planning). COAT patients appeared to have difficulty following instructions and showed a propensity toward impulsive behaviour, yet this did not reach statistical significance. It is important to note that this study reveals that COAT patients have no domain-specific deficits, which supports the notion that chronic opioid use has minor effects on psychomotor, cognitive, or neuropsychological functioning.
Reinforcement disorders
Addiction

Morphine is a highly addictive substance. Numerous studies, including one by The Lancet, ranked Morphine/Heroin as #1 most addictive substance, followed by Cocaine at #2, Nicotine #3, followed by Barbiturates at 4 and ethanol at 5. In controlled studies comparing the physiological and subjective effects of heroin and morphine in individuals formerly addicted to opiates, subjects showed no preference for one drug over the other. Equipotent, injected doses had comparable action courses, with diacetylmorphine crossing the BBB slightly quicker. No difference in subjects' self-rated feelings of euphoria, ambition, nervousness, relaxation, drowsiness, or sleepiness.[42] Short-term addiction studies by the same researchers demonstrated that tolerance developed at a similar rate to both heroin and morphine. When compared to the opioids hydromorphone, fentanyl, oxycodone, and pethidine/meperidine, former addicts showed a strong preference for heroin and morphine, suggesting that heroin and morphine are particularly susceptible to abuse and addiction. Morphine and heroin also produced higher rates of euphoria and other positive subjective effects when compared to these other opioids.[42] The choice of heroin and morphine over other opioids by former drug addicts may also be because heroin (also known as morphine diacetate, diamorphine, or diacetyl morphine) is an ester of morphine and a morphine prodrug, essentially meaning they are identical drugs in vivo. Heroin is converted to morphine before binding to the opioid receptors in the brain and spinal cord, where morphine causes the subjective effects, which is what the addicted individuals are seeking.[43]
Tolerance
Several hypotheses are given about how tolerance develops, including opioid receptor phosphorylation (which would change the receptor conformation), functional decoupling of receptors from G-proteins (leading to receptor desensitization),[44] μ-opioid receptor internalization or receptor down-regulation (reducing the number of available receptors for morphine to act on), and upregulation of the cAMP pathway (a counterregulatory mechanism to opioid effects) (For a review of these processes, see Koch and Hollt.[45])
Dependence and withdrawal
Cessation of dosing with morphine creates the prototypical opioid withdrawal syndrome, which, unlike that of barbiturates, benzodiazepines, alcohol, or sedative-hypnotics, is not fatal by itself in otherwise healthy people.
Acute morphine withdrawal, along with that of any other opioid, proceeds through a number of stages. Other opioids differ in the intensity and length of each, and weak opioids and mixed agonist-antagonists may have acute withdrawal syndromes that do not reach the highest level. As commonly cited[by whom?], they are:
- Stage I, 6 h to 14 h after last dose: Drug craving, anxiety, irritability, perspiration, and mild to moderate dysphoria
- Stage II, 14 h to 18 h after last dose: Yawning, heavy perspiration, mild depression, lacrimation, crying, headaches, runny nose, dysphoria, also intensification of the above symptoms, "yen sleep" (a waking trance-like state)
- Stage III, 16 h to 24 h after last dose: Rhinorrhea (runny nose) and increase in other of the above, dilated pupils, piloerection (goose bumps – a purported origin of the phrase, 'cold turkey,' but in fact the phrase originated outside of drug treatment),[46] muscle twitches, hot flashes, cold flashes, aching bones and muscles, loss of appetite, and the beginning of intestinal cramping
- Stage IV, 24 h to 36 h after last dose: Increase in all of the above including severe cramping and involuntary leg movements ("kicking the habit" also called restless leg syndrome), loose stool, insomnia, elevation of blood pressure, moderate elevation in body temperature, increase in frequency of breathing and tidal volume, tachycardia (elevated pulse), restlessness, nausea
- Stage V, 36 h to 72 h after last dose: Increase in the above, fetal position, vomiting, free and frequent liquid diarrhea, which sometimes can accelerate the time of passage of food from mouth to out of system, weight loss of 2 kg to 5 kg per 24 h, increased white cell count, and other blood changes
- Stage VI, after completion of above: Recovery of appetite and normal bowel function, beginning of transition to post acute and chronic symptoms that are mainly psychological, but may also include increased sensitivity to pain, hypertension, colitis or other gastrointestinal afflictions related to motility, and problems with weight control in either direction
In advanced stages of withdrawal, ultrasonographic evidence of pancreatitis has been demonstrated in some patients and is presumably attributed to spasm of the pancreatic sphincter of Oddi.[47]
The withdrawal symptoms associated with morphine addiction are usually experienced shortly before the time of the next scheduled dose, sometimes within as early as a few hours (usually 6 h to 12 h) after the last administration. Early symptoms include watery eyes, insomnia, diarrhea, runny nose, yawning, dysphoria, sweating, and in some cases a strong drug craving. Severe headache, restlessness, irritability, loss of appetite, body aches, severe abdominal pain, nausea and vomiting, tremors, and even stronger and more intense drug craving appear as the syndrome progresses. Severe depression and vomiting are very common. During the acute withdrawal period, systolic and diastolic blood pressures increase, usually beyond premorphine levels, and heart rate increases,[48] which have potential to cause a heart attack, blood clot, or stroke.
Chills or cold flashes with goose bumps ("cold turkey") alternating with flushing (hot flashes), kicking movements of the legs ("kicking the habit"[43]) and excessive sweating are also characteristic symptoms.[49] Severe pains in the bones and muscles of the back and extremities occur, as do muscle spasms. At any point during this process, a suitable narcotic can be administered that will dramatically reverse the withdrawal symptoms. Major withdrawal symptoms peak between 48 h and 96 h after the last dose and subside after about 8 to 12 days. Sudden withdrawal by heavily dependent users who are in poor health is very rarely fatal. Morphine withdrawal is considered less dangerous than alcohol, barbiturate, or benzodiazepine withdrawal.[50][51]
The psychological dependence associated with morphine addiction is complex and protracted. Long after the physical need for morphine has passed, the addict will usually continue to think and talk about the use of morphine (or other drugs) and feel strange or overwhelmed coping with daily activities without being under the influence of morphine. Psychological withdrawal from morphine is usually a very long and painful process. Addicts often experience severe depression, anxiety, insomnia, mood swings, amnesia (forgetfulness), low self-esteem, confusion, paranoia, and other psychological disorders. Without intervention, the syndrome will run its course, and most of the overt physical symptoms will disappear within 7 to 10 days including psychological dependence. A high probability of relapse exists after morphine withdrawal when neither the physical environment nor the behavioral motivators that contributed to the abuse have been altered. Testimony to morphine's addictive and reinforcing nature is its relapse rate. Abusers of morphine (and heroin) have one of the highest relapse rates among all drug users, ranging up to 98% in the estimation of some medical experts.[52]
Toxicity
| Properties of Morphine | |||||
|---|---|---|---|---|---|
| Molar mass[53] | 285.338 g/mol | ||||
| Acidity (pKa)[53] |
| ||||
| Solubility[53] | 0.15 g/L at 20 °C | ||||
| Melting point[53] | 255 °C | ||||
| Boiling point[53] | 190 °C sublimes | ||||
A large overdose can cause asphyxia and death by respiratory depression if the person does not receive medical attention immediately.[54] Overdose treatment includes the administration of naloxone. The latter completely reverses morphine's effects, but may result in immediate onset of withdrawal in opiate-addicted subjects. Multiple doses may be needed as the duration of action of morphine is longer than that of naloxone.[55]
Pharmacology
Pharmacodynamics
| Compound | Affinities (Ki) | Ratio | Ref | ||
|---|---|---|---|---|---|
| MOR | DOR | KOR | MOR:DOR:KOR | ||
| Morphine | 1.8 nM | 90 nM | 317 nM | 1:50:176 | [56] |
| (−)-Morphine | 1.24 nM | 145 nM | 23.4 nM | 1:117:19 | [57] |
| (+)-Morphine | >10 μM | >100 μM | >300 μM | ND | [57] |
| Compound | Route | Dose |
|---|---|---|
| Codeine | PO | 200 mg |
| Hydrocodone | PO | 20–30 mg |
| Hydromorphone | PO | 7.5 mg |
| Hydromorphone | IV | 1.5 mg |
| Morphine | PO | 30 mg |
| Morphine | IV | 10 mg |
| Oxycodone | PO | 20 mg |
| Oxycodone | IV | 10 mg |
| Oxymorphone | PO | 10 mg |
| Oxymorphone | IV | 1 mg |
Due to its long history and established use as a pain medication, this compound has become the benchmark to which all other opioids are compared.[61] It interacts predominantly with the μ–δ-opioid (Mu-Delta) receptor heteromer.[62][63] The μ-binding sites are discretely distributed in the human brain, with high densities in the posterior amygdala, hypothalamus, thalamus, nucleus caudatus, putamen, and certain cortical areas. They are also found on the terminal axons of primary afferents within laminae I and II (substantia gelatinosa) of the spinal cord and in the spinal nucleus of the trigeminal nerve.[64]
Morphine is a phenanthrene opioid receptor agonist – its main effect is binding to and activating the μ-opioid receptor (MOR) in the central nervous system. Its intrinsic activity at the MOR is heavily dependent on the assay and tissue being tested; in some situations it is a full agonist while in others it can be a partial agonist or even antagonist.[65] In clinical settings, morphine exerts its principal pharmacological effect on the central nervous system and gastrointestinal tract. Its primary actions of therapeutic value are analgesia and sedation. Activation of the MOR is associated with analgesia, sedation, euphoria, physical dependence, and respiratory depression. Morphine is also a κ-opioid receptor (KOR) and δ-opioid receptor (DOR) agonist. Activation of the KOR is associated with spinal analgesia, miosis (pinpoint pupils), and psychotomimetic effects. The DOR is thought to play a role in analgesia.[64][failed verification] Although morphine does not bind to the σ receptor, it has been shown that σ receptor agonists, such as (+)-pentazocine, inhibit morphine analgesia, and σ receptor antagonists enhance morphine analgesia,[66] suggesting downstream involvement of the σ receptor in the actions of morphine.
The effects of morphine can be countered with opioid receptor antagonists such as naloxone and naltrexone; the development of tolerance to morphine may be inhibited by NMDA receptor antagonists such as ketamine, dextromethorphan, and memantine.[67][68] The rotation of morphine with chemically dissimilar opioids in the long-term treatment of pain will slow down the growth of tolerance in the longer run, particularly agents known to have significantly incomplete cross-tolerance with morphine such as levorphanol, ketobemidone, piritramide, and methadone and its derivatives; all of these drugs also have NMDA antagonist properties. It is believed that the strong opioid with the most incomplete cross-tolerance with morphine is either methadone[69] or dextromoramide.[citation needed]

Gene expression
Studies have shown that morphine can alter the expression of a number of genes. A single injection of morphine has been shown to alter the expression of two major groups of genes, for proteins involved in mitochondrial respiration and for cytoskeleton-related proteins.[70]
Effects on the immune system
Morphine has long been known to act on receptors expressed on cells of the central nervous system resulting in pain relief and analgesia. In the 1970s and '80s, evidence suggesting that opioid drug addicts show increased risk of infection (such as increased pneumonia, tuberculosis, and HIV/AIDS) led scientists to believe that morphine may also affect the immune system. This possibility increased interest in the effect of chronic morphine use on the immune system.[71]
The first step of determining that morphine may affect the immune system was to establish that the opiate receptors known to be expressed on cells of the central nervous system are also expressed on cells of the immune system. One study successfully showed that dendritic cells, part of the innate immune system, display opiate receptors. Dendritic cells are responsible for producing cytokines, which are the tools for communication in the immune system. This same study showed that dendritic cells chronically treated with morphine during their differentiation produce more interleukin-12 (IL-12), a cytokine responsible for promoting the proliferation, growth, and differentiation of T-cells (another cell of the adaptive immune system) and less interleukin-10 (IL-10), a cytokine responsible for promoting a B-cell immune response (B cells produce antibodies to fight off infection).[72]
This regulation of cytokines appear to occur via the p38 MAPKs (mitogen-activated protein kinase)-dependent pathway. Usually, the p38 within the dendritic cell expresses TLR 4 (toll-like receptor 4), which is activated through the ligand LPS (lipopolysaccharide). This causes the p38 MAPK to be phosphorylated. This phosphorylation activates the p38 MAPK to begin producing IL-10 and IL-12. When the dendritic cells are chronically exposed to morphine during their differentiation process then treated with LPS, the production of cytokines is different. Once treated with morphine, the p38 MAPK does not produce IL-10, instead favoring production of IL-12. The exact mechanism through which the production of one cytokine is increased in favor over another is not known. Most likely, the morphine causes increased phosphorylation of the p38 MAPK. Transcriptional level interactions between IL-10 and IL-12 may further increase the production of IL-12 once IL-10 is not being produced. This increased production of IL-12 causes increased T-cell immune response.
Further studies on the effects of morphine on the immune system have shown that morphine influences the production of neutrophils and other cytokines. Since cytokines are produced as part of the immediate immunological response (inflammation), it has been suggested that they may also influence pain. In this way, cytokines may be a logical target for analgesic development. Recently, one study has used an animal model (hind-paw incision) to observe the effects of morphine administration on the acute immunological response. Following hind-paw incision, pain thresholds and cytokine production were measured. Normally, cytokine production in and around the wounded area increases in order to fight infection and control healing (and, possibly, to control pain), but pre-incisional morphine administration (0.1 mg/kg to 10.0 mg/kg) reduced the number of cytokines found around the wound in a dose-dependent manner. The authors suggest that morphine administration in the acute post-injury period may reduce resistance to infection and may impair the healing of the wound.[73]
Pharmacokinetics
Absorption and metabolism
Morphine can be taken orally, sublingually, bucally, rectally, subcutaneously, intranasally, intravenously, intrathecally or epidurally and inhaled via a nebulizer. As a recreational drug, it is becoming more common to inhale ("Chasing the Dragon"), but, for medical purposes, intravenous (IV) injection is the most common method of administration. Morphine is subject to extensive first-pass metabolism (a large proportion is broken down in the liver), so, if taken orally, only 40% to 50% of the dose reaches the central nervous system. Resultant plasma levels after subcutaneous (SC), intramuscular (IM), and IV injection are all comparable. After IM or SC injections, morphine plasma levels peak in approximately 20 min, and, after oral administration, levels peak in approximately 30 min.[74] Morphine is metabolised primarily in the liver and approximately 87% of a dose of morphine is excreted in the urine within 72 h of administration. Morphine is metabolized primarily into morphine-3-glucuronide (M3G) and morphine-6-glucuronide (M6G)[75] via glucuronidation by phase II metabolism enzyme UDP-glucuronosyl transferase-2B7 (UGT2B7). About 60% of morphine is converted to M3G, and 6% to 10% is converted to M6G.[76] Not only does the metabolism occur in the liver but it may also take place in the brain and the kidneys. M3G does not undergo opioid receptor binding and has no analgesic effect. M6G binds to μ-receptors and is half as potent an analgesic as morphine in humans.[76] Morphine may also be metabolized into small amounts of normorphine, codeine, and hydromorphone. Metabolism rate is determined by gender, age, diet, genetic makeup, disease state (if any), and use of other medications. The elimination half-life of morphine is approximately 120 min, though there may be slight differences between men and women. Morphine can be stored in fat, and, thus, can be detectable even after death. Morphine can cross the blood–brain barrier, but, because of poor lipid solubility, protein binding, rapid conjugation with glucuronic acid and ionization, it does not cross easily. Heroin, which is derived from morphine, crosses the blood–brain barrier more easily, making it more potent.[77]
Extended-release
There are extended-release formulations of orally administered morphine whose effect last longer, which can be given once per day. Brand names for this formulation of morphine include Avinza,[78] Kadian,[78] MS Contin[78] and Dolcontin.[79] For constant pain, the relieving effect of extended-release morphine given once (for Kadian)[80] or twice (for MS Contin)[80] every 24 hours is roughly the same as multiple administrations of immediate release (or "regular") morphine.[81] Extended-release morphine can be administered together with "rescue doses" of immediate-release morphine as needed in case of breakthrough pain, each generally consisting of 5% to 15% of the 24-hour extended-release dosage.[81]
Detection in body fluids
Morphine and its major metabolites, morphine-3-glucuronide and morphine-6-glucuronide, can be detected in blood, plasma, hair, and urine using an immunoassay. Chromatography can be used to test for each of these substances individually. Some testing procedures hydrolyze metabolic products into morphine before the immunoassay, which must be considered when comparing morphine levels in separately published results. Morphine can also be isolated from whole blood samples by solid phase extraction (SPE) and detected using liquid chromatography-mass spectrometry (LC-MS).
Ingestion of codeine or food containing poppy seeds can cause false positives.[82]
A 1999 review estimated that relatively low doses of heroin (which metabolizes immediately into morphine) are detectable by standard urine tests for 1–1.5 days after use.[83] A 2009 review determined that, when the analyte is morphine and the limit of detection is 1 ng/ml, a 20 mg intravenous (IV) dose of morphine is detectable for 12–24 hours. A limit of detection of 0.6 ng/ml had similar results.[84]
Chirality and biological activity
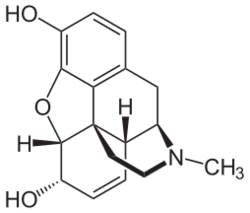
Morphine is a pentacyclic 3°amine (alkaloid) with 5 stereogenic centers and exists in 32 stereoisomeric forms. But the desired analgesic activity resides exclusively in the natural product, the (-)-enantiomer with the configuration (5R,6S,9R,13S,14R).[85][86]
Natural occurrence

Morphine is the most abundant opiate found in opium, the dried latex extracted by shallowly scoring the unripe seedpods of the Papaver somniferum poppy. Morphine is generally 8–14% of the dry weight of opium.[87] Przemko and Norman cultivars of the opium poppy, are used to produce two other alkaloids, thebaine and oripavine, which are used in the manufacture of semi-synthetic and synthetic opioids like oxycodone and etorphine. P. bracteatum does not contain morphine or codeine, or other narcotic phenanthrene-type, alkaloids. This species is rather a source of thebaine.[88] Occurrence of morphine in other Papaverales and Papaveraceae, as well as in some species of hops and mulberry trees has not been confirmed. Morphine is produced most predominantly early in the life cycle of the plant. Past the optimum point for extraction, various processes in the plant produce codeine, thebaine, and in some cases negligible amounts of hydromorphone, dihydromorphine, dihydrocodeine, tetrahydro-thebaine, and hydrocodone (these compounds are rather synthesized from thebaine and oripavine).
In the brain of mammals, morphine is detectable in trace steady-state concentrations.[9] The human body also produces endorphins, which are chemically related endogenous opioid peptides that function as neuropeptides and have similar effects to morphine.[89]
Human biosynthesis
Morphine is an endogenous opioid in humans. Various human cells are capable of synthesizing and releasing it, including white blood cells.[9][90][91] The primary biosynthetic pathway for morphine in humans consists of:[9]
- L-tyrosine → para-tyramine or L-DOPA → Dopamine
- L-tyrosine → L-DOPA → 3,4-dihydroxyphenylacetaldehyde (DOPAL)
- Dopamine + DOPAL → (S)-norlaudanosoline →→→ (S)-reticuline → 1,2-dehydroreticulinium → (R)-reticuline → salutaridine → salutaridinol → thebaine → neopinone → codeinone → codeine → morphine
The intermediate (S)-norlaudanosoline (also known as tetrahydropapaveroline) is synthesized through addition of DOPAL and dopamine.[9] CYP2D6, a cytochrome P450 isoenzyme is involved in two steps along the biosynthetic pathway, catalyzing both the biosynthesis of dopamine from tyramine and of morphine from codeine.[9][92]
Urinary concentrations of endogenous codeine and morphine have been found to significantly increase in individuals taking L-DOPA for the treatment of Parkinson's disease.[9]
Biosynthesis in the opium poppy
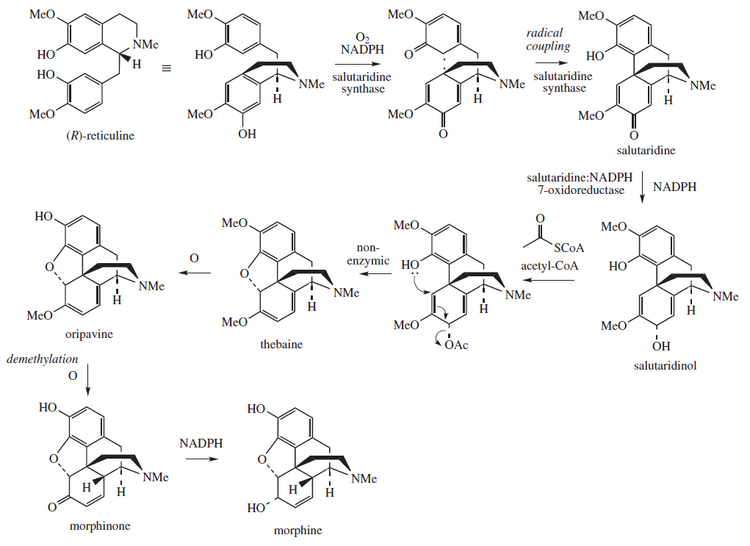
Morphine is biosynthesized in the opium poppy from the tetrahydroisoquinoline reticuline. It is converted into salutaridine, thebaine, and oripavine. The enzymes involved in this process are the salutaridine synthase, salutaridine:NADPH 7-oxidoreductase and the codeinone reductase.[93] Researchers are attempting to reproduce the biosynthetic pathway that produces morphine in genetically engineered yeast.[94] In June 2015 the S-reticuline could be produced from sugar and R-reticuline could be converted to morphine, but the intermediate reaction could not be performed.[95] In August 2015 the first complete synthesis of thebaine and hydrocodone in yeast were reported, but the process would need to be 100,000 times more productive to be suitable for commercial use.[96][97]
Chemistry
This section has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
(Learn how and when to remove this template message)No issues specified. Please specify issues, or remove this template. |
Elements of the morphine structure have been used to create completely synthetic drugs such as the morphinan family (levorphanol, dextromethorphan and others) and other groups that have many members with morphine-like qualities.[citation needed] The modification of morphine and the aforementioned synthetics has also given rise to non-narcotic drugs with other uses such as emetics, stimulants, antitussives, anticholinergics, muscle relaxants, local anaesthetics, general anaesthetics, and others.[citation needed] Morphine-derived agonist–antagonist drugs have also been developed.[citation needed]
Structure description
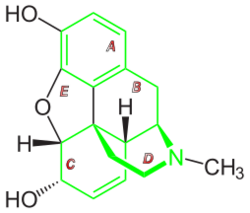


Morphine is a benzylisoquinoline alkaloid with two additional ring closures.[98] As Jack DeRuiter of the Department of Drug Discovery and Development (formerly, Pharmacal Sciences), Harrison School of Pharmacy, Auburn University stated in his Fall 2000 course notes for that earlier department's "Principles of Drug Action 2" course, "Examination of the morphine molecule reveals the following structural features important to its pharmacological profile...
- A rigid pentacyclic structure consisting of a benzene ring (A), two partially unsaturated cyclohexane rings (B and C), a piperidine ring (D) and a tetrahydrofuran ring (E). Rings A, B and C are the phenanthrene ring system. This ring system has little conformational flexibility...
- Two hydroxyl functional groups: a C3-phenolic [hydroxyl group] (pKa 9.9) and a C6-allylic [hydroxyl group],
- An ether linkage between E4 and E5,
- Unsaturation between C7 and C8,
- A basic, [tertiary]-amine function at position 17, [and]
- [Five] centers of chirality (C5, C6, C9, C13 and C14) with morphine exhibiting a high degree of stereoselectivity of analgesic action."[99][better source needed][needs update]
Morphine and most of its derivatives do not exhibit optical isomerism, although some more distant relatives like the morphinan series (levorphanol, dextorphan and the racemic parent chemical racemorphan) do,[100] and as noted above stereoselectivity in vivo is an important issue.[citation needed]
Uses and derivatives
Most of the licit morphine produced is used to make codeine by methylation.[101] It is also a precursor for many drugs including heroin (3,6-diacetylmorphine), hydromorphone (dihydromorphinone), and oxymorphone (14-hydroxydihydromorphinone).[102] Most semi-synthetic opioids, both of the morphine and codeine subgroups, are created by modifying one or more of the following:[citation needed]
- Halogenating or making other modifications at positions 1 or 2 on the morphine carbon skeleton.
- The methyl group that makes morphine into codeine can be removed or added back, or replaced with another functional group like ethyl and others to make codeine analogues of morphine-derived drugs and vice versa. Codeine analogues of morphine-based drugs often serve as prodrugs of the stronger drug, as in codeine and morphine, hydrocodone and hydromorphone, oxycodone and oxymorphone, nicocodeine and nicomorphine, dihydrocodeine and dihydromorphine, etc.
- Saturating, opening, or other changes to the bond between positions 7 and 8, as well as adding, removing, or modifying functional groups to these positions; saturating, reducing, eliminating, or otherwise modifying the 7–8 bond and attaching a functional group at 14 yields hydromorphinol; the oxidation of the hydroxyl group to a carbonyl and changing the 7–8 bond to single from double changes codeine into oxycodone.
- Attachment, removal or modification of functional groups to positions 3 or 6 (dihydrocodeine and related, hydrocodone, nicomorphine); in the case of moving the methyl functional group from position 3 to 6, codeine becomes heterocodeine, which is 72 times stronger, and therefore six times stronger than morphine
- Attachment of functional groups or other modification at position 14 (oxymorphone, oxycodone, naloxone)
- Modifications at positions 2, 4, 5 or 17, usually along with other changes to the molecule elsewhere on the morphine skeleton. Often this is done with drugs produced by catalytic reduction, hydrogenation, oxidation, or the like, producing strong derivatives of morphine and codeine.
Many morphine derivatives can also be manufactured using thebaine or codeine as a starting material.[citation needed] Replacement of the N-methyl group of morphine with an N-phenylethyl group results in a product that is 18 times more powerful than morphine in its opiate agonist potency.[citation needed] Combining this modification with the replacement of the 6-hydroxyl with a 6-methylene group produces a compound some 1,443 times more potent than morphine, stronger than the Bentley compounds such as etorphine (M99, the Immobilon tranquilliser dart) by some measures.[citation needed] Closely related to morphine are the opioids morphine-N-oxide (genomorphine), which is a pharmaceutical that is no longer in common use;[citation needed] and pseudomorphine, an alkaloid that exists in opium, form as degradation products of morphine.[citation needed]
As a result of the extensive study and use of this molecule, more than 250 morphine derivatives (also counting codeine and related drugs) have been developed since the last quarter of the 19th century.[citation needed] These drugs range from 25% the analgesic strength of codeine (or slightly more than 2% of the strength of morphine) to several thousand times the strength of morphine, to powerful opioid antagonists, including naloxone (Narcan), naltrexone (Trexan), diprenorphine (M5050, the reversing agent for the Immobilon dart) and nalorphine (Nalline).[citation needed] Some opioid agonist-antagonists, partial agonists, and inverse agonists are also derived from morphine.[citation needed] The receptor-activation profile of the semi-synthetic morphine derivatives varies widely and some, like apomorphine are devoid of narcotic effects.[citation needed]
Chemical salts of Morphine
Both morphine and its hydrated form are sparingly soluble in water.[103] For this reason, pharmaceutical companies produce sulfate and hydrochloride salts of the drug, both of which are over 300 times more water-soluble than their parent molecule.[clarification needed][citation needed] Whereas the pH of a saturated morphine hydrate solution is 8.5, the salts are acidic.[citation needed] Since they derive from a strong acid but weak base, they are both at about pH = 5;[clarification needed][citation needed] as a consequence, the morphine salts are mixed with small amounts of NaOH to make them suitable for injection.[citation needed]
A number of salts of morphine are used, with the most common in current clinical use being the hydrochloride, sulfate, tartrate, and citrate;[citation needed] less commonly methobromide, hydrobromide, hydroiodide, lactate, chloride, and bitartrate and the others listed below.[citation needed] Morphine diacetate (heroin) is not a salt, but rather a further derivative,[citation needed] see above.[104]
Morphine meconate is a major form of the alkaloid in the poppy, as is morphine pectinate, nitrate, sulfate, and some others.[citation needed] Like codeine, dihydrocodeine and other (especially older) opiates, morphine has been used as the salicylate salt by some suppliers and can be easily compounded, imparting the therapeutic advantage of both the opioid and the NSAID;[citation needed] multiple barbiturate salts of morphine were also used in the past, as was/is morphine valerate, the salt of the acid being the active principle of valerian.[citation needed] Calcium morphenate is the intermediate in various latex and poppy-straw methods of morphine production, more rarely sodium morphenate takes its place.[citation needed] Morphine ascorbate and other salts such as the tannate, citrate, and acetate, phosphate, valerate and others may be present in poppy tea depending on the method of preparation.[citation needed][105]
The salts listed by the United States Drug Enforcement Administration for reporting purposes, in addition to a few others, are as follows:[citation needed]
| Select salts of morphine with their Controlled Substances Act (CSA) schedule, Administrative Controlled Substances Code Number (ACSCN), and free base conversion ratio.[clarification needed][citation needed] | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Production

In the opium poppy, the alkaloids are bound to meconic acid. The method is to extract from the crushed plant with diluted sulfuric acid, which is a stronger acid than meconic acid, but not so strong to react with alkaloid molecules. The extraction is performed in many steps (one amount of crushed plant is extracted at least six to ten times, so practically every alkaloid goes into the solution). From the solution obtained at the last extraction step, the alkaloids are precipitated by either ammonium hydroxide or sodium carbonate. The last step is purifying and separating morphine from other opium alkaloids. The somewhat similar Gregory process was developed in the United Kingdom during the Second World War, which begins with stewing the entire plant, in most cases save the roots and leaves, in plain or mildly acidified water, then proceeding through steps of concentration, extraction, and purification of alkaloids.[citation needed] Other methods of processing "poppy straw" (i.e., dried pods and stalks) use steam, one or more of several types of alcohol, or other organic solvents.
The poppy straw methods predominate in Continental Europe and the British Commonwealth, with the latex method in most common use in India. The latex method can involve either vertical or horizontal slicing of the unripe pods with a two-to five-bladed knife with a guard developed specifically for this purpose to the depth of a fraction of a millimetre and scoring of the pods can be done up to five times. An alternative latex method sometimes used in China in the past is to cut off the poppy heads, run a large needle through them, and collect the dried latex 24 to 48 hours later.[citation needed]
In India, opium harvested by licensed poppy farmers is dehydrated to uniform levels of hydration at government processing centers, and then sold to pharmaceutical companies that extract morphine from the opium. However, in Turkey and Tasmania, morphine is obtained by harvesting and processing the fully mature dry seed pods with attached stalks, called poppy straw. In Turkey, a water extraction process is used, while in Tasmania, a solvent extraction process is used.[citation needed]
Opium poppy contains at least 50 different alkaloids, but most of them are of very low concentration. Morphine is the principal alkaloid in raw opium and constitutes roughly 8–19% of opium by dry weight (depending on growing conditions).[77] Some purpose-developed strains of poppy now produce opium that is up to 26% morphine by weight.[citation needed] A rough rule of thumb to determine the morphine content of pulverised dried poppy straw is to divide the percentage expected for the strain or crop via the latex method by eight or an empirically determined factor, which is often in the range of 5 to 15.[citation needed] The Norman strain of P. somniferum, also developed in Tasmania, produces down to 0.04% morphine but with much higher amounts of thebaine and oripavine, which can be used to synthesise semi-synthetic opioids as well as other drugs like stimulants, emetics, opioid antagonists, anticholinergics, and smooth-muscle agents.[citation needed]
In the 1950s and 1960s, Hungary supplied nearly 60% of Europe's total medication-purpose morphine production. To this day, poppy farming is legal in Hungary, but poppy farms are limited by law to 2 acres (8,100 m2). It is also legal to sell dried poppy in flower shops for use in floral arrangements.
It was announced in 1973 that a team at the National Institutes of Health in the United States had developed a method for total synthesis of morphine, codeine, and thebaine using coal tar as a starting material. A shortage in codeine-hydrocodone class cough suppressants (all of which can be made from morphine in one or more steps, as well as from codeine or thebaine) was the initial reason for the research.
Most morphine produced for pharmaceutical use around the world is actually converted into codeine as the concentration of the latter in both raw opium and poppy straw is much lower than that of morphine; in most countries, the usage of codeine (both as end-product and precursor) is at least equal or greater than that of morphine on a weight basis.
Chemical synthesis
The first morphine total synthesis, devised by Marshall D. Gates, Jr. in 1952, remains a widely used example of total synthesis.[106] Several other syntheses were reported, notably by the research groups of Rice,[107] Evans,[108] Fuchs,[109] Parker,[110] Overman,[111] Mulzer-Trauner,[112] White,[113] Taber,[114] Trost,[115] Fukuyama,[116] Guillou,[117] and Stork.[118] Because of the stereochemical complexity and consequent synthetic challenge presented by this polycyclic structure, Michael Freemantle has expressed the view that it is "highly unlikely" that a chemical synthesis will ever be cost-effective such that it could compete with the cost of producing morphine from the opium poppy.[119]
GMO synthesis
Research
Thebaine has been produced by GMO E. coli.[120]
Precursor to other opioids
Pharmaceutical
Morphine is a precursor in the manufacture in a number of opioids such as dihydromorphine, hydromorphone, hydrocodone, and oxycodone as well as codeine, which itself has a large family of semi-synthetic derivatives.[121]
Illicit
Illicit morphine is produced, though rarely, from codeine found in over-the-counter cough and pain medicines.[citation needed] Another illicit source is morphine extracted from extended-release morphine products.[122] Chemical reactions can then be used to convert morphine, dihydromorphine, and hydrocodone into heroin or other opioids [e.g., diacetyldihydromorphine (Paralaudin), and thebacon].[citation needed] Other clandestine conversions—of morphine, into ketones of the hydromorphone class, or other derivatives like dihydromorphine (Paramorfan), desomorphine (Permonid), metopon, etc., and of codeine into hydrocodone (Dicodid), dihydrocodeine (Paracodin), etc. —require greater expertise, and types and quantities of chemicals and equipment that are more difficult to source, and so are more rarely used, illicitly (but cases have been recorded).[citation needed]
History

The earliest known reference to morphine can be traced back to Theophrastus in the 3rd Century BC, however, possible references to morphine may go as far back as 2100 BC as Sumerian clay tablets which records lists of medical prescriptions include opium-based cures.[123]
An opium-based elixir has been ascribed to alchemists of Byzantine times, but the specific formula was lost during the Ottoman conquest of Constantinople (Istanbul).[124] Around 1522, Paracelsus made reference to an opium-based elixir that he called laudanum from the Latin word laudāre, meaning "to praise". He described it as a potent painkiller, but recommended that it be used sparingly. The recipe given differs substantially from that of modern-day laudanum.[125]
Morphine was discovered as the first active alkaloid extracted from the opium poppy plant in December 1804 in Paderborn by German pharmacist Friedrich Sertürner.[11][126] In 1817, Sertürner reported experiments in which he administered morphine to himself, three young boys, three dogs, and a mouse; all four people almost died.[127] Sertürner originally named the substance morphium after the Greek god of dreams, Morpheus, as it has a tendency to cause sleep.[12][128] Sertürner's morphium was six times stronger than opium. He hypothesized that, because lower doses of the drug were needed, it would be less addictive. However Sertürner became addicted to the drug, warning that "I consider it my duty to attract attention to the terrible effects of this new substance I called morphium in order that calamity may be averted."[129]
The drug was first marketed to the general public by Sertürner and Company in 1817 as a pain medication, and also as a treatment for opium and alcohol addiction. It was first used as a poison in 1822 when Edme Castaing of France was convicted of murdering a patient.[130] Commercial production began in Darmstadt, Germany, in 1827 by the pharmacy that became the pharmaceutical company Merck, with morphine sales being a large part of their early growth.[131][132] In the 1850s, Alexander Wood reported that he had injected morphine into his wife Rebecca as an experiment; the myth goes that this killed her because of respiratory depression,[127] but she outlived her husband by ten years.[133]
Later it was found that morphine was more addictive than either alcohol or opium, and its extensive use during the American Civil War allegedly resulted in over 400,000[134] people with the "soldier's disease" of morphine addiction.[135] This idea has been a subject of controversy, as there have been suggestions that such a disease was in fact a fabrication; the first documented use of the phrase "soldier's disease" was in 1915.[136][137]
Diacetylmorphine (better known as heroin) was synthesized from morphine in 1874 and brought to market by Bayer in 1898. Heroin is approximately 1.5 to 2 times more potent than morphine weight for weight. Due to the lipid solubility of diacetylmorphine, it can cross the blood–brain barrier faster than morphine, subsequently increasing the reinforcing component of addiction.[138] Using a variety of subjective and objective measures, one study estimated the relative potency of heroin to morphine administered intravenously to post-addicts to be 1.80–2.66 mg of morphine sulfate to 1 mg of diamorphine hydrochloride (heroin).[42]


Morphine became a controlled substance in the US under the Harrison Narcotics Tax Act of 1914, and possession without a prescription in the US is a criminal offense. Morphine was the most commonly abused narcotic analgesic in the world until heroin was synthesized and came into use. In general, until the synthesis of dihydromorphine (c. 1900), the dihydromorphinone class of opioids (1920s), and oxycodone (1916) and similar drugs, there were no other drugs in the same efficacy range as opium, morphine, and heroin, with synthetics still several years away (pethidine was invented in Germany in 1937) and opioid agonists among the semi-synthetics were analogues and derivatives of codeine such as dihydrocodeine (Paracodin), ethylmorphine (Dionine), and benzylmorphine (Peronine). Even today, morphine is the most sought after prescription narcotic by heroin addicts when heroin is scarce, all other things being equal; local conditions and user preference may cause hydromorphone, oxymorphone, high-dose oxycodone, or methadone as well as dextromoramide in specific instances such as 1970s Australia, to top that particular list. The stop-gap drugs used by the largest absolute number of heroin addicts is probably codeine, with significant use also of dihydrocodeine, poppy straw derivatives like poppy pod and poppy seed tea, propoxyphene, and tramadol.
The structural formula of morphine was determined by 1925 by Robert Robinson.[140] At least three methods of total synthesis of morphine from starting materials such as coal tar and petroleum distillates have been patented, the first of which was announced in 1952, by Marshall D. Gates, Jr. at the University of Rochester.[141] Still, the vast majority of morphine is derived from the opium poppy by either the traditional method of gathering latex from the scored, unripe pods of the poppy, or processes using poppy straw, the dried pods and stems of the plant, the most widespread of which was invented in Hungary in 1925 and announced in 1930 by Hungarian pharmacologist János Kabay.[142]
In 2003, there was discovery of endogenous morphine occurring naturally in the human body. Thirty years of speculation were made on this subject because there was a receptor that, it appeared, reacted only to morphine: the μ3-opioid receptor in human tissue.[143] Human cells that form in reaction to cancerous neuroblastoma cells have been found to contain trace amounts of endogenous morphine.[91]
Society and culture
Legal status
- In Australia , morphine is classified as a Schedule 8 drug under the variously titled State and Territory Poisons Acts.
- In Canada , morphine is classified as a Schedule I drug under the Controlled Drugs and Substances Act.
- In France , morphine is in the strictest schedule of controlled substances, based upon the December 1970 French controlled substances law.
- In Germany , morphine is a verkehrsfähiges und verschreibungsfähiges Betäubungsmittel listed under Anlage III (the equivalent of CSA Schedule II) of the Betäubungsmittelgesetz.[144]
- In Switzerland , morphine is scheduled similarly to Germany's legal classification of the drug.
- In Japan , morphine is classified as a narcotic under the Narcotics and Psychotropics Control Act (麻薬及び向精神薬取締法, mayaku oyobi kōseishinyaku torishimarihō).
- In the Netherlands, morphine is classified as a List 1 drug under the Opium Law.
- In New Zealand, morphine is classified as a Class B drug under the Misuse of Drugs Act 1975.[145]
- In the United Kingdom , morphine is listed as a Class A drug under the Misuse of Drugs Act 1971 and a Schedule 2 Controlled Drug under the Misuse of Drugs Regulations 2001.
- In the United States , morphine is classified as a Schedule II controlled substance under the Controlled Substances Act under main Administrative Controlled Substances Code Number 9300. Morphine pharmaceuticals are subject to annual manufacturing quotas; in 2017 these quotas were 35.0 tonnes of production for sale, and 27.3 tonnes of production as an intermediate, or chemical precursor, for conversion into other drugs.[146] Morphine produced for use in extremely dilute formulations is excluded from the manufacturing quota.[citation needed]
- United Nations (UN), morphine is a Schedule I drug under the Single Convention on Narcotic Drugs.[147]
Non-medical use
The euphoria, comprehensive alleviation of distress and therefore all aspects of suffering, promotion of sociability and empathy, "body high", and anxiolysis provided by narcotic drugs including the opioids can cause the use of high doses in the absence of pain for a protracted period, which can impart a craving for the drug in the user.[148] As the prototype of the entire opioid class of drugs, morphine has properties that may lead to its misuse. Morphine addiction is the model upon which the current perception of addiction is based.
Animal and human studies and clinical experience back up the contention that morphine is one of the most euphoric drugs known, and via all but the IV route heroin and morphine cannot be distinguished according to studies because heroin is a prodrug for the delivery of systemic morphine. Chemical changes to the morphine molecule yield other euphorigenics such as dihydromorphine, hydromorphone (Dilaudid, Hydal), and oxymorphone (Numorphan, Opana), as well as the latter three's methylated equivalents dihydrocodeine, hydrocodone, and oxycodone, respectively; in addition to heroin, there are dipropanoylmorphine, diacetyldihydromorphine, and other members of the 3,6 morphine diester category like nicomorphine and other similar semi-synthetic opiates like desomorphine, hydromorphinol, etc. used clinically in many countries of the world but also produced illicitly in rare instances.
In general, non-medical use of morphine entails taking more than prescribed or outside of medical supervision, injecting oral formulations, mixing it with unapproved potentiators such as alcohol, cocaine, and the like, or defeating the extended-release mechanism by chewing the tablets or turning into a powder for snorting or preparing injectables. The latter method can be as time-consuming and involved as traditional methods of smoking opium. This and the fact that the liver destroys a large percentage of the drug on the first pass impacts the demand side of the equation for clandestine re-sellers, as many customers are not needle users and may have been disappointed with ingesting the drug orally. As morphine is generally as hard or harder to divert than oxycodone in a lot of cases, morphine in any form is uncommon on the street, although ampoules and phials of morphine injection, pure pharmaceutical morphine powder, and soluble multi-purpose tablets are very popular where available.
Morphine is also available in a paste that is used in the production of heroin, which can be smoked by itself or turned to a soluble salt and injected; the same goes for the penultimate products of the Kompot (Polish Heroin) and black tar processes. Poppy straw as well as opium can yield morphine of purity levels ranging from poppy tea to near-pharmaceutical-grade morphine by itself or with all of the more than 50 other alkaloids. It also is the active narcotic ingredient in opium and all of its forms, derivatives, and analogues as well as forming from breakdown of heroin and otherwise present in many batches of illicit heroin as the result of incomplete acetylation.
Names
Morphine is marketed under many different brand names in various parts of the world.[1] It was formerly called Morphia in British English.[149]
Informal names for morphine include: Cube Juice, Dope, Dreamer, Emsel, First Line, God's Drug, Hard Stuff, Hocus, Hows, Lydia, Lydic, M, Miss Emma, Mister Blue, Monkey, Morf, Morph, Morphide, Morphie, Morpho, Mother, MS, Ms. Emma, Mud, New Jack Swing (if mixed with heroin), Sister, Tab, Unkie, Unkie White, and Stuff.[150]
MS Contin tablets are known as misties, and the 100 mg extended-release tablets as greys and blockbusters. The "speedball" can use morphine as the opioid component, which is combined with cocaine, amphetamines, methylphenidate, or similar drugs. "Blue Velvet" is a combination of morphine with the antihistamine tripelennamine (Pyrabenzamine, PBZ, Pelamine) taken by injection.
Access in developing countries
Although morphine is cheap, people in poorer countries often do not have access to it. According to a 2005 estimate by the International Narcotics Control Board, six countries (Australia, Canada, France, Germany, the United Kingdom, and the United States) consume 79% of the world's morphine. The less affluent countries, accounting for 80% of the world's population, consumed only about 6% of the global morphine supply.[151] Some countriesLua error: Internal error: The interpreter has terminated with signal "24". import virtually no morphine, and in othersLua error: Internal error: The interpreter has terminated with signal "24". the drug is rarely available even for relieving severe pain while dying.[152]
Experts in pain management attribute the under-distribution of morphine to an unwarranted fear of the drug's potential for addiction and abuse. While morphine is clearly addictive, Western doctors believe it is worthwhile to use the drug and then wean the patient off when the treatment is over.[153]
References
- ↑ 1.0 1.1 1.2 drugs.com "International listings for Morphine". Drugs.com. https://www.drugs.com/international/morphine.html.
- ↑ 2.0 2.1 "Morphine Use During Pregnancy". 14 October 2019. https://www.drugs.com/pregnancy/morphine.html.
- ↑ Today's Medical Assistant: Clinical and Administrative Procedures. Elsevier Health Sciences. 2012. p. 571. ISBN 9781455701506. https://books.google.com/books?id=YalYPI1KqTQC&pg=PA571.
- ↑ "The bioavailability of rectally administered morphine". Pharmacology & Toxicology 62 (4): 203–5. April 1988. doi:10.1111/j.1600-0773.1988.tb01872.x. PMID 3387374.
- ↑ Cambridge textbook of accident and emergency medicine. Cambridge: Cambridge University Press. 1997. p. 191. ISBN 978-0-521-43379-2. https://books.google.com/books?id=m0bNaDhkaukC&pg=PA191.
- ↑ Oxford textbook of palliative care for children (2 ed.). Oxford: Oxford University Press. 2012. p. 240. ISBN 978-0-19-959510-5. https://books.google.com/books?id=k3XNeNseoHIC&pg=PA240.
- ↑ 7.00 7.01 7.02 7.03 7.04 7.05 7.06 7.07 7.08 7.09 7.10 7.11 7.12 7.13 "Morphine sulfate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/morphine-sulfate.html.
- ↑ 8.0 8.1 8.2 Rockwood and Wilkins' fractures in children (7th ed.). Philadelphia, Pa.: Lippincott Williams & Wilkins. 2009. p. 54. ISBN 978-1-58255-784-7. https://books.google.com/books?id=QVIdXV_F8M4C&pg=PA54.
- ↑ 9.0 9.1 9.2 9.3 9.4 9.5 9.6 "Endogenous morphine: up-to-date review 2011". Folia Biologica 58 (2): 49–56. 2012. PMID 22578954. http://fb.cuni.cz/file/5635/FB2012A0008.pdf. Retrieved 10 October 2016. "Positive evolutionary pressure has apparently preserved the ability to synthesize chemically authentic morphine, albeit in homeopathic concentrations, throughout animal phyla.".
- ↑ 10.0 10.1 10.2 Forces of habit drugs and the making of the modern world (1 ed.). Cambridge, Mass.: Harvard University Press. 2009. pp. 36–37. ISBN 978-0-674-02990-3. https://books.google.com/books?id=GHqV3elHYvMC&pg=PA36.
- ↑ 11.0 11.1 Molecular, clinical and environmental toxicology. Springer. 2009. p. 20. ISBN 978-3-7643-8335-0. https://books.google.com/books?id=MtOiLVWBn8cC&pg=PA20.
- ↑ 12.0 12.1 12.2 Drugs and Drug Policy: The Control of Consciousness Alteration. SAGE Publications. 2013. p. 123. ISBN 978-1-4833-2188-2. https://books.google.com/books?id=2UQXBAAAQBAJ&pg=PA123.
- ↑ Encyclopedia of substance abuse prevention, treatment, & recovery. Los Angeles: SAGE. 2009. p. 564. ISBN 978-1-4522-6601-5. https://books.google.com/books?id=DFR2AwAAQBAJ&pg=PT598.
- ↑ Narcotic Drugs Estimated World Requirements for 2008, Statistics for 2006. New York: United Nations Pubns. 2008. p. 77. ISBN 9789210481199. https://books.google.com/books?id=0_9QHvacPzYC&pg=PA77.
- ↑ 15.0 15.1 15.2 15.3 Narcotic Drugs 2014. INTERNATIONAL NARCOTICS CONTROL BOARD. 2015. pp. 21, 30. ISBN 9789210481571. https://www.incb.org/documents/Narcotic-Drugs/Technical-Publications/2014/Narcotic_Drugs_Report_2014.pdf.
- ↑ 16.0 16.1 Morphine. New York: Chelsea House Publishers. 2006. pp. 20–21. ISBN 978-1-4381-0211-5. https://books.google.com/books?id=sud4ORAMNkYC&pg=PA20.
- ↑ Drug abuse handbook (2nd ed.). Boca Raton: CRC/Taylor & Francis. 2006. pp. 7–8. ISBN 978-1-4200-0346-8. https://books.google.com/books?id=F0mUte90ATUC&pg=PA7.
- ↑ Black's Medical Dictionary. 87 (40th ed.). 2002. 162. doi:10.1038/087313b0. ISBN 978-0-7136-5442-4. Bibcode: 1911Natur..87R.313.. https://books.google.com/books?id=bUnCAwAAQBAJ&pg=PA162.
- ↑ Davis's Canadian Drug Guide for Nurses. F.A. Davis. 2014. p. 1409. ISBN 978-0-8036-4086-3. https://books.google.com/books?id=0Y8QBAAAQBAJ&pg=PA1409.
- ↑ World Health Organization model list of essential medicines: 22nd list (2021). Geneva: World Health Organization. 2021. WHO/MHP/HPS/EML/2021.02.
- ↑ "The Top 300 of 2020". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Morphine - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Morphine.
- ↑ "First Generic Drug Approvals 2023". 30 May 2023. https://www.fda.gov/drugs/drug-and-biologic-approval-and-ind-activity-reports/first-generic-drug-approvals.
- ↑ 24.0 24.1 "Morphine Sulfate". The American Society of Health-System Pharmacists. https://www.drugs.com/monograph/morphine-sulfate.html.
- ↑ "Association of intravenous morphine use and outcomes in acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative". American Heart Journal 149 (6): 1043–9. June 2005. doi:10.1016/j.ahj.2005.02.010. PMID 15976786.
- ↑ "BestBets: Does the application of opiates, during an attack of Acute Cardiogenic Pulmonary Oedma, reduce patients' mortality and morbidity?". Best Evidence Topics. http://www.bestbets.org/bets/bet.php?id=376.
- ↑ "Oral morphine for cancer pain". The Cochrane Database of Systematic Reviews 4 (3): CD003868. April 2016. doi:10.1002/14651858.CD003868.pub4. PMID 27105021.
- ↑ "Emergencies in palliative care". Cancer Journal 16 (5): 514–20. 2010. doi:10.1097/PPO.0b013e3181f28a8d. PMID 20890149.
- ↑ "Evidence-based review of interventions to improve palliation of pain, dyspnea, depression". Geriatrics 64 (8): 8–10, 12–4. August 2009. PMID 20722311.
- ↑ "An official American Thoracic Society statement: update on the mechanisms, assessment, and management of dyspnea". American Journal of Respiratory and Critical Care Medicine 185 (4): 435–52. February 2012. doi:10.1164/rccm.201111-2042ST. PMID 22336677.
- ↑ "American College of Chest Physicians consensus statement on the management of dyspnea in patients with advanced lung or heart disease". Chest 137 (3): 674–91. March 2010. doi:10.1378/chest.09-1543. PMID 20202949.
- ↑ (PDF) National Evaluation of Pharmacotherapies for Opioid Dependence (NEPOD): Report of Results and Recommendation. Monograph Series No. 52. Australian Government. 2004. ISBN 978-0-642-82459-2. http://www.health.gov.au/internet/drugstrategy/publishing.nsf/Content/8BA50209EE22B9C6CA2575B40013539D/$File/mono52.pdf+.
- ↑ "Narcotic analgesic effects on the sphincter of Oddi: a review of the data and therapeutic implications in treating pancreatitis". The American Journal of Gastroenterology 96 (4): 1266–72. April 2001. doi:10.1111/j.1572-0241.2001.03536.x. PMID 11316181.
- ↑ 34.0 34.1 34.2 34.3 34.4 34.5 "Opioids for chronic noncancer pain: a meta-analysis of effectiveness and side effects". CMAJ 174 (11): 1589–94. May 2006. doi:10.1503/cmaj.051528. PMID 16717269.
- ↑ "Morphine enhances nitric oxide release in the mammalian gastrointestinal tract via the micro(3) opiate receptor subtype: a hormonal role for endogenous morphine". Journal of Physiology and Pharmacology 55 (1 Pt 2): 279–88. March 2004. PMID 15082884.
- ↑ "Endogenous nitric oxide modulates morphine-induced constipation". Biochemical and Biophysical Research Communications 181 (2): 889–93. December 1991. doi:10.1016/0006-291X(91)91274-G. PMID 1755865.
- ↑ "The effect of opioid therapy on endocrine function". The American Journal of Medicine 126 (3 Suppl 1): S12-8. March 2013. doi:10.1016/j.amjmed.2012.12.001. PMID 23414717.
- ↑ "Opioid-induced endocrinopathy". The Journal of the American Osteopathic Association 109 (1): 20–5. January 2009. PMID 19193821.
- ↑ "Concentration-related effects of morphine on cognition and motor control in human subjects". Neuropsychopharmacology 5 (3): 157–66. November 1991. PMID 1755931.
- ↑ "Acute effects of opioids on memory functions of healthy men and women". Psychopharmacology 198 (2): 243–50. June 2008. doi:10.1007/s00213-008-1123-x. PMID 18379759.
- ↑ "Effects of opioids on driving ability". Journal of Pain and Symptom Management 19 (3): 200–8. March 2000. doi:10.1016/S0885-3924(99)00158-X. PMID 10760625.
- ↑ 42.0 42.1 42.2 "A comparative study of physiological and subjective effects of heroin and morphine administered intravenously in postaddicts". The Journal of Pharmacology and Experimental Therapeutics 133: 388–99. September 1961. PMID 13767429.
- ↑ 43.0 43.1 National Institute on Drug Abuse (NIDA) (April 2013). "Heroin". U.S. National Institutes of Health. http://www.nida.nih.gov/infofacts/heroin.html.
- ↑ "Tolerance to the anticonvulsant effect of morphine in mice: blockage by ultra-low dose naltrexone". Epilepsy Research 83 (2–3): 261–4. February 2009. doi:10.1016/j.eplepsyres.2008.10.011. PMID 19059761.
- ↑ "Role of receptor internalization in opioid tolerance and dependence". Pharmacology & Therapeutics 117 (2): 199–206. February 2008. doi:10.1016/j.pharmthera.2007.10.003. PMID 18076994.
- ↑ "Why do We Quit 'Cold Turkey'?". http://www.merriam-webster.com/words-at-play/why-do-we-quit-cold-turkey.
- ↑ "Opiate Withdrawal Stages". http://www.livestrong.com/article/68215-opiate-withdrawal-stages/.
- ↑ "Cardiovascular changes during morphine administration and spontaneous withdrawal in the rat". European Journal of Pharmacology 368 (1): 25–33. February 1999. doi:10.1016/S0014-2999(98)00984-4. PMID 10096766.
- ↑ "Morphine (and Heroin)". U.S. National Traffic Safety Administration. http://www.nhtsa.dot.gov/People/injury/research/job185drugs/morphine.htm.
- ↑ "Narcotics". U.S. Drug Enforcement Administration. http://www.justice.gov/dea/concern/narcotics.html.
- ↑ Romancing Opiates: Pharmacological Lies and the Addiction Bureaucracy. Encounter. 2006. pp. 160. ISBN 978-1-59403-087-1. https://archive.org/details/romancingopiates00theo/page/160.
- ↑ The Merck index : an encyclopedia of chemicals, drugs, and biological. Whitehouse Station, N.J.: Merck. 2006. ISBN 978-0-911910-00-1.
- ↑ 53.0 53.1 53.2 53.3 53.4 CRC handbook of chemistry and physics: a ready-reference book of chemical and physical data (85 ed.). Boca Ratan Florida: CRC Press. 2004. ISBN 978-0-8493-0485-9. https://archive.org/details/crchandbookofche81lide.
- ↑ "Morphine overdose". MedlinePlus. U.S. National Library of Medicine. 2 March 2009. https://www.nlm.nih.gov/medlineplus/ency/article/002502.htm.
- ↑ "Management of opioid analgesic overdose". The New England Journal of Medicine 367 (2): 146–155. July 2012. doi:10.1056/NEJMra1202561. PMID 22784117.
- ↑ "Selectivity of Ligands for Opioid Receptors". Opioids. Handbook of Experimental Pharmacology. 104 / 1. Berlin, Heidelberg: Springer. 1993. pp. 645–679. doi:10.1007/978-3-642-77460-7_26. ISBN 978-3-642-77462-1.
- ↑ 57.0 57.1 "Serotonin and norepinephrine uptake inhibiting activity of centrally acting analgesics: structural determinants and role in antinociception". The Journal of Pharmacology and Experimental Therapeutics 274 (3): 1263–70. September 1995. PMID 7562497.
- ↑ Pharmacology for Women's Health. Jones & Bartlett Publishers. 25 October 2010. pp. 332–. ISBN 978-1-4496-1073-9. https://books.google.com/books?id=o_rHHCsIpckC&pg=PA332.
- ↑ "Postoperative and chronic pain: systemic and regional pain techniques". Chestnut's Obstetric Anesthesia: Principles and Practice E-Book. Elsevier Health Sciences. 28 February 2014. pp. 611–. ISBN 978-0-323-11374-8. https://books.google.com/books?id=FMU0AwAAQBAJ&pg=PA611.
- ↑ Havard's Nursing Guide to Drugs. Elsevier Health Sciences. 1 June 2013. pp. 933–. ISBN 978-0-7295-8162-2. https://books.google.com/books?id=XpzQAgAAQBAJ&pg=PA933.
- ↑ "Chapter 15 – Opioid Agonists and Antagonists". Pharmacology and physiology for anesthesia : foundations and clinical application. Philadelphia, PA: Elsevier/Saunders. 2013. doi:10.1016/B978-1-4377-1679-5.00015-6. ISBN 978-1-4377-1679-5.
- ↑ "Standard opioid agonists activate heteromeric opioid receptors: evidence for morphine and [d-Ala(2)-MePhe(4)-Glyol(5)enkephalin as selective μ-δ agonists"]. ACS Chemical Neuroscience 1 (2): 146–54. February 2010. doi:10.1021/cn9000236. PMID 22816017.
- ↑ "Clinically employed opioid analgesics produce antinociception via μ-δ opioid receptor heteromers in Rhesus monkeys". ACS Chemical Neuroscience 3 (9): 720–7. September 2012. doi:10.1021/cn300049m. PMID 23019498.
- ↑ 64.0 64.1 "MS-Contin (Morphine Sulfate Controlled-Release) Drug Information: Clinical Pharmacology". RxList. http://www.rxlist.com/cgi/generic/ms_cp.htm.
- ↑ "Efficacy and ligand bias at the μ-opioid receptor". British Journal of Pharmacology 169 (7): 1430–46. August 2013. doi:10.1111/bph.12222. PMID 23646826.
- ↑ "Sigma antagonists potentiate opioid analgesia in rats". Neuroscience Letters 190 (2): 137–9. May 1995. doi:10.1016/0304-3940(95)11504-P. PMID 7644123.
- ↑ "The effects of NMDA receptor antagonists and nitric oxide synthase inhibitors on opioid tolerance and withdrawal. Medication development issues for opiate addiction". Neuropsychopharmacology 13 (4): 269–293. December 1995. doi:10.1016/0893-133X(95)00140-9. PMID 8747752.
- ↑ "Clinically available NMDA receptor antagonists memantine and dextromethorphan reverse existing tolerance to the antinociceptive effects of morphine in mice". Naunyn-Schmiedeberg's Archives of Pharmacology 361 (4): 425–432. April 2000. doi:10.1007/s002109900205. PMID 10763858.
- ↑ "Clinical efficacy of methadone in patients refractory to other mu-opioid receptor agonist analgesics for management of terminal cancer pain. Case presentations and discussion of incomplete cross-tolerance among opioid agonist analgesics". Cancer 72 (7): 2266–2272. October 1993. doi:10.1002/1097-0142(19931001)72:7<2266::AID-CNCR2820720734>3.0.CO;2-P. PMID 7690683.
- ↑ "Gene expression following acute morphine administration". Physiological Genomics 6 (3): 169–81. August 2001. doi:10.1152/physiolgenomics.2001.6.3.169. PMID 11526201.
- ↑ "Opioids and the immune system". Palliative Medicine 20 (Suppl 1): s9-15. 2006. doi:10.1191/0269216306pm1124oa. PMID 16764216.
- ↑ "Morphine reciprocally regulates IL-10 and IL-12 production by monocyte-derived human dendritic cells and enhances T cell activation". Molecular Medicine 12 (11–12): 284–90. 2006. doi:10.2119/2006-00043.Messmer. PMID 17380193.
- ↑ "Morphine reduces local cytokine expression and neutrophil infiltration after incision". Molecular Pain 3: 1744-8069-3-28. October 2007. doi:10.1186/1744-8069-3-28. PMID 17908329.
- ↑ "Opioid pharmacology". Pain Physician 11 (2 Suppl): S133-53. March 2008. doi:10.36076/ppj.2008/11/S133. PMID 18443637.
- ↑ "Morphine-6-glucuronide: actions and mechanisms". Medicinal Research Reviews 25 (5): 521–44. September 2005. doi:10.1002/med.20035. PMID 15952175.
- ↑ 76.0 76.1 "Morphine-6-glucuronide: morphine's successor for postoperative pain relief?". Anesthesia and Analgesia 102 (6): 1789–97. June 2006. doi:10.1213/01.ane.0000217197.96784.c3. PMID 16717327. http://www.anesthesia-analgesia.org/cgi/content/full/102/6/1789.
- ↑ 77.0 77.1 Jenkins AJ (2008) Pharmacokinetics of specific drugs. In Karch SB (Ed), Pharmacokinetics and pharmacodynamics of abused drugs. CRC Press: Boca Raton.
- ↑ 78.0 78.1 78.2 "Morphine, slow release (By mouth)". https://umm.edu/health/medical/drug-notes/notes/morphine-slow-release-by-mouth.
- ↑ "Opioids for chronic noncancer pain: still no evidence for superiority of sustained-release opioids". Clinical Pharmacology and Therapeutics 97 (2): 114–5. February 2015. doi:10.1002/cpt.26. PMID 25670511. Last reviewed on 18 November 2015
- ↑ 80.0 80.1 "Dosing & Uses". http://reference.medscape.com/drug/ms-contin-astramorph-morphine-343319.
- ↑ 81.0 81.1 "EndLink: An Internet-based End of Life Care Education Program – Morphine Dosing". http://endoflife.northwestern.edu/pain_management/morphine.pdf.
- ↑ Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. 2008. pp. 1057–1062. ISBN 978-0-9626523-7-0.
- ↑ "Detection time of drugs of abuse in urine". Acta Clinica Belgica 55 (6): 323–33. 2000. doi:10.1080/17843286.2000.11754319. PMID 11484423.
- ↑ "Detection times of drugs of abuse in blood, urine, and oral fluid". Therapeutic Drug Monitoring 26 (2): 200–5. April 2004. doi:10.1097/00007691-200404000-00020. PMID 15228165.
- ↑ Stereochemistry of organic compounds. New York: Wiley. 1994. ISBN 0-471-01670-5. OCLC 27642721. https://www.worldcat.org/oclc/27642721.
- ↑ "Morphine – Chiralpedia". 18 July 2022. https://chiralpedia.com/blog/morphine/.
- ↑ Opium Poppy: Botany, Chemistry, and Pharmacology. United States: CRC Press. 1995. pp. 164. ISBN 978-1-56024-923-8.
- ↑ "Thebaine content of selections of Papaver bracteatum Lindl. at different ages". Journal of Pharmaceutical Sciences 66 (12): 1716–9. December 1977. doi:10.1002/jps.2600661215. PMID 925935.
- ↑ Functional Neuroscience. New York: Springer. 2000. p. 116. ISBN 978-0-387-98543-5. https://books.google.com/books?id=nNH3p29wjK4C&q=endorphins+neurotransmitter&pg=PA116.
- ↑ "μ receptor". International Union of Basic and Clinical Pharmacology. 15 March 2017. http://www.guidetopharmacology.org/GRAC/ObjectDisplayForward?objectId=319. "Morphine occurs endogenously"
- ↑ 91.0 91.1 "Endogenous formation of morphine in human cells". Proceedings of the National Academy of Sciences of the United States of America 101 (39): 14091–6. September 2004. doi:10.1073/pnas.0405430101. PMID 15383669. Bibcode: 2004PNAS..10114091P. "Without doubt, human cells can produce the alkaloid morphine.".
- ↑ "The endogenous substrates of brain CYP2D". European Journal of Pharmacology 724: 211–8. February 2014. doi:10.1016/j.ejphar.2013.12.025. PMID 24374199. "Additionally, CYP2D is involved in the synthesis of endogenous morphine from various precursors, including L-3,4-dihydroxyphenylalanine (L-DOPA), reticulin, tetrahydropapaveroline (THP), and tyramine (Kulkarni, 2001; Mantione et al., 2008; Zhu, 2008).".
- ↑ "Morphine Synthesis and Biosynthesis-An Update". Current Organic Chemistry 4 (3): 343–362. March 2000. doi:10.2174/1385272003376292. http://brocku.ca/mathematics-science/departments-and-centres/chemistry/faculty/Hudlicky/CurrOrgChem-2000-4-343.pdf.
- ↑ "Home-brew heroin: soon anyone will be able to make illegal drugs". New Scientist. 18 May 2015. https://www.newscientist.com/article/dn27546-home-brew-heroin-soon-anyone-will-be-able-to-make-illegal-drugs/.
- ↑ "Final step in sugar-to-morphine conversion deciphered". Science. 25 June 2015. https://www.science.org/content/article/final-step-sugar-morphine-conversion-deciphered.
- ↑ "Complete biosynthesis of opioids in yeast". Science 349 (6252): 1095–100. September 2015. doi:10.1126/science.aac9373. PMID 26272907. Bibcode: 2015Sci...349.1095G.
- ↑ "Yeast-Based Opioid Production Completed". 13 August 2015. http://mobile.the-scientist.com/article/43739/yeast-based-opioid-production-completed.
- ↑ Triboluminescence: Theory, Synthesis, and Application (illustrated ed.). Springer. 2016. p. 193. ISBN 9783319388427. https://books.google.com/books?id=5G6zDAAAQBAJ. Extract of page 193
- ↑ "Narcotic analgesics: morphine and "peripherally modified" morphine analogs". Auburn University. Fall 2000. http://www.auburn.edu/~deruija/opioids_morphine.pdf.
- ↑ "The biological disposition of morphine and its surrogates". Bulletin of the World Health Organization 27 (3): 359–394. 1962. PMID 13999272.
- ↑ "UNODC - Bulletin on Narcotics - 1958 Issue 3 - 005". https://www.unodc.org/unodc/en/data-and-analysis/bulletin/bulletin_1958-01-01_3_page006.html.
- ↑ "Opioids", LiverTox: Clinical and Research Information on Drug-Induced Liver Injury (Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases), 2012, PMID 31643200, http://www.ncbi.nlm.nih.gov/books/NBK547864/, retrieved 2023-11-14
- ↑ Drug Stability for Pharmaceutical Scientists. Elsevier Science & Technology. 2013. pp. 82. OCLC 1136560730. http://worldcat.org/oclc/1136560730.
- ↑ Heroin (morphine diacetate) is a Schedule I controlled substance, so it is not used clinically in the United States;Lua error: Internal error: The interpreter has terminated with signal "24". it is a sanctioned medication in the United Kingdom and in Canada and some countries in Continental Europe, its use being particularly common (nearly to the degree of the hydrochloride salt)Lua error: Internal error: The interpreter has terminated with signal "24". in the United Kingdom.Lua error: Internal error: The interpreter has terminated with signal "24".
- ↑ Morphine valerate was one ingredient of a medication available for both oral and parenteral administration popular many years ago in Europe and elsewhere called Trivalin—not to be confused with the current, unrelated herbal preparation of the same name—which also included the valerates of caffeine and cocaine.Lua error: Internal error: The interpreter has terminated with signal "24". A version containing codeine valerate as a fourth ingredient is distributed under the name Tetravalin.Lua error: Internal error: The interpreter has terminated with signal "24".
- ↑ "The Synthesis of Morphine". Journal of the American Chemical Society 78 (7): 1380–1393. April 1956. doi:10.1021/ja01588a033.
- ↑ "Synthetic opium alkaloids and derivatives. A short total synthesis of (±)-dihydrothebainone, (±)-dihydrocodeinone, and (±)-nordihydrocodeinone as an approach to a practical synthesis of morphine, codeine, and congeners". The Journal of Organic Chemistry 45 (15): 3135–3137. July 1980. doi:10.1021/jo01303a045.
- ↑ "Studies directed towards the total synthesis of morphine alkaloids". Tetrahedron Letters 23 (3): 285–288. January 1982. doi:10.1016/S0040-4039(00)86810-0.
- ↑ "Studies culminating in the total synthesis of (dl)-morphine". The Journal of Organic Chemistry 53 (20): 4694–4708. September 1988. doi:10.1021/jo00255a008.
- ↑ "Convergent synthesis of (±)-dihydroisocodeine in 11 steps by the tandem radical cyclization strategy. A formal total synthesis of (±)-morphine". Journal of the American Chemical Society 114 (24): 9688–9689. November 1992. doi:10.1021/ja00050a075.
- ↑ "Asymmetric synthesis of either enantiomer of opium alkaloids and morphinans. Total synthesis of (−)- and (+)-dihydrocodeinone and (−)- and (+)-morphine". Journal of the American Chemical Society 115 (23): 11028–11029. November 1993. doi:10.1021/ja00076a086.
- ↑ "Formal Total Synthesis of(—)-Morphine by Cuprate Conjugate Addition". Angewandte Chemie International Edition in English 35 (2324): 2830–2832. December 1996. doi:10.1002/anie.199628301.
- ↑ "Asymmetric Total Synthesis of (+)-Codeine via Intramolecular Carbenoid Insertion". The Journal of Organic Chemistry 64 (21): 7871–7884. October 1999. doi:10.1021/jo990905z.
- ↑ "Synthesis of (-)-morphine". Journal of the American Chemical Society 124 (42): 12416–7. October 2002. doi:10.1021/ja027882h. PMID 12381175.
- ↑ "Enantioselective synthesis of (-)-codeine and (-)-morphine". Journal of the American Chemical Society 124 (49): 14542–3. December 2002. doi:10.1021/ja0283394. PMID 12465957. https://figshare.com/articles/Enantioselective_Synthesis_of_-Codeine_and_-Morphine/3646293.
- ↑ "Total synthesis of (+/-)-morphine". Organic Letters 8 (23): 5311–3. November 2006. doi:10.1021/ol062112m. PMID 17078705.
- ↑ "Diastereoselective total synthesis of (+/-)-codeine". Chemistry: A European Journal 14 (22): 6606–8. 2008. doi:10.1002/chem.200800744. PMID 18561354.
- ↑ "Regiospecific and stereoselective syntheses of (+/-) morphine, codeine, and thebaine via a highly stereocontrolled intramolecular 4 + 2 cycloaddition leading to a phenanthrofuran system". Journal of the American Chemical Society 131 (32): 11402–6. August 2009. doi:10.1021/ja9038505. PMID 19624126. https://figshare.com/articles/Regiospecific_and_Stereoselective_Syntheses_of_Morphine_Codeine_and_Thebaine_via_a_Highly_Stereocontrolled_Intramolecular_4_2_Cycloaddition_Leading_to_a_Phenanthrofuran_System/2834536.
- ↑ "The Top Pharmaceuticals That Changed The World-Morphine". Chemical and Engineering News. 20 June 2005. http://pubs.acs.org/cen/coverstory/83/8325/8325morphine.html.
- ↑ "Genetically modified E. coli pump out morphine precursor: Bacteria yield 300 times more opiates than yeast" (in en). https://www.sciencedaily.com/releases/2016/02/160225101103.htm.
- ↑ "Narcotics (Opioids) | DEA". https://www.dea.gov/taxonomy/term/331.
- ↑ "Recovery of morphine from a controlled-release preparation. A source of opioid abuse". Cancer 66 (12): 2642–4. December 1990. doi:10.1002/1097-0142(19901215)66:12<2642::AID-CNCR2820661229>3.0.CO;2-B. PMID 2249204.
- ↑ "[History of opium poppy and morphine"]. Dansk Medicinhistorisk Arbog 33: 171–184. 2005. PMID 17152761. https://pubmed.ncbi.nlm.nih.gov/17152761.
- ↑ "Pain relief and sedation in Roman Byzantine texts: Mandragoras officinarum, Hyoscyamos niger and Atropa belladonna". International Congress Series 1242: 43–50. December 2002. doi:10.1016/S0531-5131(02)00699-4.
- ↑ "Laudanum in the Works of Paracelsus". Bull. Hist. Med. 9: 530–544. 1941. http://www.samorini.it/doc1/alt_aut/sz/sigerist-laudanum-in-the-work-of-paracelsus.pdf. Retrieved 5 September 2018.
- ↑ "Friedrich Sertürner (Untitled letter to the editor)" (in de). Journal der Pharmacie für Aerzte, Apotheker und Chemisten 13: 229–243; see especially "III. Säure im Opium" (acid in opium), pp. 234–235, and "I. Nachtrag zur Charakteristik der Säure im Opium" (Addendum on the characteristics of the acid in opium), pp. 236–241. 1805. https://books.google.com/books?id=8A09AAAAcAAJ&pg=PA229.
- ↑ 127.0 127.1 "Incidence, Reversal, and Prevention of Opioid-induced Respiratory Depression". Anesthesiology 112 (1): 226–38. January 2010. doi:10.1097/ALN.0b013e3181c38c25. PMID 20010421.
- ↑ Sertürner coined the term morphium in: Sertuerner (1817) "Ueber das Morphium, eine neue salzfähige Grundlage, und die Mekonsäure, als Hauptbestandtheile des Opiums" (On morphine, a new salifiable [i.e., precipitable], fundamental substance, and meconic acid, as principal components of opium), Annalen der Physik, 55 : 56–89. It was Gay-Lussac, a French chemist and editor of Annales de Chimie et de Physique, who coined the word morphine in a French translation of Sertuener's original German article: Sertuener (1817) "Analyse de l'opium: De la morphine et de l'acide méconique, considérés comme parties essentielles de l'opium" (Analysis of opium: On morphine and on meconic acid, considered as essential constituents of opium), Annales de Chimie et de Physique, 2nd series, 5 : 21–42. From p. 22: " ... car il a pris pour cette substance, que j'appelle morphine (morphium), ce qui n'en était qu'une combinaison avec l'acide de l'opium." ( ... for he [i.e., French chemist and pharmacist Charles Derosne (1780–1846)] took as that substance [i.e., the active ingredient in opium], which I call "morphine" (or morphium), what was only a compound of it with acid of opium.)
- ↑ "God's Own Medicine". Skeptical Inquirer 41 (2): 44. March–April 2017.
- ↑ Annual Register. J. Dodsley. 1824. p. 1. https://archive.org/details/annualregister03dodsgoog. Retrieved 1 September 2015. "Edme."
- ↑ Drug Hunters.. Arcade Publishing. 2016. ISBN 978-1-62872-719-7. OCLC 966360188. https://www.worldcat.org/oclc/966360188.
- ↑ Cocaine global histories. London: Routledge. 1999. pp. 90. ISBN 92-9078-018-5. OCLC 1162209949. https://www.worldcat.org/oclc/1162209949.
- ↑ The Pursuit of Oblivion: A Global History of Narcotics. W.W. Norton. 2003. p. 68. ISBN 978-0-393-32545-4.
- ↑ "Lewis H. Wright Memorial Lecture". ASA Newsletter 68 (7): 9–10. July 2004. http://www.asahq.org/For-Members/Publications-and-Research/Periodicals/ASA-Newsletter/July-2004-ASA-Newsletter.aspx.
- ↑ "Opiate Narcotics". Canadian Government Commission. http://www.druglibrary.org/schaffer/library/studies/ledain/nonmed4.htm.
- ↑ "Mythical Roots of US Drug Policy – Soldier's Disease and Addicts in the Civil War". http://www.druglibrary.org/schaffer/history/soldis.htm.
- ↑ "Soldiers Disease A Historical Hoax?". iPromote Media Inc. 2006. http://www.amusingfacts.com/facts/Detail/soldiers-disease-morphine.html.
- ↑ "Relative reinforcing strength of three N-methyl-D-aspartate antagonists with different onsets of action". The Journal of Pharmacology and Experimental Therapeutics 301 (2): 690–7. May 2002. doi:10.1124/jpet.301.2.690. PMID 11961074.
- ↑ "Morphine Easy Home Cure". Overland Monthly 35 (205): 14. 1900. http://quod.lib.umich.edu/m/moajrnl/ahj1472.2-35.205/22:6?page=root;rgn=full+text;size=100;view=image.
- ↑ "Constitution of codeine and thebaine". Memoirs and Proceedings of the Literary and Philosophical Society of Manchester 69: 79–86. 1925.
- ↑ "Marshall D. Gates, Chemist to First Synthesize Morphine, Dies". University of Rochester. 3 October 2003. http://www.rochester.edu/news/show.php?id=20.
- ↑ "[János Kabay and the poppy straw process. Commemoration on the 50th anniversary of his death]". Acta Pharmaceutica Hungarica 57 (3–4): 105–10. July 1987. PMID 3314338.
- ↑ "Human white blood cells synthesize morphine: CYP2D6 modulation". Journal of Immunology 175 (11): 7357–62. December 2005. doi:10.4049/jimmunol.175.11.7357. PMID 16301642.
- ↑ "Anlage III (zu § 1 Abs. 1) verkehrsfähige und verschreibungsfähige Betäubungsmittel" (in de). Bundesamt für Justiz (Federal Office of Justice). http://www.gesetze-im-internet.de/btmg_1981/anlage_iii_61.html.
- ↑ "Misuse of Drugs Act 1975, sch 2". 13 January 2022. https://www.legislation.govt.nz/act/public/1975/0116/latest/DLM436586.html.
- ↑ 82 FR 51293
- ↑ "List of narcotic drugs under international control". Yellow List: 5. March 2011. http://www.incb.org/documents/Narcotic-Drugs/Yellow_List/NAR_2011_YellowList_50edition_EN.pdf.
- ↑ "Chapter 22 - The opioid epidemic", Global Emergency of Mental Disorders (Academic Press): pp. 401–418, 2021-01-01, doi:10.1016/B978-0-323-85837-3.00019-4, ISBN 978-0-323-85837-3, https://www.sciencedirect.com/science/article/pii/B9780323858373000194, retrieved 2024-01-11
- ↑ "Morphia | Definition of Morphia by Lexico". https://www.lexico.com/en/definition/morphia.
- ↑ The Encyclopedia of Addictive Drugs. Greenwood Publishing Group. 1 January 2002. p. 306. ISBN 978-0-313-31807-8. https://books.google.com/books?id=G7As-qawdzMC&q=happy+powder&pg=PA95.
- ↑ Scholten (3rd ed.). Switzerland: World Health Organization. pp. 1–22. http://apps.who.int/medicinedocs/en/d/Js18062en/. Retrieved 17 January 2018.
- ↑ Human Rights Watch (2 June 2011). "Global State of Pain Treatment; Access to Palliative Care as a Human Right". Human Rights Watch. https://www.hrw.org/report/2011/06/02/global-state-pain-treatment/access-medicines-and-palliative-care#_ftn22.
- ↑ "Drugs Banned, Many of World's Poor Suffer in Pain". New York Times. 10 September 2007. https://www.nytimes.com/2007/09/10/health/10pain.html.
Lua error: Internal error: The interpreter has terminated with signal "24".
External links
Lua error: Internal error: The interpreter has terminated with signal "24".
- Morphine and Heroin at The Periodic Table of Videos (University of Nottingham)
- Video: Intravenous morphine loading (Vimeo) (YouTube) – A short education video teaching health professionals the main points about intravenous loading of analgesics, in particular morphine.
Lua error: Internal error: The interpreter has terminated with signal "24". Lua error: Internal error: The interpreter has terminated with signal "24". Lua error: Internal error: The interpreter has terminated with signal "24". Lua error: Internal error: The interpreter has terminated with signal "24". Lua error: Internal error: The interpreter has terminated with signal "24".
Lua error: Internal error: The interpreter has terminated with signal "24".

