Chemistry:Levocabastine
 | |
| Clinical data | |
|---|---|
| Trade names | Livostin |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | Ophthalmic, intranasal[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
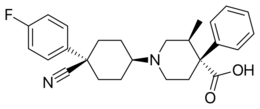
| Formula | C26H29FN2O2 |
| Molar mass | 420.528 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Levocabastine (trade name Livostin or Livocab, depending on the region) is a selective second-generation H1 receptor antagonist which was discovered at Janssen Pharmaceutica in 1979. It is used for allergic conjunctivitis.[3]
As well as acting as an antihistamine, levocabastine has also subsequently been found to act as a potent and selective antagonist for the neurotensin receptor NTS2, and was the first drug used to characterise the different neurotensin subtypes.[4][5] This has made it a useful tool for the study of this receptor.[6][7][8][9][10]
The pharmaceutical drug Bilina is a combination of Levocabastine, benzalkonium chloride, and other components and is typically used in a 0.5 mg/ml suspension as eye-drops, dispensed in 4ml bottles for the treatment of allergic conjunctivitis or similar allergic ocular conditions.[11]
References
- ↑ "Livostin Nasal Spray". RxMed: Pharmaceutical Information. http://www.rxmed.com/b.main/b2.pharmaceutical/b2.1.monographs/CPS-%20Monographs/CPS-%20(General%20Monographs-%20L)/LIVOSTIN%20NASAL%20SPRAY.html.
- ↑ "Livostin - levocabastine hydrochloride suspension". DailyMed. U.S. National Library of Medicine. http://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=420b3f66-87f0-47d8-947c-541e84eb594a.
- ↑ "A double-blind evaluation of topical levocabastine, a new specific H1 antagonist in patients with allergic conjunctivitis". Allergy 40 (7): 491–496. October 1985. doi:10.1111/j.1398-9995.1985.tb00255.x. PMID 2866725.
- ↑ "Evidence for a displaceable non-specific [3H]neurotensin binding site in rat brain". Naunyn-Schmiedeberg's Archives of Pharmacology 333 (4): 400–405. August 1986. doi:10.1007/BF00500016. PMID 3022160.
- ↑ "Two populations of neurotensin binding sites in murine brain: discrimination by the antihistamine levocabastine reveals markedly different radioautographic distribution". European Journal of Pharmacology 140 (3): 285–293. August 1987. doi:10.1016/0014-2999(87)90285-8. PMID 2888670.
- ↑ "Molecular cloning of a levocabastine-sensitive neurotensin binding site". FEBS Letters 386 (2–3): 91–94. May 1996. doi:10.1016/0014-5793(96)00397-3. PMID 8647296.
- ↑ "Structure, functional expression, and cerebral localization of the levocabastine-sensitive neurotensin/neuromedin N receptor from mouse brain". The Journal of Neuroscience 16 (18): 5613–5620. September 1996. doi:10.1523/JNEUROSCI.16-18-05613.1996. PMID 8795617.
- ↑ "Potent spinal analgesia elicited through stimulation of NTS2 neurotensin receptors". The Journal of Neuroscience 25 (36): 8188–8196. September 2005. doi:10.1523/JNEUROSCI.0810-05.2005. PMID 16148226.
- ↑ "Interactions between NTS2 neurotensin and opioid receptors on two nociceptive responses assessed on the hot plate test in mice". Behavioural Brain Research 175 (2): 399–407. December 2006. doi:10.1016/j.bbr.2006.09.016. PMID 17074405.
- ↑ "Neurotensin type 2 receptor is involved in fear memory in mice". Journal of Neurochemistry 102 (5): 1669–1676. September 2007. doi:10.1111/j.1471-4159.2007.04805.x. PMID 17697051.
- ↑ "Levocabastine ophthalmic". http://www.vademecum.es/principios-activos-levocabastina+oftalmica-s01gx02.
External links
- "Levocabastine". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/rn/79516-68-0.
- "Levocabastine hydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/levocabastine%20hydrochloride.
 |
