Chemistry:Rupatadine
 | |
| Clinical data | |
|---|---|
| Trade names | Rupafin, others |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 98–99% |
| Metabolism | Liver, CYP-mediated |
| Elimination half-life | 5.9 hours |
| Excretion | 34.6% urine, 60.9% faeces |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
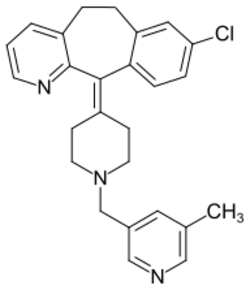
| Formula | C26H26ClN3 |
| Molar mass | 415.97 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Rupatadine is a second generation antihistamine and platelet-activating factor antagonist used to treat allergies. It was discovered and developed by Uriach[2] and is marketed as Rupafin and under several other trade names.
Medical uses
Rupatadine fumarate has been approved for the treatment of allergic rhinitis and chronic urticaria in adults and children over 12 years. It is available as round, light salmon coloured tablets containing 10 mg of rupatadine (as fumarate) to be administered orally, once a day.[3]
The efficacy of rupatadine as treatment for allergic rhinitis (AR) and chronic idiopathic urticaria (CIU) has been investigated in adults and adolescents (aged over 12 years) in several controlled studies, showing a rapid onset of action and a good safety profile even in prolonged treatment periods of a year.[4][5][6]
Side effects
Rupatadine is a non-sedating antihistamine. However, as in other non sedating second-generation antihistamines, the most common side effects in controlled clinical studies were somnolence, headaches and fatigue.
Pharmacology
Mechanism of action
Rupatadine is a second generation, non-sedating, long-acting histamine antagonist with selective peripheral H1 receptor antagonist activity. It further blocks the receptors of the platelet-activating factor (PAF) according to in vitro and in vivo studies.[7]
Rupatadine possesses anti-allergic properties such as the inhibition of the degranulation of mast cells induced by immunological and non-immunological stimuli, and inhibition of the release of cytokines, particularly of the tumor necrosis factors (TNF) in human mast cells and monocytes.[4]
Pharmacokinetics
Rupatadine has several active metabolites such as desloratadine, 3-hydroxydesloratadine,[8] 5-hydroxydesloratadine and 6-hydroxydesloratadine.[9]
History
Rupatadine discovery, pre-clinical and clinical development was performed by Uriach, a Spanish pharmaceutical company. It was launched in 2003 in Spain under the brand name of Rupafin. It was launched in Canada under the name Rupall.[10]
Brand names
Brand names include Rupafin, Rupall,[10] Rupanase, Rinialer, Pafinur, Rupax, Urtimed, Wystamm and Ralif, among others.[11]
References
- ↑ "Active Substance: rupatadine". List of the nationally authorised medicinal products. European Medicines Agency. 2 September 2021. https://www.ema.europa.eu/documents/psusa/rupatadine-list-nationally-authorised-medicinal-products-psusa/00002673/202012_en.pdf.
- ↑ Patents: EP patent 577957, US patent 5407941, US patent 5476856
- ↑ UK package leaflet for Rupafin .
- ↑ 4.0 4.1 "Rupatadine: pharmacological profile and its use in the treatment of allergic disorders". Expert Opinion on Pharmacotherapy 7 (14): 1989–2001. October 2006. doi:10.1517/14656566.7.14.1989. PMID 17020424.
- ↑ "Rupatadine: a review of its use in the management of allergic disorders". Drugs 67 (3): 457–474. 2007. doi:10.2165/00003495-200767030-00008. PMID 17335300.
- ↑ "Rupatadine in allergic rhinitis and chronic urticaria". Allergy 63 (Suppl 87): 5–28. April 2008. doi:10.1111/j.1398-9995.2008.01640.x. PMID 18339040.
- ↑ "Rupatadine, a new potent, orally active dual antagonist of histamine and platelet-activating factor (PAF)". The Journal of Pharmacology and Experimental Therapeutics 280 (1): 114–121. January 1997. PMID 8996188.
- ↑ "Influence of food on the oral bioavailability of rupatadine tablets in healthy volunteers: a single-dose, randomized, open-label, two-way crossover study". Clinical Therapeutics 29 (5): 900–908. May 2007. doi:10.1016/j.clinthera.2007.05.004. PMID 17697908.
- ↑ "Structural and clinical impact of anti-allergy agents: An overview". Bioorganic Chemistry 94: 103351. January 2020. doi:10.1016/j.bioorg.2019.103351. PMID 31668464.
- ↑ 10.0 10.1 "Pediapharm Announces the Commercial Launch of Rupall (rupatadine) in Canada". Pediapharm. January 25, 2017. http://www.pedia-pharm.com/en/press-releases/pediapharm-announces-the-commercial-launch-of-rupall-rupatadine-in-canada/.
- ↑ International Drug Names: Rupatadine.
External links
- "Rupatadine". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/rn/158876-82-5.
- "Rupatadine trihydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/rupatadine%20trihydrochloride.
 |
