Chemistry:Nicotine
Nicotine is an alkaloid found primarily in plants of the nightshade family, notably in tobacco; it is also synthesized.[1] Nicotine is used recreationally for its stimulant and anxiolytic effects. In tobacco leaves, nicotine constitutes about 0.6–3.0% of the dry weight,[2] and smaller, trace quantities occur in other Solanaceae crops such as tomatoes, potatoes, and eggplants. In pure form, nicotine is a colorless to yellowish, oily liquid that readily penetrates biological membranes and acts as a potent neurotoxin in insects, where it serves as a antiherbivore toxin. Historically, it was widely used as an insecticide, and its structure provided the basis for synthetic neonicotinoid pesticides.[3]
In humans, nicotine acts primarily as a stimulant by binding to and activating nicotinic acetylcholine receptors (nAChRs) in the central nervous system and peripheral tissues. This results in the release of neurotransmitters such as dopamine, acetylcholine, and norepinephrine, producing effects including increased alertness, reduced anxiety, and mild euphoria.[4] Nicotine is typically consumed through tobacco smoking, vaping, or other nicotine delivery systems. An average cigarette outside of the European Union yields about 2 mg of absorbed nicotine, a dose sufficient to produce reinforcement and dependence while remaining far below toxic levels,[5] but in the EU it is maximum 1 mg absorbed.[6][7]
Nicotine is highly addictive, and nicotine dependence is characterized by tolerance, physical dependence, psychological dependence, and nicotine withdrawal symptoms such as irritability, anxiety, and difficulty concentrating.[8][9] Nicotine replacement therapy (NRT) products, including gums, patches, and lozenges, deliver the compound in slower, lower doses that are less addictive and are used medically to help people quit smoking.[10][11] Synthetic derivatives of nicotine, such as varenicline, act as partial agonists at nicotinic receptors and are also used as smoking cessation aids.[12]
Although nicotine itself is not classified as a carcinogen by either the International Agency for Research on Cancer or the Surgeon General of the United States,[13] high doses can cause nicotine poisoning and respiratory paralysis. Chronic exposure may also affect the immune, cardiovascular, and nervous systems.[14][15] Nicotine is also a known teratogen, associated with adverse developmental effects during pregnancy,[16] and may impair adolescent neurodevelopment, though the extent of this effect in humans remains debated.[17]
Uses
Medical
The primary therapeutic use of nicotine is treating nicotine dependence to eliminate smoking and the damage it does to health. Nicotine itself is not a standalone cessation tool; its efficacy in smoking cessation relies on nicotine replacement therapy (NRT) delivery systems, which vary formulations (e.g., transdermal patches and lozenges for steady release versus oral gum, inhalers, and nasal sprays for acute relief) to control and modify how much nicotine is delivered and absorbed, and to mimic tobacco pharmacokinetics without harmful byproducts.[18]
A 2018 Cochrane Collaboration review found high-quality evidence that all current forms of nicotine replacement therapy (gum, patch, lozenges, inhaler, and nasal spray) increase the chances of successfully quitting smoking by 50–60%, regardless of setting.[18]
Combining nicotine patch use with a faster acting nicotine replacement, like gum or spray, improves the odds of treatment success.[19]
In contrast to recreational nicotine products, which have been designed to maximize the likelihood of addiction, nicotine replacement products (NRTs) are designed to minimize addictiveness.[13]: 112 The more quickly a dose of nicotine is delivered and absorbed, the higher the addiction risk.[20]
Pesticide
Nicotine has been used as an insecticide since at least 1690, in the form of tobacco extracts or as pure nicotine sulfate[3][21][22] (although other components of tobacco also seem to have pesticide effects).[23] It acts on the nicotinic acetylcholine receptor, and gave the receptor its name. Nicotine is in IRAC group 4B. Nicotine insecticides have been banned in the US since 2014,[24] including use on organic crops,[25] and caution is recommended for small gardeners.[26] Nicotine pesticides have been banned in the EU since 2009.[27] Foods are imported from countries in which nicotine pesticides are allowed, such as China, but foods may not exceed maximum nicotine levels.[27][28] Neonicotinoids, such as imidacloprid, which are derived from and structurally similar to nicotine, are widely used as agricultural and veterinary pesticides as of 2016.[29][21]
Performance
Nicotine-containing products are sometimes used for the performance-enhancing effects of nicotine on cognition.[30] A 2010 meta-analysis of 41 double-blind, placebo-controlled studies concluded that nicotine or smoking had significant positive effects on aspects of fine motor abilities, alerting and orienting attention, and episodic and working memory.[31] A 2015 review noted that stimulation of the α4β2 nicotinic receptor is responsible for certain improvements in attentional performance;[32] among the nicotinic receptor subtypes, nicotine has the highest binding affinity at the α4β2 receptor (ki=1 nM), which is also the biological target that mediates nicotine's addictive properties.[33] Nicotine has potential beneficial effects, but it also has paradoxical effects, which may be due to the inverted U-shape of the dose-response curve or pharmacokinetic features.[34]
Recreational
Nicotine is used as a recreational drug.[35] It is widely used, highly addictive and hard to discontinue.[36] Nicotine is often used compulsively,[37] and dependence can develop within days.[37][38] Recreational drug users commonly use nicotine for its mood-altering effects.[20] Recreational nicotine products include chewing tobacco, cigars,[39] cigarettes,[39] e-cigarettes,[40] snuff, pipe tobacco,[39] snus, and nicotine pouches.[41]
Alcohol infused with nicotine is called nicotini.[42]
Contraindications
Nicotine use for tobacco cessation has few contraindications.[43]
It is not known whether nicotine replacement therapy is effective for smoking cessation in adolescents, as of 2014.[44] It is therefore not recommended to adolescents.[45] It is not safe to use nicotine during pregnancy or breastfeeding, although it is safer than smoking. The desirability of NRT use in pregnancy is therefore debated.[46][47][48]
Randomized trials and observational studies of nicotine replacement therapy in cardiovascular patients show no increase in adverse cardiovascular events compared to those treated with placebo.[49] Using nicotine products during cancer treatment may be contraindicated, as nicotine may promote tumour growth, but temporary use of NRTs to quit smoking may be advised for harm reduction.[50]
Nicotine gum is contraindicated in individuals with temporomandibular joint disease.[51] People with chronic nasal disorders and severe reactive airway disease require additional precautions when using nicotine nasal sprays.[45] Nicotine in any form is contraindicated in individuals with a known hypersensitivity to nicotine.[51][45]
Adverse effects
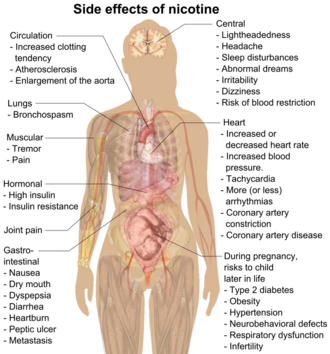
Nicotine is classified as a poison,[53][54] and it is "extremely hazardous".[55] The CDC says it is "toxic to developing fetuses and is a health danger for pregnant women." It can harm brain development up to age twenty-five, and early use of nicotine can predispose young people to smoking and drug use.[56] However, at doses typically used by consumers, it presents little if any hazard to adult users.[57][58][59] Although at low amounts nicotine has a mild analgesic effect,[60] at sufficiently high doses nicotine may result in nausea, vomiting, diarrhea, salivation, bradycardia, and possibly seizures, hypoventilation, and death.[61]
Sleep
Nicotine reduces the amount of rapid eye movement (REM) sleep, slow-wave sleep (SWS), and total sleep time in healthy nonsmokers given nicotine via a transdermal patch, and the reduction is dose-dependent.[62] Acute nicotine intoxication has been found to significantly reduce total sleep time and increase REM latency, sleep onset latency, and non-rapid eye movement (NREM) stage 2 sleep time.[62][63] Depressive non-smokers experience mood and sleep improvements under nicotine administration; however, subsequent nicotine withdrawal has a negative effect on both mood and sleep.[64]
Cardiovascular system
Nicotine exerts several significant effects on the cardiovascular system. Primarily, it stimulates the sympathetic nervous system, leading to the release of catecholamines. This activation results in an increase in heart rate and blood pressure, as well as enhanced myocardial contractility, which raises the workload on the heart. Additionally, nicotine causes systemic vasoconstriction, including constriction of coronary arteries, which can reduce blood flow to the heart. Long-term exposure to nicotine may impair endothelial function, potentially contributing to atherosclerosis. Furthermore, nicotine has been associated with the development of cardiac arrhythmias, particularly in individuals who already have underlying heart disease.[65]
The effects of nicotine can be differentiated between short-term and long-term use. Short-term nicotine use, such as that associated with nicotine replacement therapy (NRT) for smoking cessation, appears to pose little cardiovascular risk, even for patients with known cardiovascular conditions. In contrast, longer-term nicotine use may not accelerate atherosclerosis but could contribute to acute cardiovascular events in those with pre-existing cardiovascular disease. Many severe cardiovascular effects traditionally associated with smoking may not be solely attributable to nicotine itself. Cigarette smoke contains numerous other potentially cardiotoxic substances, including carbon monoxide and oxidant gases.[65]
A 2016 review of the cardiovascular toxicity of nicotine concluded, "Based on current knowledge, we believe that the cardiovascular risks of nicotine from e-cigarette use in people without cardiovascular disease are quite low. We have concerns that nicotine from e-cigarettes could pose some risk for users with cardiovascular disease."[65]
A 2018 Cochrane review found that, in rare cases, nicotine replacement therapy can cause non-ischemic chest pain (i.e., chest pain that is unrelated to a heart attack) and heart palpitations, but does not increase the incidence of serious cardiac adverse events (i.e., myocardial infarction, stroke, and cardiac death) relative to controls.[18]
Blood pressure
In the short term, nicotine causes a transient increase in blood pressure. Long term, epidemiological studies generally show increased blood pressure and hypertension among nicotine users.[65]
Reinforcement disorders
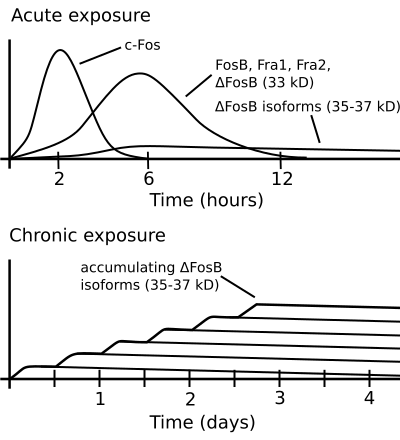
ΔFosB accumulation from excessive drug use
Top: this depicts the initial effects of high dose exposure to an addictive drug on gene expression in the nucleus accumbens for various Fos family proteins (i.e., c-Fos, FosB, ΔFosB, Fra1, and Fra2).
Bottom: this illustrates the progressive increase in ΔFosB expression in the nucleus accumbens following repeated twice daily drug binges, where these phosphorylated (35–37 kilodalton) ΔFosB isoforms persist in the D1-type medium spiny neurons of the nucleus accumbens for up to 2 months.[66][67] |
Nicotine is highly addictive but paradoxically has quite weak reinforcing property compared to other drugs of abuse in various animals.[68][36][69][70] Its addictiveness depends on how it is administered and also depends upon form in which nicotine is used.[71] Animal research suggests that monoamine oxidase inhibitors, acetaldehyde[70][72] and other constituents in tobacco smoke may enhance its addictiveness.[73][74] Nicotine dependence involves aspects of both psychological dependence and physical dependence, since discontinuation of extended use has been shown to produce both affective (e.g., anxiety, irritability, craving, anhedonia) and somatic (mild motor dysfunctions such as tremor) withdrawal symptoms.[75] Withdrawal symptoms peak in one to three days[76] and can persist for several weeks.[77] Even though other drugs of dependence can have withdrawal states lasting 6 months or longer, this does not appear to occur with cigarette withdrawal.[78]
Normal between-cigarettes discontinuation, in unrestricted smokers, causes mild but measurable nicotine withdrawal symptoms.[79] These include mildly worse mood, stress, anxiety, cognition, and sleep, all of which briefly return to normal with the next cigarette.[79] Smokers have a worse mood than they typically would have if they were not nicotine-dependent; they experience normal moods only immediately after smoking.[79] Nicotine dependence is associated with poor sleep quality and shorter sleep duration among smokers.[80][81]
In dependent smokers, withdrawal causes impairments in memory and attention, and smoking during withdrawal returns these cognitive abilities to pre-withdrawal levels.[82] The temporarily increased cognitive levels of smokers after inhaling smoke are offset by periods of cognitive decline during nicotine withdrawal.[79] Therefore, the overall daily cognitive levels of smokers and non-smokers are roughly similar.[79]
Nicotine activates the mesolimbic pathway and induces long-term ΔFosB expression (i.e., produces phosphorylated ΔFosB isoforms) in the nucleus accumbens when inhaled or injected frequently or at high doses, but not necessarily when ingested.[83][84][85] Consequently, high daily exposure (possibly excluding oral route) to nicotine can cause ΔFosB overexpression in the nucleus accumbens, resulting in nicotine addiction.[83][84]
Cancer
Contrary to popular belief, nicotine itself does not cause cancer in humans,[86][87] although it is unclear whether it functions as a tumor promoter as of 2012[update].[88] A 2018 report by the US National Academies of Sciences, Engineering, and Medicine concludes, "Template:Wj[w]hile it is biologically plausible that nicotine can act as a tumor promoter, the existing body of evidence indicates this is unlikely to translate into increased risk of human cancer."[89]
Although nicotine is classified as a non-carcinogenic substance, it can still theoretically promote tumor growth and metastasis as evidenced from alterations. Nicotine induces several processes, some of them via nicotine's effects on immune function, that contribute to cancer progression in both smoking-related and non-smoking-related cancers, including cell cycle progression, epithelial-to-mesenchymal transition, migration, invasion, angiogenesis, and evasion of apoptosis.[90][15] These effects are primarily mediated through nicotinic acetylcholine receptors (nAChRs), particularly the α7 subtype, and to a lesser extent, β-adrenergic receptors (β-ARs). Activation of these receptors triggers several signaling cascades crucial in cancer biology, notably the MAPK/ERK pathway, PI3K/AKT pathway, and JAK-STAT signaling.[90]
Nicotine potentially promotes lung cancer development by enhancing proliferation, angiogenesis, migration, invasion, and epithelial–mesenchymal transition (EMT) via nAChRs, which are present in lung cancer cells.[91] Additionally, nicotine-induced EMT contributes to drug resistance in cancer cells.[92]
Nicotine in tobacco can form carcinogenic tobacco-specific nitrosamines through a nitrosation reaction. This occurs mostly in the curing and processing of tobacco. However, nicotine in the mouth and stomach can react to form N-nitrosonornicotine,[93] a known type 1 carcinogen,[94] suggesting that consumption of non-tobacco forms of nicotine may still play a role in carcinogenesis.[95]
Genotoxicity
Nicotine causes DNA damage in several types of human cells as judged by assays for genotoxicity such as the comet assay, cytokinesis-block micronucleus test and chromosome aberrations test. In humans, this damage can happen in primary parotid gland cells,[96] lymphocytes,[97] and respiratory tract cells.[98]
Pregnancy and breastfeeding
Nicotine has been shown to produce birth defects in some animal species, but not others;[99] consequently, it is considered to be a possible teratogen in humans.[99] In animal studies that resulted in birth defects, researchers found that nicotine negatively affects fetal brain development and pregnancy outcomes;[99][13] the negative effects on early brain development are associated with abnormalities in brain metabolism and neurotransmitter system function.[100] Nicotine crosses the placenta and is found in the breast milk of mothers who smoke as well as mothers who inhale passive smoke.[101]
Nicotine exposure in utero is responsible for several complications of pregnancy and birth: pregnant women who smoke are at greater risk for both miscarriage and stillbirth and infants exposed to nicotine in utero tend to have lower birth weights.[102] A McMaster University research group observed in 2010 that rats exposed to nicotine in the womb (via parenteral infusion) later in life had conditions including type 2 diabetes, obesity, hypertension, neurobehavioral defects, respiratory dysfunction, and infertility.[103]
Overdose
It is unlikely that a person would overdose on nicotine through smoking alone. The US Food and Drug Administration (FDA) stated in 2013 that there are no significant safety concerns associated with the use of more than one form of over-the-counter (OTC) nicotine replacement therapy at the same time, or using OTC NRT at the same time as another nicotine-containing product, like cigarettes.[104] The median lethal dose of nicotine in humans is unknown.[105][5] Nevertheless, nicotine has a relatively high toxicity in comparison to many other alkaloids such as caffeine, which has an LD50 of 127 mg/kg when administered to mice.[106] At sufficiently high doses, it is associated with nicotine poisoning,[13] which, while common in children (in whom poisonous and lethal levels occur at lower doses per kilogram of body weight[60]) rarely results in significant morbidity or death.[99] The estimated lower dose limit for fatal outcomes is 500–1,000 mg of ingested nicotine for an adult (6.5–13 mg/kg).[73][5]
The initial symptoms of a nicotine overdose typically include nausea, vomiting, diarrhea, hypersalivation, abdominal pain, tachycardia (rapid heart rate), hypertension (high blood pressure), tachypnea (rapid breathing), headache, dizziness, pallor (pale skin), auditory or visual disturbances, and perspiration, followed shortly after by marked bradycardia (slow heart rate), bradypnea (slow breathing), and hypotension (low blood pressure).[99] An increased respiratory rate (i.e., tachypnea) is one of the primary signs of nicotine poisoning.[99] At sufficiently high doses, somnolence (sleepiness or drowsiness), confusion, syncope (loss of consciousness from fainting), shortness of breath, marked weakness, seizures, and coma may occur.[107][99] Lethal nicotine poisoning rapidly produces seizures, and death – which may occur within minutes – is believed to be due to respiratory paralysis.[99]
Toxicity
Today nicotine is less commonly used in agricultural insecticides, which was a main source of poisoning. More recent cases of poisoning typically appear to be in the form of Green Tobacco Sickness (GTS),[99] accidental ingestion of tobacco or tobacco products, or ingestion of nicotine-containing plants.[108][109][110] People who harvest or cultivate tobacco may experience GTS, a type of nicotine poisoning caused by dermal exposure to wet tobacco leaves. This occurs most commonly in young, inexperienced tobacco harvesters who do not consume tobacco.[108][111] People can be exposed to nicotine in the workplace by breathing it in, skin absorption, swallowing it, or eye contact. The Occupational Safety and Health Administration (OSHA) has set the legal limit (permissible exposure limit) for nicotine exposure in the workplace as 0.5 mg/m3 skin exposure over an 8-hour workday. The US National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) of 0.5 mg/m3 skin exposure over an 8-hour workday. At environmental levels of 5 mg/m3, nicotine is immediately dangerous to life and health.[112]
Drug interactions
Pharmacodynamic
- Potential interaction with sympathomimetic drugs (adrenergic agonists) and sympatholytic drugs (alpha-blockers and beta-blockers).[51]
Pharmacokinetic
Nicotine and cigarette smoke both induce the expression of liver enzymes (e.g., certain cytochrome P450 proteins) which metabolize drugs, leading to the potential for alterations in drug metabolism.[51]
- Smoking cessation may decrease the metabolism of acetaminophen, beta-blockers, caffeine, oxazepam, pentazocine, propoxyphene, theophylline, and tricyclic antidepressants, leading to higher plasma concentrations of these drugs.[51]
- Possible alteration of nicotine absorption through the skin from the transdermal nicotine patch by drugs that cause vasodilation or vasoconstriction.[51]
- Possible alteration of nicotine absorption through the nasal cavity from the nicotine nasal spray by nasal vasoconstrictors (e.g., xylometazoline).[51]
- Possible alteration of nicotine absorption through oral mucosa from nicotine gum and lozenges by food and drink that modify salivary pH.[51]
Pharmacology
Pharmacodynamics
Nicotine acts as a receptor agonist at most nicotinic acetylcholine receptors (nAChRs),[113][114] except at two nicotinic receptor subunits (nAChRα9 and nAChRα10) where it acts as a receptor antagonist.[115][113] Such antagonism results in mild analgesia.
The stereochemistry of nicotine is crucial to its biological effects. Due to the chiral nature of its receptors in the body, the (S)-enantiomer is substantially more active. For this reason, nearly all pharmacological and toxicological data is based on studies of (S)-nicotine. (S)-Nicotine is 4–28 times more potent than (R)-nicotine in standard nicotinic receptor binding and functional assays and elicits stronger nasal irritation, stinging, and mucosal responses at lower detection thresholds—yet smokers rated it as more pleasant in the only human sensory study.[116][117] The pharmacological, metabolic, and toxicological effects of (R)-nicotine and of racemic (R)/(S)-nicotine mixtures in humans remain poorly understood, with data largely limited to animal studies.[116]
Central nervous system
Acute effects on CNS
By binding to nicotinic acetylcholine receptors in the brain, nicotine elicits its psychoactive effects and increases the levels of several neurotransmitters in various brain structures – acting as a sort of "volume control".[118][119] Nicotine has a higher affinity for nicotinic receptors in the brain than those in skeletal muscle, though at toxic doses it can induce contractions and respiratory paralysis.[120] Nicotine's selectivity is thought to be due to a particular amino acid difference on these receptor subtypes.[121] Nicotine is unusual in comparison to most drugs, as its profile changes from stimulant to sedative with increasing dosages, a phenomenon known as "Nesbitt's paradox" after the doctor who first described it in 1969.[122][123] At very high doses it dampens neuronal activity.[124] Nicotine induces both behavioral stimulation and anxiety in animals.[107] Research into nicotine's most predominant metabolite, cotinine, suggests that some of nicotine's psychoactive effects are mediated by cotinine.[125]
Nicotine activates nicotinic receptors (particularly α4β2 nicotinic receptors, but also α5 nAChRs) on neurons that innervate the ventral tegmental area and within the mesolimbic pathway where it appears to cause the release of dopamine.[126][127] This nicotine-induced dopamine release occurs at least partially through activation of the cholinergic–dopaminergic reward link in the ventral tegmental area.[127][128] Nicotine can modulate the firing rate of the ventral tegmental area neurons.[128] These actions are largely responsible for the strongly reinforcing effects of nicotine, which often occur in the absence of euphoria;[126] however, mild euphoria from nicotine use can occur in some individuals.[126]
Long-term effects on CNS
Chronic exposure to nicotine induces several molecular changes in neuronal systems, particularly within the mesolimbic dopamine pathway and associated circuits. These adaptations include desensitization and upregulation of nAChRs and downregulation of related enzymes (e.g. class I and II histone deacetylases in the striatum), alterations in transcription factors, and modifications to dopamine synthesis and release.[129][130]
Nicotine binds to presynaptic and postsynaptic nAChRs, leading to initial activation followed by desensitization—a conformational shift rendering receptors temporarily unresponsive.[131][132] Chronic nicotine exposure promotes upregulation of nAChRs in brain regions like the ventral tegmental area and striatum, with increased receptor density observed within 1–7 days and peaking after 10–14 days in rodent models.[133] Human imaging studies show this upregulation is temporary and returns to baseline levels in nonsmokers by approximately 21 days after smoking cessation but full recovery taking 6-12 weeks.[134][135]
Chronic nicotine use also leads to accumulation of the transcription factor ΔFosB in dopamine D1-type medium spiny neurons of the nucleus accumbens, a process implicated in sustained reward pathway modifications.[136] This elevation is longer-lasting and persists "for weeks and months even when substance use has ceased."[137]
Additionally, positron emission tomography (PET) studies indicate reduced presynaptic dopamine synthesis capacity in the striatum of chronic smokers, as measured by 18F-DOPA uptake. This deficit, approximately 15–20% lower than in nonsmokers, normalizes after about 3 months of abstinence.[138][139]
A 2016 study found that nicotine exposure creates long-lasting malleable circuits 7 months after the initial exposure to nicotine and 6 months after stopping its administration.[140] Other studies suggest broader neuronal recovery, such as normalization of dopamine transporter (DAT) levels in reward centers, may extend up to 12–14 months in some cases of substance dependence affecting dopamine levels, though specific data for nicotine are limited.[141]
Sympathetic nervous system
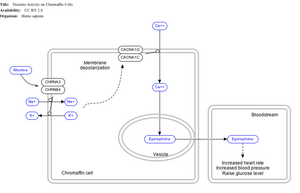
Nicotine also activates the sympathetic nervous system,[142] acting via splanchnic nerves to the adrenal medulla, stimulating the release of epinephrine. Acetylcholine released by preganglionic sympathetic fibers of these nerves acts on nicotinic acetylcholine receptors, causing the release of epinephrine (and norepinephrine) into the bloodstream.
Adrenal medulla
By binding to ganglion type nicotinic receptors in the adrenal medulla, nicotine increases flow of adrenaline (epinephrine), a stimulating hormone and neurotransmitter. By binding to the receptors, it causes cell depolarization and an influx of calcium through voltage-gated calcium channels. Calcium triggers the exocytosis of chromaffin granules and thus the release of epinephrine (and norepinephrine) into the bloodstream. The release of epinephrine (adrenaline) causes an increase in heart rate, blood pressure and respiration, as well as higher blood glucose levels.[143]
Pharmacokinetics

As nicotine enters the body, it is distributed quickly through the bloodstream and crosses the blood–brain barrier reaching the brain within 10–20 seconds after inhalation.[145] The elimination half-life of nicotine in the body is around two hours.[146][147] Nicotine is primarily excreted in urine and urinary concentrations vary depending upon urine flow rate and urine pH.[107]
The amount of nicotine absorbed by the body from smoking can depend on many factors, including the types of tobacco, whether the smoke is inhaled, and whether a filter is used. However, it has been found that the nicotine yield of individual products has only a small effect (4.4%) on the blood concentration of nicotine,[148] suggesting "the assumed health advantage of switching to lower-tar and lower-nicotine cigarettes may be largely offset by the tendency of smokers to compensate by increasing inhalation".
Cotinine is an active metabolite of nicotine that remains in the blood with a half-life of 18–20 hours, making it easier to analyze due to longer half-life than that of nicotine itself.[149]
Nicotine is metabolized in the liver by cytochrome P450 enzymes (mostly CYP2A6, and also by CYP2B6) and FMO3, which selectively metabolizes (S)-nicotine. A major metabolite is cotinine. Other primary metabolites include nicotine N-oxide, nornicotine, nicotine isomethonium ion, 2-hydroxynicotine and nicotine glucuronide.[150] Under some conditions, other substances may be formed such as myosmine.[151][152]
Glucuronidation and oxidative metabolism of nicotine to cotinine are both inhibited by menthol, an additive to mentholated cigarettes, thus increasing the half-life of nicotine in vivo.[153]
Influence of ionization state
Nicotine's absorption is modulated by its ionization state, governed by pH relative to pKa values (8.10 for pyrrolidine nitrogen, 3.41 for pyridine). At physiological pH (~7.4), it is mostly monoprotonated (cationic); above pH 8, it becomes unprotonated free-base, which is lipophilic and volatile[154]. The distribution of nicotine among its free-base and protonated forms in aerosolised nicotine affects inhalability; it has been manipulated in tobacco smoke and now in electronic cigarettes by the use of acids to de-freebase nicotine and form 'nicotine salts'[155]. Pod mod electronic cigarettes use nicotine in the form of a protonated nicotine, rather than free-base nicotine found in earlier generations.[156]
Free-base nicotine enables rapid membrane diffusion and higher bioavailability in early tobacco/oral studies. Yet, recent e-cigarette research contradicts this: protonated salts (e.g., nicotine benzoate, lactate, levulinate from acid addition) yield higher Cmax and faster onset than equivalent free-base[154]. For example, 2% benzoate salt produced 3x higher Cmax in human puffing trials. Notably, Cmax of protonated nicotine salts appears independent of the composition and identity of the counter anions (e.g., benzoate, lactate, levulinate) forming the salts for higher administered nicotine formulations[157].
These effects stem from aerosol dynamics—salts form low-volatility submicron particles for deeper lung deposition and less exhalation loss, versus free-base's superficial deposition. Sensorily, free-base delivers a harsh throat hit, while salts allow smoother high-dose inhalation, boosting appeal and intake[154].
Metabolism
Nicotine decreases hunger and as a consequence food consumption, alongside increasing energy expenditure.[158][159] The majority of research shows that nicotine reduces body weight, but some researchers have found that nicotine may result in weight gain under specific types of eating habits in animal models.[159] Nicotine effect on weight appears to result from nicotine's stimulation of α3β4 nAChR receptors located in the POMC neurons in the arcuate nucleus and subsequently the melanocortin system, especially the melanocortin-4 receptors on second-order neurons in the paraventricular nucleus of the hypothalamus, thus modulating feeding inhibition.[128][159] POMC neurons are a precursor of the melanocortin system, a critical regulator of body weight and peripheral tissue such as skin and hair.[159]
Chemistry
| NFPA 704 fire diamond | |
|---|---|
The fire diamond hazard sign for nicotine[160] |
Nicotine is a very hygroscopic, colorless to pale yellow, oily liquid that gradually turns brown on exposure to air or light.[55][161][162] It develops a characteristic pungent, fishy odor of pyridine and has an acrid burning taste.[161] It is very soluble in alcohol, chloroform, ether, light petroleum, kerosene, or oils.[55][161] It is miscible with water in its neutral amine base form between 60 °C and 210 °C. It is a dibasic nitrogenous base, having Kb1=1×10−6, Kb2=1×10−11.[163] It readily forms ammonium salts with acids that are usually solid and water-soluble. Its flash point is 95 °C, and its auto-ignition temperature is 244 °C.[164] Nicotine is volatile (vapor pressure 5.5 Pa at 25 °C)[163] On exposure to ultraviolet light or various oxidizing agents, nicotine is converted to nicotine oxide, nicotinic acid (niacin, a B3 vitamer), and methylamine.[165]
Anabasine is a structural isomer of nicotine, as both compounds have the molecular formula C
10H
14N
2.
Stereochemistry
Nicotine has a chiral center at the C2' position of the pyrrolidine ring, and therefore exists as two enantiomers: (S)-nicotine and (R)-nicotine.[166]
- (S)-Nicotine − this naturally occurring form of nicotine, found in tobacco plants at over 99% purity, is levorotatory with a specific rotation of [α]D(20°C)=–169.3°.[161][167]
- (R)-Nicotine − this is the dextrorotatory form that is physiologically less active and less toxic than (S)-nicotine.[168][169]
The salts of (S)-nicotine are usually dextrorotatory; this conversion between levorotatory and dextrorotatory upon protonation is common among alkaloids.[165] The hydrochloride and sulfate salts become optically inactive if heated in a closed vessel above 180 °C.[165]
The most common chemistry synthetic methods for generating nicotine yield a product that is approximately equal proportions of the S- and R-enantiomers.[170] Tobacco-derived nicotine (>99% (S)-enantiomer) is distinguishable from synthetic nicotine (typically racemic, 50:50 (S)/(R)) by enantiomeric ratio analysis, although strategies exist for adjusting the relative levels of the enantiomers or performing a synthesis that only leads to the pure S-enantiomer.[167][171] Synthetic stereospecific (S)-nicotine has become available on the market to consumers of electronic cigarette products.[171][116] Nicotine enantiomers differ in their biological effects on animals.[116][117]

Preparation
The first laboratory preparation of nicotine (as its racemate) was described in 1904.[172]
The starting material was an N-substituted pyrrole derivative, which was heated to convert it by a [1,5] sigmatropic shift to the isomer with a carbon bond between the pyrrole and pyridine rings, followed by methylation and selective reduction of the pyrrole ring using tin and hydrochloric acid.[172][173] Many other syntheses of nicotine, in both racemic and chiral forms have since been published.[174]
Biosynthesis
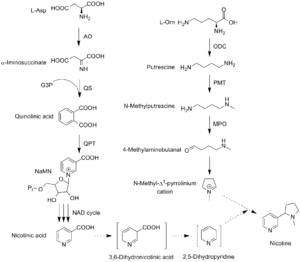
The biosynthetic pathway of nicotine involves a coupling reaction between the two cyclic structures that comprise nicotine. Metabolic studies show that the pyridine ring of nicotine is derived from nicotinic acid, while the pyrrolidine is derived from N-methyl-Δ1-pyrrollidium cation.[175][176] Biosynthesis of the two component structures proceeds via two independent syntheses, the NAD pathway for nicotinic acid and the tropane pathway for N-methyl-Δ1-pyrrollidium cation.
The final step in the synthesis of nicotine is the coupling between N-methyl-Δ1-pyrrollidium cation and nicotinic acid. Although studies conclude some form of coupling between the two component structures, the definite process and mechanism remains undetermined. The current agreed theory involves the conversion of nicotinic acid into 2,5-dihydropyridine through 3,6-dihydronicotinic acid. The 2,5-dihydropyridine intermediate would then react with N-methyl-Δ1-pyrrollidium cation to form enantiomerically pure (−)-nicotine.[177]
Detection in body fluids
Nicotine can be quantified in blood, plasma, or urine to confirm a diagnosis of poisoning or to facilitate a medicolegal death investigation. Urinary or salivary cotinine concentrations are frequently measured for the purposes of pre-employment and health insurance medical screening programs. Careful interpretation of results is important, since passive exposure to cigarette smoke can result in significant accumulation of nicotine, followed by the appearance of its metabolites in various body fluids.[178][179] Nicotine use is not regulated in competitive sports programs.[180]
Methods for analysis of enantiomers
Methods for measuring the two enantiomers are straightforward and include normal-phase liquid chromatography,[167] liquid chromatography with a chiral column.[181] However, since methods can be used to alter the two enantiomers, it may not be possible to distinguish tobacco-derived from synthetic nicotine simply by measuring the levels of the two enantiomers. A new approach uses hydrogen and deuterium nuclear magnetic resonance to distinguish tobacco-derived and synthetic nicotine based on differences the substrates used in the natural synthetic pathway performed in the tobacco plant and the substrates most used in synthesis.[182] Another approach measures the carbon-14 content which also differs between natural and laboratory-based tobacco.[183] These methods remain to be fully evaluated and validated using a wide range of samples.
Analogues and derivatives
Analogues and derivatives of nicotine are known.[184][185][186][187][188] These compounds, often structurally similar and sharing affinity for nicotinic acetylcholine receptors, have applications in pharmacology (e.g., smoking cessation), pest control, and neuroscience research (e.g., multiple domain cognitive enhancement, neuroprotection).[189][190]
Natural analogues
Natural analogues of nicotine, often found in plants or other biological sources, include anabasine (from Anabasis aphylla), anatabine (from tobacco), arecoline (from betel nut), cotinine (major metabolite of nicotine), cytisine (from Laburnum species), and epibatidine (from frog skin), among others.[191][184]
Synthetic analogues
Synthetic analogues and derivatives, typically developed for research or therapeutic purposes, include altinicline, 6-chloronicotine, dianicline, levamisole, RJR-2429, TC-1698, UB-165, GTS-21, and varenicline, among others.[191][184]
Natural occurrence
Nicotine is a secondary metabolite produced in a variety of plants in the family Solanaceae, most notably in tobacco Nicotiana tabacum, where it can be found at high concentrations of 0.5 to 7.5%.[192] Nicotine is also present in other tobacco species, such as Nicotiana rustica (in amounts of 2–14%).[163] Nicotine production is strongly induced in response to wounding as part of a jasmonate-dependent reaction.[193] Specialist insects on tobacco, such as the tobacco hornworm (Manduca sexta), have a number of adaptations to the detoxification and even adaptive re-purposing of nicotine.[194] Nicotine is also found at low concentrations in the nectar of tobacco plants, where it may promote outcrossing by affecting the behavior of hummingbird pollinators.[195]
Nicotine occurs in smaller amounts (varying from 2–7 μg/kg, or 20–70 millionths of a percent wet weight[196]) in other Solanaceaeous plants, including some crop species such as potatoes, tomatoes, eggplant, and peppers,[196][197] as well as non-crop species such as Duboisia hopwoodii.[163] The amounts of nicotine in tomatoes lowers substantially as the fruit ripens.[196] A 1999 report found "In some papers it is suggested that the contribution of dietary nicotine intake is significant when compared with exposure to ETS [environmental tobacco smoke] or by active smoking of small numbers of cigarettes. Others consider the dietary intake to be negligible unless inordinately large amounts of specific vegetables are consumed."[196] The amount of nicotine eaten per day is roughly around 1.4 and 2.25 μg/day at the 95th percentile.[196] These numbers may be low due to insufficient food intake data.[196] The concentrations of nicotine in vegetables are difficult to measure accurately, since they are very low (parts per billion range).[198] Pure nicotine tastes "terrible".[55]
History

Nicotine was originally isolated from the tobacco plant in 1828 by chemists Wilhelm Heinrich Posselt and Karl Ludwig Reimann from Germany, who believed it was a poison.[199][200] Its chemical empirical formula was described by Melsens in 1843,[201] its structure was discovered by Adolf Pinner and Richard Wolffenstein in 1893,[202][203][204][clarification needed] and it was first synthesized by Amé Pictet and A. Rotschy in 1904.[172][205]
Nicotine is named after the tobacco plant Nicotiana tabacum, which in turn is named after the French ambassador in Portugal, Jean Nicot de Villemain, who sent tobacco and seeds to Paris in 1560, presented to the French King,[206] and who promoted their medicinal use. Smoking was believed to protect against illness, particularly the plague.[206] However, the "holy herb", tobacco, had first reached Europe by the early 1530s, brought by Spanish explorers.[207]
Following its introduction, tobacco rapidly gained popularity in Europe for its stimulating effects, fueled by nicotine's addictive and pharmacological properties and tobacco's widespread embrace as a cure-all.[208] By the early 17th century, this allure fueled its mercilessly laborious cultivation as a cash crop in the Virginia colonies, where John Rolfe's introduction in 1612 rescued Jamestown from economic collapse and famine, transforming it into a prosperous export hub with over 20,000 pounds shipped by 1619 and laying the groundwork for transatlantic trade.[208] From the 17th century onward, tobacco smoking became virtually central to European social, economic, and cultural history and beyond, entrenching itself in daily rituals, trade networks, and even international conflicts.[208]
By the late 17th century, tobacco was used not only for smoking but also as an insecticide. After World War II, over 2,500 tons of nicotine insecticide were used worldwide, but by the 1980s the use of nicotine insecticide had declined below 200 tons. This was due to the availability of other insecticides that are cheaper and less harmful to mammals.[3]
The nicotine content of popular American-brand cigarettes has increased over time, and one study found that there was an average increase of 1.78% per year between the years of 1998 and 2005.[209]
Although methods of production of synthetic nicotine have existed for decades,[210] it was believed that the cost of making nicotine by laboratory synthesis was cost prohibitive compared to extracting nicotine from tobacco.[211] However, recently synthetic nicotine started to be found in different brands of e-cigarettes and oral pouches and marketed as "tobacco-free".[212]
Society and culture
Regulation
In the United States, the Food and Drug Administration (FDA) regulates nicotine as a tobacco product under the 2009 Family Smoking Prevention and Tobacco Control Act.[213] The FDA is tasked with reviewing tobacco products such as e-cigarettes and determining which can be authorized for sale.
In March 2022, the US Congress passed a law (the Consolidated Appropriations Act, 2022) that expanded FDA's tobacco regulatory authority to include tobacco products containing nicotine from any source, thereby including products made with synthetic nicotine.[213]
On January 17, 2025, the FDA proposed a strongly endorsed rule to reduce nicotine in cigarettes and certain combusted tobacco products to minimally or non-addictive levels, capping nicotine yield at 0.7 mg per gram of tobacco, approximately a 95% reduction from current commercial levels.[214][215]
In the European Union, the Tobacco Products Directive (2014/40/EU) regulates the manufacture, presentation, and sale of tobacco and related products.[216]
In the United Kingdom, the Tobacco and Related Products Regulations 2016 implemented the European directive 2014/40/EU, amended by Tobacco Products and Nicotine Inhaling Products (Amendment etc.) (EU Exit) Regulations 2019 and the Tobacco Products and Nicotine Inhaling Products (Amendment) (EU Exit) Regulations 2020. Additionally other regulations limit advertising, sale and display of tobacco products and other products containing nicotine for human consumption. The Sunak government proposed banning disposable vapes to limit their appeal and affordability for children and to reduce the amount of waste generated.
Age limits on purchase
In the United States, over-the-counter nicotine replacement therapy products are only available to people aged 18 and above, and it is not for sale in vending machines or from any source where proof of age cannot be verified. The minimum age to purchase tobacco products in the US is 21 at the federal level.[217]
In the European Union, the minimum age to purchase nicotine products is 18 in all member states except Latvia, where it is 20.[218] However, there is no minimum age requirement to use tobacco or nicotine products.[219]
In the United Kingdom, the minimum age to purchase tobacco products is 18.[220]
In media
The public's lack of understanding regarding nicotine's biological effects frequently results in inaccurate claims disseminated by the media and general public.[221]
In some anti-smoking literature, the harm that tobacco smoking and nicotine addiction does is personified as Nick O'Teen, represented as a humanoid with some aspect of a cigarette or cigarette butt about him or his clothes and hat.[222] Nick O'Teen was a villain that was created for the Health Education Council. The character was featured in three animated anti-smoking public service announcements in which he tries to get kids addicted to cigarettes before being foiled by the DC Comics character Superman.[222]
Nicotine was often compared to caffeine in advertisements in the 1980s by the tobacco industry, and later in the 2010s by the electronic cigarettes industry, in an effort to reduce the stigmatization and the public perception of the risks associated with nicotine use.[223]
Research
Central nervous system
While acute/initial nicotine intake causes activation of neuronal nicotinic receptors, chronic low doses of nicotine use leads to desensitization of those receptors (due to the development of tolerance) and results in an antidepressant effect, with early research showing low dose nicotine patches could be an effective treatment of major depressive disorder in non-smokers.[224] Nicotine anti-depressant effects have been documented in murine research.[225]
Though tobacco smoking is associated with an increased risk of Alzheimer's disease,[226] there is evidence that nicotine itself has the potential to prevent and treat Alzheimer's disease.[227]
Smoking is associated with a decreased risk of Parkinson's disease; however, it is unknown whether this is due to people with healthier brain dopaminergic reward centers (the area of the brain affected by Parkinson's) being more likely to enjoy smoking and thus pick up the habit, nicotine directly acting as a neuroprotective agent, or other compounds in cigarette smoke acting as neuroprotective agents.[228]
Nicotine may partly attenuate sensory gating and attentional deficits associated with schizophrenia. Short-term use of transdermal nicotine was found to improve subjects' reaction time and alertness in given tasks. Nicotine was not found to improve negative, positive, or other cognitive symptoms of schizophrenia.[229]
Nicotine dependence pathophysiology in heavy smokers suggests less efficient network architecture in the brain and disruptions in the topological organization of brain networks, with the altered brain network metrics correlated with the duration of cigarette use and the severity of nicotine dependence.[230]
Some long-term effects of nicotine may be irreversible because "it is entirely possible that doses of nicotine achieved in the brains of human smokers can damage or kill mHb [medial habenula] neurons that regulate nicotine avoidance behaviors", but more studies are needed to elucidate this underlying mechanism of nicotine-induced degeneration of the mHb-IPn circuit .[231]
Immune system
Immune cells of both the innate immune system and adaptive immune systems frequently express the α2, α5, α6, α7, α9, and α10 subunits of nicotinic acetylcholine receptors.[232] Evidence suggests that nicotinic receptors which contain these subunits are involved in the regulation of immune function.[232]
Although some of its effects are pro-inflammatory (e.g., inducing prostaglandin E2 production), nicotine effects are mostly anti-inflammatory.[168][233] Nicotine suppresses the innate and adaptive immune response by reducing the secretion of pro-inflammatory cytokines (IL-1, IL-6, TNF-α, IL-17, IL-21, and IL-22), reducing proliferation and activation of T-cells, and suppressing the activation of dendritic cells.[168] As a result, cell-mediated immunity against infection and neoplastic diseases is downregulated.[168] In vitro and animal studies also showed that nicotine reduces T cell receptor (TCR) signaling and suppresses the production and secretion of antibodies.[168]
Nicotine effects on immune system function can aggravate tumors (growth and metastases) in cancer patients and is found to have many positive effects in the treating autoimmune disease (e.g. inflammatory bowel disease/ulcerative colitis, arthritis), requiring further studies.[234][15]
Optopharmacology
A photoactivatable form of nicotine, which releases nicotine when exposed to ultraviolet light with certain conditions, has been developed for studying nicotinic acetylcholine receptors in brain tissue.[235]
Oral health
Many studies have shown the pro-inflammatory effect of nicotine on oral diseases.[234] Nicotine promotes and aggravates some diseases such as periodontitis and gingivitis, especially when there are harmful microorganisms in the oral cavity, however, the data is insufficient, especially in vivo.[234] Understanding the potential role of nicotine in oral health has become increasingly important given the recent introduction of novel nicotine products.[236]
See also
- 6-Chloronicotine
- Nicotine marketing
References
- ↑ "Nicotine: Pharmacology, Toxicity and Therapeutic use". Journal of Smoking Cessation 9 (2): 53–59. December 2014. doi:10.1017/jsc.2014.27.
- ↑ "Chemistry and toxicology.". Smoking and Tobacco Control Monograph No. 9. U.S. Department of Health and Human Services, Public Health Service, National Institutes of Health, National Cancer Institute. 1998. pp. 55–104. ISBN 978-0-7881-7301-1. https://cancercontrol.cancer.gov/sites/default/files/2020-06/m9_3.pdf. Retrieved 19 December 2012.
- ↑ 3.0 3.1 3.2 "Nicotine and Other Insecticidal Alkaloids". Nicotinoid Insecticides and the Nicotinic Acetylcholine Receptor. Tokyo: Springer-Verlag. 1999. pp. 29–69. doi:10.1007/978-4-431-67933-2_2. ISBN 978-4-431-67933-2.
- ↑ "Drugs of abuse and blood-brain barrier endothelial dysfunction: A focus on the role of oxidative stress". Journal of Cerebral Blood Flow and Metabolism 36 (3): 539–554. March 2016. doi:10.1177/0271678X15616978. PMID 26661236.
- ↑ 5.0 5.1 5.2 "How much nicotine kills a human? Tracing back the generally accepted lethal dose to dubious self-experiments in the nineteenth century". Archives of Toxicology 88 (1): 5–7. January 2014. doi:10.1007/s00204-013-1127-0. PMID 24091634. Bibcode: 2014ArTox..88....5M.
- ↑ https://ash.org.uk/resources/view/whats-in-a-cigarette#:~:text=12-,Under%20the%20terms%20of%20the%202001%20EU%20Tobacco%20Products%20Directive%2C%20maximum%20yields%20for%20cigarettes%20were%20set%20for%20tar%20(10mg)%2C%20carbon%20monoxide%20(10mg)%20and%20nicotine%20(1mg).,-13
- ↑ https://eur-lex.europa.eu/EN/legal-content/summary/tighter-eu-rules-on-tobacco.html#:~:text=Ingredients%20and%20maximum,of%20carbon%20monoxide.
- ↑ CDC (2025-01-31). "Health Effects of Vaping" (in en-us). https://www.cdc.gov/tobacco/e-cigarettes/health-effects.html.
- ↑ "Reinforcement enhancing effects of nicotine via smoking". Psychopharmacology 228 (3): 479–486. August 2013. doi:10.1007/s00213-013-3054-4. PMID 23494236.
- ↑ "Nicotine: Clinical data". International Union of Basic and Clinical Pharmacology. http://www.guidetopharmacology.org/GRAC/LigandDisplayForward?tab=clinical&ligandId=2585. "Used as an aid to smoking cessation and for the relief of nicotine withdrawal symptoms."
- ↑ "Addiction to the nicotine gum in never smokers". BMC Public Health 7. July 2007. doi:10.1186/1471-2458-7-159. PMID 17640334.
- ↑ "Varenicline: an alpha4beta2 nicotinic receptor partial agonist for smoking cessation". Journal of Medicinal Chemistry 48 (10): 3474–3477. May 2005. doi:10.1021/jm050069n. PMID 15887955. Bibcode: 2005JMedC..48.3474C.
- ↑ 13.0 13.1 13.2 13.3 "Chapter 5 - Nicotine". The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. Surgeon General of the United States. 2014. pp. 107–138. https://stacks.cdc.gov/view/cdc/21569/Share. Retrieved 26 July 2019.
- ↑ "Pulmonary effects of e-liquid flavors: a systematic review". Journal of Toxicology and Environmental Health Part B: Critical Reviews 25 (7): 343–371. October 2022. doi:10.1080/10937404.2022.2124563. PMID 36154615. Bibcode: 2022JTEHB..25..343E.
- ↑ 15.0 15.1 15.2 "Effect of Nicotine on Immune System Function". Advanced Pharmaceutical Bulletin 13 (1): 69–78. January 2023. doi:10.34172/apb.2023.008. PMID 36721811.
- ↑ "Nicotine during pregnancy: changes induced in neurotransmission, which could heighten proclivity to addict and induce maladaptive control of attention". Journal of Developmental Origins of Health and Disease 6 (3): 169–181. June 2015. doi:10.1017/S2040174414000531. PMID 25385318.
- ↑ U.S. Department of Health and Human Services. (2016). E-Cigarette Use Among Youth and Young Adults. A Report of the Surgeon General.. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic, Disease Prevention and Health Promotion, Office on Smoking and Health. https://e-cigarettes.surgeongeneral.gov/documents/2016_SGR_Full_Report_508.pdf.
- ↑ 18.0 18.1 18.2 "Nicotine replacement therapy versus control for smoking cessation". The Cochrane Database of Systematic Reviews 5 (5). May 2018. doi:10.1002/14651858.CD000146.pub5. PMID 29852054. "There is high-quality evidence that all of the licensed forms of NRT (gum, transdermal patch, nasal spray, inhalator and sublingual tablets/lozenges) can help people who make a quit attempt to increase their chances of successfully stopping smoking. NRTs increase the rate of quitting by 50% to 60%, regardless of setting, and further research is very unlikely to change our confidence in the estimate of the effect. The relative effectiveness of NRT appears to be largely independent of the intensity of additional support provided to the individual.
A meta-analysis of adverse events associated with NRT included 92 RCTs and 28 observational studies, and addressed a possible excess of chest pains and heart palpitations among users of NRT compared with placebo groups (Mills 2010). The authors report an OR of 2.06 (95% CI 1.51 to 2.82) across 12 studies. We replicated this data collection exercise and analysis where data were available (included and excluded) in this review, and detected a similar but slightly lower estimate, OR 1.88 (95% CI 1.37 to 2.57; 15 studies; 11,074 participants; OR rather than RR calculated for comparison; Analysis 6.1). Chest pains and heart palpitations were an extremely rare event, occurring at a rate of 2.5% in the NRT groups compared with 1.4% in the control groups in the 15 trials in which they were reported at all. A recent network meta-analysis of cardiovascular events associated with smoking cessation pharmacotherapies (Mills 2014), including 21 RCTs comparing NRT with placebo, found statistically significant evidence that the rate of cardiovascular events with NRT was higher (RR 2.29 95% CI 1.39 to 3.82). However, when only serious adverse cardiac events (myocardial infarction, stroke and cardiovascular death) were considered, the finding was not statistically significant (RR 1.95 95% CI 0.26 to 4.30).". - ↑ "Different doses, durations and modes of delivery of nicotine replacement therapy for smoking cessation". The Cochrane Database of Systematic Reviews 2023 (6). June 2023. doi:10.1002/14651858.CD013308.pub2. PMID 37335995.
- ↑ 20.0 20.1 "Why all stimulant drugs are damaging to recreational users: an empirical overview and psychobiological explanation". Human Psychopharmacology 30 (4): 213–224. July 2015. doi:10.1002/hup.2468. PMID 26216554.
- ↑ 21.0 21.1 "Neonicotinoid insecticide toxicology: mechanisms of selective action". Annual Review of Pharmacology and Toxicology 45: 247–268. 2005. doi:10.1146/annurev.pharmtox.45.120403.095930. PMID 15822177.
- ↑ The chemical components of tobacco and tobacco smoke. Boca Raton, FL: CRC Press. 2009. ISBN 978-1-4200-7883-1.
- ↑ "Tobacco and its evil cousin nicotine are good as a pesticide – American Chemical Society" (in en). https://www.acs.org/content/acs/en/pressroom/presspacs/2010/acs-presspac-october-27-2010/tobacco-and-its-evil-cousin-nicotine-are-good-as-a-pesticide.html.
- ↑ "Nicotine; Product Cancellation Order". Federal Register: 26695–26696. 3 June 2009. https://federalregister.gov/a/E9-12561. Retrieved 8 April 2012.
- ↑ US Code of Federal Regulations. 7 CFR 205.602 – Nonsynthetic substances prohibited for use in organic crop production
- ↑ "Safety for Homemade Remedies for Pest Control". Montana State University. 5 September 2014. http://www.pesticides.montana.edu/documents/mt-pesticide-bulletins/2009_05_MPB.pdf.
- ↑ 27.0 27.1 "[How does a pesticide residue turn into a contaminant?]" (in de). Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz 60 (7): 768–773. July 2017. doi:10.1007/s00103-017-2556-3. PMID 28508955.
- ↑ "Potential risks for public health due to the presence of nicotine in wild mushrooms". EFSA Journal 7 (5): 286r. 7 May 2009. doi:10.2903/j.efsa.2009.286r.
- ↑ "Developmental neurotoxicity of succeeding generations of insecticides". Environment International 99: 55–77. February 2017. doi:10.1016/j.envint.2016.11.019. PMID 27908457. Bibcode: 2017EnInt..99...55A.
- ↑ "Cognitive Effects of Nicotine: Recent Progress". Current Neuropharmacology (Bentham Science Publishers) 16 (4): 403–414. May 2018. doi:10.2174/1570159X15666171103152136. PMID 29110618.
- ↑ "Meta-analysis of the acute effects of nicotine and smoking on human performance". Psychopharmacology 210 (4): 453–469. July 2010. doi:10.1007/s00213-010-1848-1. PMID 20414766.
- ↑ "Behavioral-Cognitive Targets for Cholinergic Enhancement". Current Opinion in Behavioral Sciences 4: 22–26. August 2015. doi:10.1016/j.cobeha.2015.01.004. PMID 28607947.
- ↑ "Nicotine: Biological activity". International Union of Basic and Clinical Pharmacology. http://www.guidetopharmacology.org/GRAC/LigandDisplayForward?tab=biology&ligandId=2585. "Kis as follows; α2β4=9900nM [5], α3β2=14nM [1], α3β4=187nM [1], α4β2=1nM [4,6]. Due to the heterogeneity of nACh channels we have not tagged a primary drug target for nicotine, although the α4β2 is reported to be the predominant high affinity subtype in the brain which mediates nicotine addiction"
- ↑ "Revisiting nicotine's role in the ageing brain and cognitive impairment". Reviews in the Neurosciences 28 (7): 767–781. October 2017. doi:10.1515/revneuro-2017-0008. PMID 28586306. https://findresearcher.sdu.dk/ws/files/140909555/Revisiting_nicotine_s_role_in_the_ageing_brain_and_cognitive_impairment.pdf.
- ↑ "Biospecimens and the ABCD study: Rationale, methods of collection, measurement and early data". Developmental Cognitive Neuroscience 32: 97–106. August 2018. doi:10.1016/j.dcn.2018.03.005. PMID 29606560.
- ↑ 36.0 36.1 "Nicotine and Tobacco as Substances of Abuse in Children and Adolescents". Pediatrics 139 (1). January 2017. doi:10.1542/peds.2016-3436. PMID 27994114.
- ↑ 37.0 37.1 "The scientific case that nicotine is addictive". Psychopharmacology 117 (1): 2–10; discussion 14–20. January 1995. doi:10.1007/BF02245088. PMID 7724697.
- ↑ "Nicotine without smoke: Tobacco harm reduction". UK: Royal College of Physicians. April 2016. pp. 58, 125. https://www.rcplondon.ac.uk/projects/outputs/nicotine-without-smoke-tobacco-harm-reduction-0.
- ↑ 39.0 39.1 39.2 "Biocatalytic and semisynthetic studies of the anticancer tobacco cembranoids". Expert Opinion on Investigational Drugs 16 (6): 877–887. June 2007. doi:10.1517/13543784.16.6.877. PMID 17501699.
- ↑ "Electronic cigarettes: patterns of use, health effects, use in smoking cessation and regulatory issues". Tobacco Induced Diseases 12 (1): 21. 2014. doi:10.1186/1617-9625-12-21. PMID 25745382.
- ↑ "The Potential Impact of Oral Nicotine Pouches on Public Health: A Scoping Review". Nicotine & Tobacco Research 27 (4): 598–610. March 2025. doi:10.1093/ntr/ntae131. PMID 38880491.
- ↑ "2003: The 3rd Annual Year In Ideas; Nicotini, The". The New York Times Magazine. 14 December 2003. https://www.nytimes.com/2003/12/14/magazine/2003-the-3rd-annual-year-in-ideas-nicotini-the.html.
- ↑ "The safety of treatments for tobacco use disorder". Expert Opinion on Drug Safety 15 (3): 333–341. 2016. doi:10.1517/14740338.2016.1131817. PMID 26715118.
- ↑ "Pharmacotherapy for smoking cessation: pharmacological principles and clinical practice". British Journal of Clinical Pharmacology 77 (2): 324–336. February 2014. doi:10.1111/bcp.12116. PMID 23488726.
- ↑ 45.0 45.1 45.2 "Efficacy and tolerability of pharmacotherapies to aid smoking cessation in adolescents". Paediatric Drugs 14 (2): 91–108. April 2012. doi:10.2165/11594370-000000000-00000. PMID 22248234.
- ↑ "Electronic Cigarettes – What are the health effects of using e-cigarettes?". Centers for Disease Control and Prevention. 22 February 2018. https://www.cdc.gov/tobacco/basic_information/e-cigarettes/pdfs/Electronic-Cigarettes-Infographic-508.pdf. "Nicotine is a health danger for pregnant women and their developing babies."
- ↑ "Long-term consequences of fetal and neonatal nicotine exposure: a critical review". Toxicological Sciences 116 (2): 364–374. August 2010. doi:10.1093/toxsci/kfq103. PMID 20363831. "there is no safe dose of nicotine during pregnancy... The general consensus among clinicians is that more information is needed about the risks of NRT use during pregnancy before well-informed definitive recommendations can be made to pregnant women... Overall, the evidence provided in this review overwhelmingly indicates that nicotine should no longer be considered the safe component of cigarette smoke. In fact, many of the adverse postnatal health outcomes associated with maternal smoking during pregnancy may be attributable, at least in part, to nicotine alone.".
- ↑ "Controversy and evidence about nicotine replacement therapy in pregnancy". MCN: The American Journal of Maternal/Child Nursing 35 (2): 89–95. 1 March 2010. doi:10.1097/NMC.0b013e3181cafba4. PMID 20215949.
- ↑ "2018 ACC Expert Consensus Decision Pathway on Tobacco Cessation Treatment: A Report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents". Journal of the American College of Cardiology 72 (25): 3332–3365. December 2018. doi:10.1016/j.jacc.2018.10.027. PMID 30527452.
- ↑ "Nicotine: Carcinogenicity and Effects on Response to Cancer Treatment - A Review". Frontiers in Oncology 5: 196. 2015. doi:10.3389/fonc.2015.00196. PMID 26380225.
- ↑ 51.0 51.1 51.2 51.3 51.4 51.5 51.6 51.7 "Nicotine". American Society of Health-System Pharmacists. https://www.drugs.com/monograph/nicotine.html.
- ↑ Detailed reference list is located on a separate image page.
- ↑ Textbook of Forensic Medicine & Toxicology: Principles & Practice (5th ed.). Elsevier Health Sciences. 2014. p. 525. ISBN 978-81-312-3623-9. https://books.google.com/books?id=Ip1rAwAAQBAJ. Extract of page 525
- ↑ "NICOTINE: Systemic Agent". 8 July 2021. https://www.cdc.gov/niosh/ershdb/emergencyresponsecard_29750028.html.
- ↑ 55.0 55.1 55.2 55.3 "Nicotine" (in en). Dictionary of Toxicology. Singapore: Springer Nature. 2024. p. 691. doi:10.1007/978-981-99-9283-6_1860. ISBN 978-981-99-9282-9. https://link.springer.com/10.1007/978-981-99-9283-6_1860. Retrieved 2024-10-19. "Nicotine is a colorless, water-soluble, and extremely hazardous alkaloid. It also has a terrible taste."
- ↑ CDC (2025-01-31). "Health Effects of Vaping" (in en-us). https://www.cdc.gov/tobacco/e-cigarettes/health-effects.html.
- ↑ Royal College of Physicians. "Nicotine Without Smoke -- Tobacco Harm Reduction". p. 125. https://www.rcplondon.ac.uk/file/3563/download?token=Mu0K_ZR0. "Use of nicotine alone, in the doses used by smokers, represents little if any hazard to the user."
- ↑ "The American Cancer Society public health statement on eliminating combustible tobacco use in the United States". CA 68 (4): 240–245. July 2018. doi:10.3322/caac.21455. PMID 29889305. "It is the smoke from combustible tobacco products—not nicotine—that injures and kills millions of smokers.".
- ↑ "The Health Effects of Electronic Cigarettes". The New England Journal of Medicine 375 (14): 1372–1381. October 2016. doi:10.1056/NEJMra1502466. PMID 27705269. "Beyond its addictive properties, short-term or long-term exposure to nicotine in adults has not been established as dangerous".
- ↑ 60.0 60.1 "Electronic Cigarettes: Vulnerability of Youth". Pediatric Allergy, Immunology, and Pulmonology 28 (1): 2–6. March 2015. doi:10.1089/ped.2015.0490. PMID 25830075.
- ↑ "Nicotine and the Developing Human: A Neglected Element in the Electronic Cigarette Debate". American Journal of Preventive Medicine 49 (2): 286–293. August 2015. doi:10.1016/j.amepre.2015.01.015. PMID 25794473.
- ↑ 62.0 62.1 "Polysomnographic sleep disturbances in nicotine, caffeine, alcohol, cocaine, opioid, and cannabis use: A focused review". The American Journal on Addictions 24 (7): 590–598. October 2015. doi:10.1111/ajad.12291. PMID 26346395.
- ↑ "What keeps us awake: the neuropharmacology of stimulants and wakefulness-promoting medications". Sleep 27 (6): 1181–1194. September 2004. doi:10.1093/sleep/27.6.1181. PMID 15532213.
- ↑ "Effects of nicotine on sleep during consumption, withdrawal and replacement therapy". Sleep Medicine Reviews 13 (5): 363–377. October 2009. doi:10.1016/j.smrv.2008.12.003. PMID 19345124.
- ↑ 65.0 65.1 65.2 65.3 "Cardiovascular toxicity of nicotine: Implications for electronic cigarette use". Trends in Cardiovascular Medicine 26 (6): 515–523. August 2016. doi:10.1016/j.tcm.2016.03.001. PMID 27079891.
- ↑ "DeltaFosB: a sustained molecular switch for addiction". Proceedings of the National Academy of Sciences of the United States of America 98 (20): 11042–11046. September 2001. doi:10.1073/pnas.191352698. PMID 11572966. Bibcode: 2001PNAS...9811042N. "Although the ΔFosB signal is relatively long-lived, it is not permanent. ΔFosB degrades gradually and can no longer be detected in brain after 1–2 months of drug withdrawal ... Indeed, ΔFosB is the longest-lived adaptation known to occur in adult brain, not only in response to drugs of abuse, but to any other perturbation (that doesn't involve lesions) as well.".
- ↑ "Transcriptional mechanisms of drug addiction". Clinical Psychopharmacology and Neuroscience 10 (3): 136–143. December 2012. doi:10.9758/cpn.2012.10.3.136. PMID 23430970. "The 35–37 kD ΔFosB isoforms accumulate with chronic drug exposure due to their extraordinarily long half-lives. ... As a result of its stability, the ΔFosB protein persists in neurons for at least several weeks after cessation of drug exposure. ... ΔFosB overexpression in nucleus accumbens induces NFκB".
- ↑ "E-cigarettes: a scientific review". Circulation 129 (19): 1972–1986. May 2014. doi:10.1161/circulationaha.114.007667. PMID 24821826.
- ↑ "Reinforcing and other behavioral effects of nicotine". Neuroscience and Biobehavioral Reviews 5 (4): 487–495. December 1981. doi:10.1016/0149-7634(81)90019-1. PMID 7322454.
- ↑ 70.0 70.1 "Acetaldehyde enhances acquisition of nicotine self-administration in adolescent rats". Neuropsychopharmacology 30 (4): 705–712. April 2005. doi:10.1038/sj.npp.1300586. PMID 15496937.
- ↑ "Evidence Review of E-Cigarettes and Heated Tobacco Products". Public Health England.. 2018. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/684963/Evidence_review_of_e-cigarettes_and_heated_tobacco_products_2018.pdf.
- ↑ "Acetaldehyde". Rijksinstituut voor Volksgezondheid en Milieu (RIVM). Netherlands. https://www.rivm.nl/en/tobacco/harmful-substances-in-tobacco-smoke/acetaldehyde.
- ↑ 73.0 73.1 "Nicotine without smoke: Tobacco harm reduction". RCP London. 28 April 2016. https://www.rcplondon.ac.uk/projects/outputs/nicotine-without-smoke-tobacco-harm-reduction.
- ↑ "Effects of Monoamine Oxidase Inhibition on the Reinforcing Properties of Low-Dose Nicotine". Neuropsychopharmacology 41 (9): 2335–2343. August 2016. doi:10.1038/npp.2016.36. PMID 26955970.
- ↑ "Neuronal mechanisms underlying development of nicotine dependence: implications for novel smoking-cessation treatments". Addiction Science & Clinical Practice 6 (1): 4–16. July 2011. PMID 22003417. "Withdrawal symptoms upon cessation of nicotine intake: Chronic nicotine use induces neuroadaptations in the brain's reward system that result in the development of nicotine dependence. Thus, nicotine-dependent smokers must continue nicotine intake to avoid distressing somatic and affective withdrawal symptoms. Newly abstinent smokers experience symptoms such as depressed mood, anxiety, irritability, difficulty concentrating, craving, bradycardia, insomnia, gastrointestinal discomfort, and weight gain (Shiffman and Jarvik, 1976; Hughes et al., 1991). Experimental animals, such as rats and mice, exhibit a nicotine withdrawal syndrome that, like the human syndrome, includes both somatic signs and a negative affective state (Watkins et al., 2000; Malin et al., 2006). The somatic signs of nicotine withdrawal include rearing, jumping, shakes, abdominal constrictions, chewing, scratching, and facial tremors. The negative affective state of nicotine withdrawal is characterized by decreased responsiveness to previously rewarding stimuli, a state called anhedonia.".
- ↑ "Innovative approaches to support smoking cessation for individuals with mental illness and co-occurring substance use disorders". Expert Review of Respiratory Medicine 11 (10): 841–850. October 2017. doi:10.1080/17476348.2017.1361823. PMID 28756728.
- ↑ "Meta-analysis of the acute effects of nicotine and smoking on human performance". Psychopharmacology 210 (4): 453–469. July 2010. doi:10.1007/s00213-010-1848-1. PMID 20414766. "The significant effects of nicotine on motor abilities, attention, and memory likely represent true performance enhancement because they are not confounded by withdrawal relief. The beneficial cognitive effects of nicotine have implications for initiation of smoking and maintenance of tobacco dependence.".
- ↑ "Effects of abstinence from tobacco: valid symptoms and time course". Nicotine & Tobacco Research 9 (3): 315–327. March 2007. doi:10.1080/14622200701188919. PMID 17365764.
- ↑ 79.0 79.1 79.2 79.3 79.4 "Cigarette-derived nicotine is not a medicine". The World Journal of Biological Psychiatry 4 (2): 49–55. April 2003. doi:10.3109/15622970309167951. PMID 12692774.
- ↑ "Nicotine dependence and sleep quality in young adults". Addictive Behaviors 65: 154–160. February 2017. doi:10.1016/j.addbeh.2016.10.020. PMID 27816041.
- ↑ "Impaired sleep quality and sleep duration in smokers-results from the German Multicenter Study on Nicotine Dependence". Addiction Biology 19 (3): 486–496. May 2014. doi:10.1111/j.1369-1600.2012.00487.x. PMID 22913370.
- ↑ "Tobacco addiction and the dysregulation of brain stress systems". Neuroscience and Biobehavioral Reviews 36 (5): 1418–1441. May 2012. doi:10.1016/j.neubiorev.2012.02.015. PMID 22405889. "Discontinuation of smoking leads to negative affective symptoms such as depressed mood, increased anxiety, and impaired memory and attention...Smoking cessation leads to a relatively mild somatic withdrawal syndrome and a severe affective withdrawal syndrome that is characterized by a decrease in positive affect, an increase in negative affect, craving for tobacco, irritability, anxiety, difficulty concentrating, hyperphagia, restlessness, and a disruption of sleep. Smoking during the acute withdrawal phase reduces craving for cigarettes and returns cognitive abilities to pre-smoking cessation level".
- ↑ 83.0 83.1 "Cellular basis of memory for addiction". Dialogues in Clinical Neuroscience 15 (4): 431–443. December 2013. doi:10.31887/DCNS.2013.15.4/enestler. PMID 24459410.
- ↑ 84.0 84.1 "Molecular neurobiology of addiction: what's all the (Δ)FosB about?". The American Journal of Drug and Alcohol Abuse 40 (6): 428–437. November 2014. doi:10.3109/00952990.2014.933840. PMID 25083822. "The knowledge of ΔFosB induction in chronic drug exposure provides a novel method for the evaluation of substance addiction profiles (i.e. how addictive they are). Xiong et al. used this premise to evaluate the potential addictive profile of propofol (119). Propofol is a general anaesthetic, however its abuse for recreational purpose has been documented (120). Using control drugs implicated in both ΔFosB induction and addiction (ethanol and nicotine), ...
Conclusions
ΔFosB is an essential transcription factor implicated in the molecular and behavioral pathways of addiction following repeated drug exposure. The formation of ΔFosB in multiple brain regions, and the molecular pathway leading to the formation of AP-1 complexes is well understood. The establishment of a functional purpose for ΔFosB has allowed further determination as to some of the key aspects of its molecular cascades, involving effectors such as GluR2 (87,88), Cdk5 (93) and NFkB (100). Moreover, many of these molecular changes identified are now directly linked to the structural, physiological and behavioral changes observed following chronic drug exposure (60,95,97,102). New frontiers of research investigating the molecular roles of ΔFosB have been opened by epigenetic studies, and recent advances have illustrated the role of ΔFosB acting on DNA and histones, truly as a molecular switch (34). As a consequence of our improved understanding of ΔFosB in addiction, it is possible to evaluate the addictive potential of current medications (119), as well as use it as a biomarker for assessing the efficacy of therapeutic interventions (121,122,124).". - ↑ "Effects of chronic nicotine administration and its withdrawal on striatal FosB/DeltaFosB and c-Fos expression in rats and mice". Neuropharmacology 51 (1): 44–51. July 2006. doi:10.1016/j.neuropharm.2006.02.014. PMID 16631212.
- ↑ "Does nicotine cause cancer?". World Health Organization – International Agency for Research on Cancer. https://cancer-code-europe.iarc.fr/index.php/en/ecac-12-ways/tobacco/199-nicotine-cause-cancer.
- ↑ "The Promise of Vaping and the Rise of Juul". The New Yorker. May 7, 2018. https://www.newyorker.com/magazine/2018/05/14/the-promise-of-vaping-and-the-rise-of-juul. Retrieved June 29, 2024.
- ↑ "Nicotine: specific role in angiogenesis, proliferation and apoptosis". Critical Reviews in Toxicology 42 (1): 68–89. January 2012. doi:10.3109/10408444.2011.623150. PMID 22050423.
- ↑ "Chapter 4: Nicotine". Public Health Consequences of E-Cigarettes (Review). National Academies Press. 2018. ISBN 978-0-309-46834-3. https://www.ncbi.nlm.nih.gov/books/NBK507191/.
- ↑ 90.0 90.1 "Nicotine-mediated cell proliferation and tumor progression in smoking-related cancers". Molecular Cancer Research 12 (1): 14–23. January 2014. doi:10.1158/1541-7786.MCR-13-0541. PMID 24398389.
- ↑ "A Summary of In Vitro and In Vivo Studies Evaluating the Impact of E-Cigarette Exposure on Living Organisms and the Environment". International Journal of Molecular Sciences 21 (2): 652. January 2020. doi:10.3390/ijms21020652. PMID 31963832.
 This article incorporates text by Merecz-Sadowska A, Sitarek P, Zielinska-Blizniewska H, Malinowska K, Zajdel K, Zakonnik L, Zajdel R available under the CC BY 4.0 license.
This article incorporates text by Merecz-Sadowska A, Sitarek P, Zielinska-Blizniewska H, Malinowska K, Zajdel K, Zakonnik L, Zajdel R available under the CC BY 4.0 license.
- ↑ "Novel clinical therapeutics targeting the epithelial to mesenchymal transition". Clinical and Translational Medicine 3. 2014. doi:10.1186/s40169-014-0035-0. PMID 25343018.
- ↑ "Nornicotine nitrosation in saliva and its relation to endogenous synthesis of N'-nitrosonornicotine in humans". Nicotine & Tobacco Research 15 (2): 591–595. February 2013. doi:10.1093/ntr/nts172. PMID 22923602.
- ↑ "List of Classifications: N'-Nitrosonornicotine". IARC Monographs on the Identification of Carcinogenic Hazards to Humans. The International Agency for Research on Cancer (IARC); World Health Organization. https://monographs.iarc.fr/list-of-classifications. Retrieved 2020-07-22.
- ↑ "Nicotine: Carcinogenicity and Effects on Response to Cancer Treatment - A Review". Frontiers in Oncology 5: 196. 2015-08-31. doi:10.3389/fonc.2015.00196. PMID 26380225.
- ↑ "Nicotine derived genotoxic effects in human primary parotid gland cells as assessed in vitro by comet assay, cytokinesis-block micronucleus test and chromosome aberrations test". Toxicology in Vitro 28 (5): 838–846. August 2014. doi:10.1016/j.tiv.2014.03.012. PMID 24698733. Bibcode: 2014ToxVi..28..838G.
- ↑ "Assessment of nicotine-induced DNA damage in a genotoxicological test battery". Mutation Research 751 (1): 34–39. February 2013. doi:10.1016/j.mrgentox.2012.11.004. PMID 23200805. Bibcode: 2013MRGTE.751...34G.
- ↑ "Analysis of nicotine-induced DNA damage in cells of the human respiratory tract". Toxicology Letters 208 (1): 23–29. January 2012. doi:10.1016/j.toxlet.2011.09.029. PMID 22001448. Bibcode: 2012ToxL..208...23G.
- ↑ 99.0 99.1 99.2 99.3 99.4 99.5 99.6 99.7 99.8 "Nicotine". United States National Library of Medicine – Toxicology Data Network. Hazardous Substances Data Bank. 20 August 2009. https://www.nlm.nih.gov/toxnet/index.html.
- ↑ "Prenatal substance abuse: short- and long-term effects on the exposed fetus". Pediatrics 131 (3): e1009-24. March 2013. doi:10.1542/peds.2012-3931. PMID 23439891. Bibcode: 2013Pedia.131e1009B.
- ↑ "State Health Officer's Report on E-Cigarettes: A Community Health Threat". California Department of Public Health. January 2015. https://www.cdph.ca.gov/Programs/CCDPHP/DCDIC/CTCB/CDPH%20Document%20Library/Policy/ElectronicSmokingDevices/StateHealthEcigReport.pdf.
- ↑ "The effects of nicotine on human fetal development". Birth Defects Research. Part C, Embryo Today 108 (2): 181–192. June 2016. doi:10.1002/bdrc.21128. PMID 27297020.
- ↑ "Long-term consequences of fetal and neonatal nicotine exposure: a critical review". Toxicological Sciences 116 (2): 364–374. August 2010. doi:10.1093/toxsci/kfq103. PMID 20363831.
- ↑ "Consumer Updates: Nicotine Replacement Therapy Labels May Change". FDA. 1 April 2013. https://www.fda.gov/forconsumers/consumerupdates/ucm345087.htm.
- ↑ "Nicotine". European Chemicals Agency: Committee for Risk Assessment. September 2015. https://echa.europa.eu/documents/10162/31694def-b7c3-208d-5aaf-3db9681ec3b9.
- ↑ Toxicology and Applied Pharmacology. Vol. 44, Pg. 1, 1978.
- ↑ 107.0 107.1 107.2 Cite error: Invalid
<ref>tag; no text was provided for refs namedinchem - ↑ 108.0 108.1 "Nicotinic plant poisoning". Clinical Toxicology 47 (8): 771–81. September 2009. doi:10.1080/15563650903252186. PMID 19778187.
- ↑ "Cigarette and nicotine chewing gum toxicity in children". Human Toxicology 7 (1): 27–31. January 1988. doi:10.1177/096032718800700105. PMID 3346035. Bibcode: 1988HETox...7...27S.
- ↑ "Nicotiana glauca (tree tobacco) intoxication--two cases in one family". Journal of Medical Toxicology 7 (1): 47–51. March 2011. doi:10.1007/s13181-010-0102-x. PMID 20652661.
- ↑ "Green-tobacco sickness. An illness of tobacco harvesters". JAMA 229 (14): 1880–3. September 1974. doi:10.1001/jama.1974.03230520022024. PMID 4479133.
- ↑ "CDC – NIOSH Pocket Guide to Chemical Hazards – Nicotine". https://www.cdc.gov/niosh/npg/npgd0446.html.
- ↑ 113.0 113.1 "Nicotinic acetylcholine receptors: Introduction". International Union of Basic and Clinical Pharmacology. http://www.iuphar-db.org/DATABASE/FamilyIntroductionForward?familyId=76&familyType=IC.
- ↑ "Chapter 9: Autonomic Nervous System". Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. 2009. p. 234. ISBN 978-0-07-148127-4. "Nicotine ... is a natural alkaloid of the tobacco plant. Lobeline is a natural alkaloid of Indian tobacco. Both drugs are agonists are nicotinic cholinergic receptors ..."
- ↑ "Nicotine effects and the endogenous opioid system". Journal of Pharmacological Sciences 125 (2): 117–124. 2014. doi:10.1254/jphs.14R03CP. PMID 24882143.
- ↑ 116.0 116.1 116.2 116.3 "WHO study group on tobacco product regulation: report on the scientific basis of tobacco product regulation: ninth report of a WHO study group" (in en) (PDF). 23 August 2023. https://www.who.int/publications/i/item/9789240079410.
- ↑ 117.0 117.1 "A Systematic Review of Analytical Methods for the Separation of Nicotine Enantiomers and Evaluation of Nicotine Sources". Chemical Research in Toxicology 36 (3): 334–341. March 2023. doi:10.1021/acs.chemrestox.2c00310. PMID 36897818.
- ↑ "Neuroregulators and the reinforcement of smoking: towards a biobehavioral explanation". Neuroscience and Biobehavioral Reviews 8 (4): 503–13. 1984. doi:10.1016/0149-7634(84)90007-1. PMID 6151160.
- ↑ "Neuroregulatory effects of nicotine". Psychoneuroendocrinology 14 (6): 407–23. 1989. doi:10.1016/0306-4530(89)90040-1. PMID 2560221.
- ↑ Basic and Clinical Pharmacology. New York: McGraw-Hill Medical. 2006. pp. 99–105.
- ↑ "Nicotine binding to brain receptors requires a strong cation-pi interaction". Nature 458 (7237): 534–7. March 2009. doi:10.1038/nature07768. PMID 19252481. Bibcode: 2009Natur.458..534X.
- ↑ Nesbitt P (1969). Smoking, physiological arousal, and emotional response. Unpublished doctoral dissertation, Columbia University.
- ↑ "Nesbitt's Paradox resolved? Stress and arousal modulation during cigarette smoking". Addiction 93 (1): 27–39. January 1998. doi:10.1046/j.1360-0443.1998.931274.x. PMID 9624709.
- ↑ "Nicotine Replacement Therapy: An Overview". International Journal of Health Sciences 10 (3): 425–35. July 2016. doi:10.12816/0048737. PMID 27610066.
- ↑ "New Insights into the Mechanisms of Action of Cotinine and its Distinctive Effects from Nicotine". Neurochemical Research 40 (10): 2032–46. October 2015. doi:10.1007/s11064-014-1359-2. PMID 24970109.
- ↑ 126.0 126.1 126.2 Molecular Neuropharmacology: A Foundation for Clinical Neuroscience (2nd ed.). New York: McGraw-Hill Medical. 2009. pp. 369, 372–373. ISBN 978-0-07-148127-4.
- ↑ 127.0 127.1 "The role of the central ghrelin system in reward from food and chemical drugs". Molecular and Cellular Endocrinology 340 (1): 80–7. June 2011. doi:10.1016/j.mce.2011.02.017. PMID 21354264. https://gupea.ub.gu.se/bitstream/2077/26318/1/gupea_2077_26318_1.pdf. Retrieved 23 September 2019. "This reward link comprises a dopamine projection from the ventral tegmental area (VTA) to the nucleus accumbens together with a cholinergic input, arising primarily from the laterodorsal tegmental area.".
- ↑ 128.0 128.1 128.2 "Molecules and circuits involved in nicotine addiction: The many faces of smoking". Neuropharmacology 76 (Pt B): 545–53. January 2014. doi:10.1016/j.neuropharm.2013.04.028. PMID 23632083. "Rat studies have shown that nicotine administration can decrease food intake and body weight, with greater effects in female animals (Grunberg et al., 1987). A similar nicotine regimen also decreases body weight and fat mass in mice as a result of β4* nAChR-mediated activation of POMC neurons and subsequent activation of MC4 receptors on second order neurons in the paraventricular nucleus of the hypothalamus (Mineur et al., 2011).".
- ↑ "Epigenetics of Stress, Addiction, and Resilience: Therapeutic Implications". Molecular Neurobiology 53 (1): 545–560. January 2016. doi:10.1007/s12035-014-9040-y. PMID 25502297.
- ↑ "Molecules and circuits involved in nicotine addiction: The many faces of smoking". Neuropharmacology 76 (Pt B): 545–553. January 2014. doi:10.1016/j.neuropharm.2013.04.028. PMID 23632083.
- ↑ "Chronic exposure to nicotine upregulates the human (alpha)4((beta)2 nicotinic acetylcholine receptor function". The Journal of Neuroscience 21 (6): 1819–1829. March 2001. doi:10.1523/JNEUROSCI.21-06-01819.2001. PMID 11245666.
- ↑ "Addiction-related neuroadaptations following chronic nicotine exposure". Journal of Neurochemistry 157 (5): 1652–1673. June 2021. doi:10.1111/jnc.15356. PMID 33742685.
- ↑ "Inside-out neuropharmacology of nicotinic drugs". Neuropharmacology. The Nicotinic Acetylcholine Receptor: From Molecular Biology to Cognition 96 (Pt B): 178–193. September 2015. doi:10.1016/j.neuropharm.2015.01.022. PMID 25660637.
- ↑ "beta2-Nicotinic acetylcholine receptor availability during acute and prolonged abstinence from tobacco smoking". Archives of General Psychiatry 66 (6): 666–676. June 2009. doi:10.1001/archgenpsychiatry.2009.41. PMID 19487632.
- ↑ "New mechanisms and perspectives in nicotine withdrawal". Neuropharmacology. The Nicotinic Acetylcholine Receptor: From Molecular Biology to Cognition 96 (Pt B): 223–234. September 2015. doi:10.1016/j.neuropharm.2014.11.009. PMID 25433149.
- ↑ "ΔFOSB: A Potentially Druggable Master Orchestrator of Activity-Dependent Gene Expression". ACS Chemical Neuroscience 13 (3): 296–307. February 2022. doi:10.1021/acschemneuro.1c00723. PMID 35020364.
- ↑ "Exploring the potential link between ΔFosB and N-acetylcysteine in craving/relapse dynamics: can N-acetylcysteine stand out as a possible treatment candidate?". Acta Neuropsychiatrica 37: e31. October 2024. doi:10.1017/neu.2024.38. PMID 39415655.
- ↑ "Tobacco smoking and dopaminergic function in humans: a meta-analysis of molecular imaging studies". Psychopharmacology 236 (4): 1119–1129. April 2019. doi:10.1007/s00213-019-05196-1. PMID 30887059.
- ↑ "Effects of Smoking Cessation on Presynaptic Dopamine Function of Addicted Male Smokers". Biological Psychiatry 80 (3): 198–206. August 2016. doi:10.1016/j.biopsych.2015.11.009. PMID 26803340.
- ↑ "Nicotine produces chronic behavioral sensitization with changes in accumbal neurotransmission and increased sensitivity to re-exposure". Addiction Biology 21 (2): 397–406. March 2016. doi:10.1111/adb.12219. PMID 25581387.
- ↑ "Loss of dopamine transporters in methamphetamine abusers recovers with protracted abstinence". The Journal of Neuroscience 21 (23): 9414–9418. December 2001. doi:10.1523/JNEUROSCI.21-23-09414.2001. PMID 11717374.
- ↑ "Effect of nicotine on sympathetic nervous system activity of mice subjected to immobilization stress". Physiology & Behavior 55 (1): 53–7. January 1994. doi:10.1016/0031-9384(94)90009-4. PMID 8140174.
- ↑ Human Anatomy & Physiology (7th Ed.). Pearson. 2007. pp. ?. ISBN 978-0-8053-5909-1. https://archive.org/details/humananatomyphys00mari_4.
- ↑ Nicotine Psychopharmacology. Handbook of Experimental Pharmacology. 192. Springer. 2009. pp. 35, 37. doi:10.1007/978-3-540-69248-5. ISBN 978-3-540-69248-5.
- ↑ "Role of nicotine pharmacokinetics in nicotine addiction and nicotine replacement therapy: a review". The International Journal of Tuberculosis and Lung Disease 7 (9): 811–9. September 2003. PMID 12971663.
- ↑ "Deconvolution of Systemic Pharmacokinetics Predicts Inhaled Aerosol Dosimetry of Nicotine". European Journal of Pharmaceutical Sciences 180. January 2023. doi:10.1016/j.ejps.2022.106321. PMID 36336278.
- ↑ "Interindividual variability in the metabolism and cardiovascular effects of nicotine in man". The Journal of Pharmacology and Experimental Therapeutics 221 (2): 368–72. May 1982. doi:10.1016/S0022-3565(25)33068-5. PMID 7077531.
- ↑ "Relation of nicotine yield of cigarettes to blood nicotine concentrations in smokers". British Medical Journal 280 (6219): 972–976. April 1980. doi:10.1136/bmj.280.6219.972. PMID 7417765.
- ↑ "Detection of Cotinine in Blood Plasma by HPLC MS/MS". MIT Undergraduate Research Journal 8: 45–50. Spring 2003. http://www.docstoc.com/docs/89426297/Detection-of-Cotinine-in-Blood-Plasma-by-HPLC-MS-MS.
- ↑ "Metabolism and disposition kinetics of nicotine". Pharmacological Reviews 57 (1): 79–115. March 2005. doi:10.1124/pr.57.1.3. PMID 15734728.
- ↑ "Thirdhand smoke: heterogeneous oxidation of nicotine and secondary aerosol formation in the indoor environment". Environmental Science & Technology 45 (1): 328–33. January 2011. doi:10.1021/es102060v. PMID 21141815. Bibcode: 2011EnST...45..328P.
- ↑ "The danger of third-hand smoke: Plain language summary" (in en). The Column (Chromatography Online) 7 (3). 22 February 2011. http://www.chromatographyonline.com/danger-third-hand-smoke.
- ↑ "Mentholated cigarette smoking inhibits nicotine metabolism". The Journal of Pharmacology and Experimental Therapeutics 310 (3): 1208–15. September 2004. doi:10.1124/jpet.104.066902. PMID 15084646.
- ↑ 154.0 154.1 154.2 "Nicotine forms: why and how do they matter in nicotine delivery from electronic cigarettes?". Expert Opinion on Drug Delivery 17 (12): 1727–1736. December 2020. doi:10.1080/17425247.2020.1814736. PMID 32842785.
- ↑ "Nicotine in tobacco product aerosols: 'It's déjà vu all over again'". Tobacco Control 29 (6): 656–662. November 2020. doi:10.1136/tobaccocontrol-2019-055275. PMID 31848312.
- ↑ "Electronic Cigarettes and Youth in the United States: A Call to Action (at the Local, National and Global Levels)". Children 6 (2): 30. February 2019. doi:10.3390/children6020030. PMID 30791645.
 This article incorporates text by Jenssen BP, Boykan R available under the CC BY 4.0 license.
This article incorporates text by Jenssen BP, Boykan R available under the CC BY 4.0 license.
- ↑ "Assessing the impact of protonating acid combinations in e-cigarette liquids: a randomised, crossover study on nicotine pharmacokinetics". Scientific Reports 13 (1). June 2023. doi:10.1038/s41598-023-37539-6. PMID 37386281. Bibcode: 2023NatSR..1310563F.
- ↑ "Nicotine' actions on energy balance: Friend or foe?". Pharmacology & Therapeutics 219. March 2021.
- ↑ 159.0 159.1 159.2 159.3 "Pharmacological Effects and Regulatory Mechanisms of Tobacco Smoking Effects on Food Intake and Weight Control". Journal of Neuroimmune Pharmacology 13 (4): 453–466. December 2018. doi:10.1007/s11481-018-9800-y. PMID 30054897. "Nicotine's weight effects appear to result especially from the drug's stimulation of α3β4 nicotine acetylcholine receptors (nAChRs), which are located on pro-opiomelanocortin (POMC) neurons in the arcuate nucleus (ARC), leading to activation of the melanocortin circuit, which is associated with body weight. Further, α7- and α4β2-containing nAChRs have been implicated in weight control by nicotine.".
- ↑ "NFPA Hazard Rating Information for Common Chemicals". http://www.nmsu.edu/safety/programs/chem_safety/NFPA-ratingJ-R.htm.
- ↑ 161.0 161.1 161.2 161.3 (in en) The Merck index: an encyclopedia of chemicals, drugs, and biologicals (15th ed.). Cambridge: The Royal Society of Chemistry. 2013. pp. 1214. ISBN 978-1-84973-670-1.
- ↑ "Introduction" (in en). NIEHS Report on the Toxicity Studies of Nicotine Bitartrate Dihydrate (CASRN 6019-06-3) Administered in Drinking Water to Sprague Dawley Rats and Swiss Mice: NIEHS Report 11 [Internet] (National Institute of Environmental Health Sciences). 2024-10-01. PMID 39441944. https://www.ncbi.nlm.nih.gov/books/NBK608400/. Retrieved 2025-10-28.
- ↑ 163.0 163.1 163.2 163.3 "Insect Control", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, 2007, p. 9
- ↑ "L-Nicotine Material Safety Data Sheet". Sciencelab.com, Inc.. http://www.sciencelab.com/msds.php?msdsId=9926222.
- ↑ 165.0 165.1 165.2 The Plant Alkaloids (4th ed.). Philadelphia, Toronto: The Blakiston Company. 1949. pp. 36–43. http://library.sciencemadness.org/library/books/the_plant_alkaloids.pdf.
- ↑ "A Systematic Review of Analytical Methods for the Separation of Nicotine Enantiomers and Evaluation of Nicotine Sources". Chemical Research in Toxicology 36 (3): 334–341. March 2023. doi:10.1021/acs.chemrestox.2c00310. PMID 36897818.
- ↑ 167.0 167.1 167.2 "Enantiomeric composition of nicotine in tobacco leaf, cigarette, smokeless tobacco, and e-liquid by normal phase high-performance liquid chromatography". Chirality 30 (7): 923–931. July 2018. doi:10.1002/chir.22866. PMID 29722457.
- ↑ 168.0 168.1 168.2 168.3 168.4 "Nicotine from a Different Angle: Biological Effects from a Psychoneuroimmunological Perspective". International Journal of Molecular Sciences 26 (13): 6437. July 2025. doi:10.3390/ijms26136437. PMID 40650213.
 This article incorporates text from this source, which is available under the CC BY 4.0 license.
This article incorporates text from this source, which is available under the CC BY 4.0 license.
- ↑ "Chapter V: Analysis of various biological processes by the study of the differential action of optical isomers". Optical Activity and Living Matter. A series of monographs on general physiology. 2. Normandy, Missouri: Biodynamica. 1941. https://archive.org/stream/opticalactivityl00gauz/opticalactivityl00gauz_djvu.txt.
- ↑ "Evaluation of nicotine in tobacco-free-nicotine commercial products". Drug Testing and Analysis 9 (6): 944–948. June 2017. doi:10.1002/dta.2145. PMID 27943582.
- ↑ 171.0 171.1 "Synthetic nicotine has arrived". Tobacco Control 32 (e1): e113–e117. April 2023. doi:10.1136/tobaccocontrol-2021-056626. PMID 34493630.
- ↑ 172.0 172.1 172.2 "Synthese des Nicotins" (in de). Berichte der Deutschen Chemischen Gesellschaft 37 (2): 1225–1235. 1904. doi:10.1002/cber.19040370206. https://zenodo.org/record/1426104.
- ↑ "A New Approach to Nicotine: Symmetry Consideration for Synthesis Design". Helvetica Chimica Acta 87 (10): 2712–2716. 2004. doi:10.1002/hlca.200490241. Bibcode: 2004HChAc..87.2712H.
- ↑ "Research Progress in the Pharmacological Effects and Synthesis of Nicotine". ChemistrySelect 7 (12). 2022. doi:10.1002/slct.202104425.
- ↑ "Ornithine as a precursor for the pyrrolidine ring of nicotine". Biochimica et Biophysica Acta 33 (1): 22–6. May 1959. doi:10.1016/0006-3002(59)90492-5. PMID 13651178.
- ↑ "The Biosynthesis of Nicotine from Isotopically Labeled Nicotinic Acids". Journal of the American Chemical Society 82 (10): 2628–2633. 1960. doi:10.1021/ja01495a059. Bibcode: 1960JAChS..82.2628D.
- ↑ Plant metabolism and biotechnology. Cambridge: Wiley. 7 June 2011. ISBN 978-0-470-74703-2.
- ↑ "Nicotine Chemistry, Metabolism, Kinetics and Biomarkers". Nicotine Psychopharmacology. Handbook of Experimental Pharmacology. 192. 1 January 2009. pp. 29–60. doi:10.1007/978-3-540-69248-5_2. ISBN 978-3-540-69246-1.
- ↑ Disposition of Toxic Drugs and Chemicals in Man (10th ed.). Biomedical Publications. 2014. pp. 1452–6. ISBN 978-0-9626523-9-4.
- ↑ "Effect of transdermal nicotine administration on exercise endurance in men". Experimental Physiology 91 (4): 705–13. July 2006. doi:10.1113/expphysiol.2006.033373. PMID 16627574.
- ↑ "A comprehensive methodology for the chiral separation of 40 tobacco alkaloids and their carcinogenic E/Z-(R,S)-tobacco-specific nitrosamine metabolites". Talanta 181: 132–141. May 2018. doi:10.1016/j.talanta.2017.12.060. PMID 29426492.
- ↑ "Site-specific peak intensity ratio (SPIR) from 1D 2H/1H NMR spectra for rapid distinction between natural and synthetic nicotine and detection of possible adulteration". Analytical and Bioanalytical Chemistry 411 (24): 6427–6434. September 2019. doi:10.1007/s00216-019-02023-6. PMID 31321470.
- ↑ "Analysis and differentiation of tobacco-derived and synthetic nicotine products: Addressing an urgent regulatory issue". PLOS ONE 17 (4). 2022-04-14. doi:10.1371/journal.pone.0267049. PMID 35421170. Bibcode: 2022PLoSO..1767049C.
- ↑ 184.0 184.1 184.2 "Recent developments in the synthesis of nicotinic acetylcholine receptor ligands". Curr Top Med Chem 4 (6): 609–629. 2004. doi:10.2174/1568026043451131. PMID 14965298.
- ↑ "Nicotine analogues: a review of tobacco industry research interests". Addiction 100 (5): 701–712. May 2005. doi:10.1111/j.1360-0443.2005.01014.x. PMID 15847628.
- ↑ "Application of nicotine enantiomers, derivatives and analogues in therapy of neurodegenerative disorders". Eur J Pharmacol 563 (1–3): 18–39. June 2007. doi:10.1016/j.ejphar.2007.02.038. PMID 17376429.
- ↑ "Synthetic Methods for the Preparation of Conformationally Restricted Analogues of Nicotine". Molecules 26 (24): 7544. December 2021. doi:10.3390/molecules26247544. PMID 34946630.
- ↑ "A comparative toxicological evaluation of emerging nicotine analogs 6-methyl nicotine and nicotinamide: a scoping review". Arch Toxicol 99 (4): 1333–1340. February 2025. doi:10.1007/s00204-025-03960-1. PMID 39937258. Bibcode: 2025ArTox..99.1333E.
- ↑ "Nicotinic acetylcholine receptors: Therapeutic targets for novel ligands to treat pain and inflammation". Pharmacological Research 190. April 2023. doi:10.1016/j.phrs.2023.106715. PMID 36868367.
- ↑ "Nicotinic Acetylcholine Receptor Ligands, Cognitive Function, and Preclinical Approaches to Drug Discovery". Nicotine & Tobacco Research 21 (3): 383–394. February 2019. doi:10.1093/ntr/nty166. PMID 30137518.
- ↑ 191.0 191.1 "Cytisine derivatives as ligands for neuronal nicotine receptors and with various pharmacological activities". Farmaco 58 (3): 265–277. March 2003. doi:10.1016/S0014-827X(03)00017-X. PMID 12620422.
- ↑ "Tobacco (leaf tobacco)". Transportation Information Service. Berlin: The German Insurance Association (GDV e.V.). http://www.tis-gdv.de/tis_e/ware/genuss/tabak/tabak.htm.
- ↑ "An ecologically motivated analysis of plant-herbivore interactions in native tobacco". Plant Physiology 127 (4): 1449–1458. December 2001. doi:10.1104/pp.010762. PMID 11743088. Bibcode: 2001PlanP.127.1449B.
- ↑ "Natural history-driven, plant-mediated RNAi-based study reveals CYP6B46's role in a nicotine-mediated antipredator herbivore defense". Proceedings of the National Academy of Sciences of the United States of America 111 (4): 1245–1252. January 2014. doi:10.1073/pnas.1314848111. PMID 24379363. Bibcode: 2014PNAS..111.1245K.
- ↑ "Unpredictability of nectar nicotine promotes outcrossing by hummingbirds in Nicotiana attenuata". The Plant Journal 71 (4): 529–538. August 2012. doi:10.1111/j.1365-313X.2012.05008.x. PMID 22448647.
- ↑ 196.0 196.1 196.2 196.3 196.4 196.5 "Determination of the nicotine content of various edible nightshades (Solanaceae) and their products and estimation of the associated dietary nicotine intake". Journal of Agricultural and Food Chemistry 47 (8): 3113–20. August 1999. doi:10.1021/jf990089w. PMID 10552617. Bibcode: 1999JAFC...47.3113S.
- ↑ "The nicotine content of common vegetables". The New England Journal of Medicine 329 (6): 437. August 1993. doi:10.1056/NEJM199308053290619. PMID 8326992.
- ↑ "Nicotine Analysis in Several Non-Tobacco Plant Materials". Beiträge zur Tabakforschung International/Contributions to Tobacco Research 27 (2): 54–59. April 2016. doi:10.1515/cttr-2016-0008.
- ↑ "Nicotine psychopharmacology research contributions to United States and global tobacco regulation: a look back and a look forward". Psychopharmacology 184 (3–4): 286–91. March 2006. doi:10.1007/s00213-006-0308-4. PMID 16463054.
- ↑ "Chemische Untersuchung des Tabaks und Darstellung eines eigenthümlich wirksamen Prinzips dieser Pflanze" (in de). Magazin für Pharmacie 6 (24): 138–161. 1828. https://books.google.com/books?id=cgkCAAAAYAAJ&pg=RA1-PA138.
- ↑ "Note sur la nicotine" (in fr). Annales de Chimie et de Physique. third series 9: 465–479; see especially page 470. 1843. https://books.google.com/books?id=j-E3AAAAMAAJ&pg=PA465. [Note: The empirical formula that Melsens provides is incorrect because at that time, chemists used the wrong atomic mass for carbon (6 instead of 12).]
- ↑ "Ueber Nicotin" (in de). Berichte der Deutschen Chemischen Gesellschaft 24: 1373–1377. 1891. doi:10.1002/cber.189102401242.
- ↑ "Ueber Nicotin. Die Constitution des Alkaloïds" (in de). Berichte der Deutschen Chemischen Gesellschaft 26: 292–305. 1893. doi:10.1002/cber.18930260165. https://zenodo.org/record/1425696.
- ↑ "Ueber Nicotin. I. Mitteilung". Archiv der Pharmazie 231 (5–6): 378–448. 1893. doi:10.1002/ardp.18932310508. https://zenodo.org/record/1424557.
- ↑ "E-Cigs Are Going Tobacco-Free With Synthetic Nicotine" (in en-US). Wired. ISSN 1059-1028. https://www.wired.com/2016/06/vaping-industry-wants-go-post-tobacco-synthetic-nicotine/. Retrieved 2022-10-11.
- ↑ 206.0 206.1 Rang & Dale's Pharmacology (6th ed.). Churchill Livingstone. p. 598. ISBN 978-0-8089-2354-1.
- ↑ (in es) Primera y segunda y tercera partes de la Historia medicinal de las cosas que se traen de nuestras Indias occidentales. National Central Library of Rome. Seville: en casa de Fernando Diaz. 1580. http://archive.org/details/bub_gb_eAtm3cE1smAC.
- ↑ 208.0 208.1 208.2 (in en) Tobacco in History: The Cultures of Dependence. New York: Taylor and Francis. 2005. pp. 62-102, 127-140, 171. ISBN 978-0-415-11669-5.
- ↑ "Trends in nicotine yield in smoke and its relationship with design characteristics among popular US cigarette brands, 1997-2005". Tobacco Control 16 (5). October 2007. doi:10.1136/tc.2006.019695. PMID 17897974.
- ↑ "Industry Documents Library". https://www.industrydocuments.ucsf.edu/tobacco/docs/.
- ↑ "The Loophole That's Fueling a Return to Teenage Vaping" (in en-US). The New York Times. 2022-03-08. ISSN 0362-4331. https://www.nytimes.com/2022/03/08/health/vaping-fda-nicotine.html.
- ↑ "The Loophole That's Fueling a Return to Teenage Vaping" (in en-US). The New York Times. 2022-03-08. ISSN 0362-4331. https://www.nytimes.com/2022/03/08/health/vaping-fda-nicotine.html.
- ↑ 213.0 213.1 Center for Tobacco Products (2024-09-26). "Family Smoking Prevention and Tobacco Control Act - An Overview" (in en). FDA. https://www.fda.gov/tobacco-products/rules-regulations-and-guidance-related-tobacco-products/family-smoking-prevention-and-tobacco-control-act-overview.
- ↑ Commissioner, Office of the (2025-01-15). "FDA Proposes Significant Step Toward Reducing Nicotine to Minimally or Nonaddictive Level in Cigarettes and Certain Other Combusted Tobacco Products" (in en). https://www.fda.gov/news-events/press-announcements/fda-proposes-significant-step-toward-reducing-nicotine-minimally-or-nonaddictive-level-cigarettes.
- ↑ "AACR Expresses Support for FDA's Proposed Rule to Decrease Nicotine Content in Certain Combusted Tobacco Products" (in en). https://www.aacr.org/about-the-aacr/newsroom/news-releases/aacr-expresses-support-for-fdas-proposed-rule-to-decrease-nicotine-content-in-certain-combusted-tobacco-products/.
- ↑ "Product regulation - Public Health - European Commission" (in en). 2024-12-03. https://health.ec.europa.eu/tobacco/product-regulation_en.
- ↑ Products, Center for Tobacco (2025-08-07). "Tobacco 21" (in en). FDA. https://www.fda.gov/tobacco-products/retail-sales-tobacco-products/tobacco-21.
- ↑ "Smoking age limit to be raised to 20 as of 2025" (in en). https://eng.lsm.lv/article/society/health/11.01.2024-smoking-age-limit-to-be-raised-to-20-as-of-2025.a538525/.
- ↑ "21, 18, or 14: A look at the legal age for smoking around the world". 3 October 2017. https://www.straitstimes.com/world/21-18-or-14-a-look-at-the-legal-age-for-smoking-around-the-world.
- ↑ "The Children and Young Persons (Sale of Tobacco etc.) Order 2007" (in en). https://www.legislation.gov.uk/ukdsi/2007/9780110757384.
- ↑ "Nicotine from a Different Angle: Biological Effects from a Psychoneuroimmunological Perspective". International Journal of Molecular Sciences 26 (13): 6437. July 2025. doi:10.3390/ijms26136437. PMID 40650213.
- ↑ 222.0 222.1 "Superman versus Nick O'Teen — a children's anti-smoking campaign". Health Education Journal 44 (1): 15–18. 1 March 1985. doi:10.1177/001789698504400104.
- ↑ "Why Big Tobacco and Big Vape love comparing nicotine to caffeine" (in en). 26 April 2019. https://www.theverge.com/2019/4/26/18513312/vape-tobacco-big-companies-nicotine-caffeine-comparison-drugs-chemicals.
- ↑ "Nicotine receptors and depression: revisiting and revising the cholinergic hypothesis". Trends in Pharmacological Sciences 31 (12): 580–6. December 2010. doi:10.1016/j.tips.2010.09.004. PMID 20965579.
- ↑ "Neurobehavioral effects of selected tobacco constituents in rodents following subchronic administration". European Journal of Pharmacology 865. December 2019. doi:10.1016/j.ejphar.2019.172809. PMID 31738931.
- ↑ "Smoking, dementia and cognitive decline in the elderly, a systematic review". BMC Geriatrics 8. December 2008. doi:10.1186/1471-2318-8-36. PMID 19105840.
- ↑ "Nicotine Psychopharmacology: Policy and Regulatory". Nicotine Psychopharmacology. Handbook of Experimental Pharmacology. 192. 2009. pp. 511–34. doi:10.1007/978-3-540-69248-5_18. ISBN 978-3-540-69246-1.
- ↑ "Nicotine and Parkinson's disease: implications for therapy". Movement Disorders 23 (12): 1641–52. September 2008. doi:10.1002/mds.21900. PMID 18683238.
- ↑ "Effects of nicotine on cognitive deficits in schizophrenia". Neuropsychopharmacology 29 (7): 1378–1385. July 2004. doi:10.1038/sj.npp.1300450. PMID 15138435.
- ↑ "Altered brain functional networks in heavy smokers". Addiction Biology 20 (4): 809–819. July 2015. doi:10.1111/adb.12155. PMID 24962385.
- ↑ "Addiction-related neuroadaptations following chronic nicotine exposure". Journal of Neurochemistry 157 (5): 1652–1673. June 2021. doi:10.1111/jnc.15356. PMID 33742685.
- ↑ 232.0 232.1 "Expression and Function of the Cholinergic System in Immune Cells". Frontiers in Immunology 8. 2017. doi:10.3389/fimmu.2017.01085. PMID 28932225.
- ↑ "Effect of nicotine on IL-18-initiated immune response in human monocytes". Journal of Leukocyte Biology 80 (6): 1388–1394. December 2006. doi:10.1189/jlb.0406236. PMID 16966384.
- ↑ 234.0 234.1 234.2 "Nicotine in Inflammatory Diseases: Anti-Inflammatory and Pro-Inflammatory Effects". Frontiers in Immunology 13. 2022. doi:10.3389/fimmu.2022.826889. PMID 35251010.
- ↑ "Photoactivatable drugs for nicotinic optopharmacology". Nature Methods 15 (5): 347–350. May 2018. doi:10.1038/nmeth.4637. PMID 29578537.
- ↑ "A feasibility study with embedded pilot randomised controlled trial and process evaluation of electronic cigarettes for smoking cessation in patients with periodontitis". Pilot and Feasibility Studies 5 (1). 2019-06-04. doi:10.1186/s40814-019-0451-4. PMID 31171977.
External links
| Wikisource has the text of the 1911 Encyclopædia Britannica article Nicotine. |
- Chemical Hazards monograph for Nicotine from the National Institute for Occupational Safety and Health