Chemistry:Famotidine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /fəˈmɒtɪdiːn/ |
| Trade names | Pepcid, Zantac 360, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a687011 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 40–45% (by mouth)[1] |
| Protein binding | 15–20%[1] |
| Elimination half-life | 2.5–3.5 hours[1] |
| Excretion | Kidney (25–30% unchanged [Oral])[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| Chemical and physical data | |
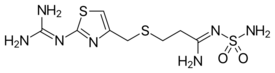
| Formula | C8H15N7O2S3 |
| Molar mass | 337.44 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Famotidine, sold under the brand name Pepcid among others, is a histamine H2 receptor antagonist medication that decreases stomach acid production.[3] It is used to treat peptic ulcer disease, gastroesophageal reflux disease, and Zollinger-Ellison syndrome.[3] It is taken by mouth or by injection into a vein.[3] It begins working within an hour.[3]
Common side effects include headache, intestinal upset, and dizziness.[3] Serious side effects may include pneumonia and seizures.[3][4] Use in pregnancy appears safe but has not been well studied, while use during breastfeeding is not recommended.[5]
Famotidine was patented in 1979 and came into medical use in 1985.[6] It is available as a generic medication.[4] In 2021, it was the 57th most commonly prescribed medication in the United States, with more than 11 million prescriptions.[7][8]
Medical uses
- Relief of heartburn, acid indigestion, and sour stomach
- Treatment for gastric and duodenal ulcers
- Treatment for pathologic gastrointestinal hypersecretory conditions such as Zollinger–Ellison syndrome and multiple endocrine adenomas
- Treatment for gastroesophageal reflux disease (GERD)
- Treatment for esophagitis
- Part of a multidrug regimen for Helicobacter pylori eradication, although omeprazole may be somewhat more effective.[9][10][11][12][13][14]
- Prevention of NSAID-induced peptic ulcers.[15][16]
- Given to surgery patients before operations to reduce the risk of aspiration pneumonitis.[17][18][19]
Pharmacokinetics
Famotidine has a delayed onset of action, beginning after 90 minutes. However, famotidine has a duration of effect of at least 540 minutes (9.0 h). At its peak effect, 210 minutes (3.5 h) after administration, famotidine reduces acid secretion by 7.3 mmol per 30 minutes.[20]
Side effects
The most common side effects associated with famotidine use include headache, dizziness, and constipation or diarrhea.[21][22]
Famotidine may contribute to QT prolongation,[23] particularly when used with other QT-elongating drugs, or in people with poor kidney function.[24]
Mechanism of action
Activation of H2 receptors located on parietal cells stimulates proton pumps to secrete acid into the stomach lumen. Famotidine, an H2 antagonist, blocks the action of histamine on the parietal cells, ultimately reducing acid secretion into the stomach.
Interactions
Unlike cimetidine, the first H2 antagonist, famotidine has a minimal effect on the cytochrome P450 enzyme system, and does not appear to interact with as many drugs as other medications in its class. Some exceptions include antiretrovirals such as atazanavir, chemotherapeutics such as doxorubicin, and antifungal medications such as itraconazole. [25][26][27]
History
Famotidine was developed by Yamanouchi Pharmaceutical Co.[28] It was licensed in the mid-1980s by Merck & Co.[29] and is marketed by a joint venture between Merck and Johnson & Johnson. The imidazole ring of cimetidine was replaced with a 2-guanidinothiazole ring. Famotidine proved to be nine times more potent than ranitidine, and thirty-two times more potent than cimetidine.[30]
It was first marketed in 1981. Pepcid RPD orally disintegrating tablets were released in 1999. Generic preparations became available in 2001, e.g. Fluxid (Schwarz) or Quamatel (Gedeon Richter Ltd.).
In the United States and Canada, a product called Pepcid Complete, which combines famotidine with an antacid in a chewable tablet to quickly relieve the symptoms of excess stomach acid, is available. In the UK, this product was known as PepcidTwo until its discontinuation in April 2015.[31]
Famotidine has poor bioavailibility (50%) due to low gastroretention time. Famotidine is less soluble at higher pH, and when used in combination with antacids gastroretention time is increased. This promotes local delivery of these drugs to receptors in the parietal cell membrane and increases bioavailibility. Researchers are developing tablet formulations that rely on other gastroretentive drug delivery systems such as floating tablets to further increase bioavailibility.[32]
Society and culture
Certain preparations of famotidine are available over the counter (OTC) in various countries. In the United States and Canada, 10 mg and 20 mg tablets, sometimes in combination with an antacid,[33][34] are available OTC. Larger doses still require a prescription.
Formulations of famotidine in combination with ibuprofen were marketed by Horizon Pharma under the trade name Duexis.[35]
Research
COVID-19
At the start of the COVID-19 pandemic, some doctors observed that anecdotally some hospitalized patients in China may have had better outcomes on famotidine than other patients that were not taking famotidine. This led to hypotheses about use of famotidine in treatment of COVID-19.[36][37] Famotidine was considered a possible treatment for COVID-19 due to its potential anti-inflammatory effects. It was thought that famotidine could modify lung inflammation caused by coronaviruses. However, studies have shown that famotidine is not effective in reducing mortality or improving recovery in COVID-19 patients.[38] Famotidine primarily works by blocking the effects of histamine and has some potential mechanisms of action that may contribute to its anti-inflammatory properties, including the inhibition of the production of certain pro-inflammatory cytokines such as TNF-alpha and IL-6.[39][40] Another hypothesis was that famotidine might activate the vagus nerve inflammatory reflex to attenuate cytokine storm.[40] Yet another hypothesis was that famotidine can reduce the activation of mast cells and the subsequent release of inflammatory mediators, therefore acting as a mast cell stabilizer.[41][39] However, while famotidine may have some anti-inflammatory effects, there is currently insufficient evidence to support its use for treating inflammation associated with COVID-19.[38] Therefore, it is not recommended for this purpose.[42]
Other
Small-scale studies have shown inconsistent and inconclusive evidence of efficacy in treatment-refractory schizophrenia.[43]
Veterinary uses
Famotidine is given to dogs and cats with acid reflux.[44]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 "Famotidine tablet". https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=f149ecc1-d66c-42f9-a3f4-2ab6a522942b.
- ↑ "Zantac 360- famotidine tablet, film coated". 17 May 2022. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=89f60eb1-359b-40e8-ae8b-65aec9cd5b1f.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 "Famotidine Monograph for Professionals" (in en). American Society of Health-System Pharmacists. https://www.drugs.com/monograph/famotidine.html.
- ↑ 4.0 4.1 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 74–75. ISBN 9780857113382.
- ↑ "Famotidine Use During Pregnancy" (in en). https://www.drugs.com/pregnancy/famotidine.html.
- ↑ (in en) Analogue-based Drug Discovery. John Wiley & Sons. 2006. p. 444. ISBN 9783527607495. https://books.google.com/books?id=FjKfqkaKkAAC&pg=PA444. Retrieved 7 May 2020.
- ↑ "The Top 300 of 2021". https://clincalc.com/DrugStats/Top300Drugs.aspx.
- ↑ "Famotidine - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Famotidine.
- ↑ "[Proton-pump inhibitors versus H2-receptor antagonists in triple therapy for Helicobacter pylori eradication]". Nihon Rinsho. Japanese Journal of Clinical Medicine 57 (1): 153–6. January 1999. PMID 10036954.
- ↑ "Is a proton pump inhibitor necessary for the treatment of lower-grade reflux esophagitis?". Journal of Gastroenterology 34 (4): 435–40. August 1999. doi:10.1007/s005350050292. PMID 10452673.
- ↑ "Omeprazole enhances efficacy of triple therapy in eradicating Helicobacter pylori". Gut 37 (4): 477–81. October 1995. doi:10.1136/gut.37.4.477. PMID 7489931.
- ↑ "Comparison of H2-receptor antagonist- and proton-pump inhibitor-based triple regimens for the eradication of Helicobacter pylori in Chinese patients with gastritis or peptic ulcer". The Journal of International Medical Research 31 (6): 469–74. 2003. doi:10.1177/147323000303100601. PMID 14708410.
- ↑ "[Efficacy of different schemes of anti-helicobacter therapy in duodenal ulcer]". Terapevticheskii Arkhiv 76 (2): 18–22. 2004. PMID 15106408.
- ↑ "Famotidine vs. omeprazole: a prospective randomized multicentre trial to determine efficacy in non-erosive gastro-oesophageal reflux disease". Alimentary Pharmacology & Therapeutics 21 (Suppl 2): 10–8. June 2005. doi:10.1111/j.1365-2036.2005.02468.x. PMID 15943841.
- ↑ "Prophylaxis and treatment of NSAID-induced gastroduodenal disorders". Drug Safety 20 (6): 527–43. June 1999. doi:10.2165/00002018-199920060-00006. PMID 10392669.
- ↑ "Double-blind randomized trials of single-tablet ibuprofen/high-dose famotidine vs. ibuprofen alone for reduction of gastric and duodenal ulcers". The American Journal of Gastroenterology 107 (3): 379–86. March 2012. doi:10.1038/ajg.2011.443. PMID 22186979.
- ↑ "Effects of omeprazole, ranitidine, famotidine and placebo on gastric secretion in patients undergoing elective surgery". British Journal of Anaesthesia 69 (4): 404–6. October 1992. doi:10.1093/bja/69.4.404. PMID 1419452.
- ↑ "Acid aspiration prophylaxis in morbidly obese patients: famotidine vs. ranitidine". Anaesthesia 46 (11): 967–9. November 1991. doi:10.1111/j.1365-2044.1991.tb09860.x. PMID 1750602.
- ↑ "Effects of famotidine on gastric pH and residual volume in pediatric surgery". Acta Anaesthesiologica Scandinavica 35 (5): 457–60. July 1991. doi:10.1111/j.1399-6576.1991.tb03328.x. PMID 1887750.
- ↑ "Comparison of the effects of over-the-counter famotidine and calcium carbonate antacid on postprandial gastric acid. A randomized controlled trial". JAMA 275 (18): 1428–1431. May 1996. doi:10.1001/jama.1996.03530420056036. PMID 8618369.
- ↑ "Common Side Effects of Pepcid (Famotidine) Drug Center" (in en). https://www.rxlist.com/pepcid-side-effects-drug-center.htm.
- ↑ "Drugs & Medications" (in en). https://www.webmd.com/drugs/2/drug-5035-2033/famotidine-oral/famotidine-oral/details.
- ↑ "Drugs to be avoided in patients with long QT syndrome: Focus on the anaesthesiological management.". World Journal of Cardiology 5 (4): 87–93. 26 April 2013. doi:10.4330/wjc.v5.i4.87. PMID 23675554.
- ↑ "Famotidine and long QT syndrome". The American Journal of Cardiology 93 (10): 1325–1327. May 2004. doi:10.1016/j.amjcard.2004.02.025. PMID 15135720.
- ↑ "Effects of the H2-receptor antagonist famotidine on the pharmacokinetics of atazanavir-ritonavir with or without tenofovir in HIV-infected patients". AIDS Patient Care and STDs 25 (9): 509–515. September 2011. doi:10.1089/apc.2011.0113. PMID 21770762.
- ↑ "Comparative randomized trial evaluating the effect of proton pump inhibitor versus histamine 2 receptor antagonist as an adjuvant therapy in diffuse large B-cell lymphoma". Medical Oncology 38 (1): 4. January 2021. doi:10.1007/s12032-020-01452-z. PMID 33394214.
- ↑ "Short report: the absorption of fluconazole and itraconazole under conditions of low intragastric acidity". Alimentary Pharmacology & Therapeutics 7 (3): 317–321. June 1993. doi:10.1111/j.1365-2036.1993.tb00103.x. PMID 8117350.
- ↑ US patent 4283408, Yasufumi Hirata, Isao Yanagisawa, Yoshio Ishii, Shinichi Tsukamoto, Noriki Ito, Yasuo Isomura and Masaaki Takeda, "Guanidinothiazole compounds, process for preparation and gastric inhibiting compositions containing them", issued 11 August 1981
- ↑ "Sankyo Pharma". Skyscape Mediwire. 2002. http://mediwire.skyscape.com/main/Default.aspx?P=Content&ArticleID=23882.
- ↑ "Famotidine, a new, potent, long-acting histamine H2-receptor antagonist: comparison with cimetidine and ranitidine in the treatment of Zollinger-Ellison syndrome". Gastroenterology 88 (4): 1026–33. April 1985. doi:10.1016/s0016-5085(85)80024-x. PMID 2857672.
- ↑ "PepcidTwo Chewable Tablet". http://www.medicines.org.uk/emc/medicine/20012.
- ↑ "Formulation and Evaluation of Gastroretentive Floating Tablets of Famotidine". Farmavita.Net. 2008. http://www.farmavita.net/content/view/1012/84/.
- ↑ Pepcid Complete
- ↑ "Famotidine". Medline Plus. https://www.nlm.nih.gov/medlineplus/druginfo/meds/a687011.html.
- ↑ "Duexis". https://www.drugs.com/duexis.html.
- ↑ "New York clinical trial quietly tests heartburn remedy against coronavirus". Science Magazine. 26 April 2020. https://www.science.org/content/article/new-york-clinical-trial-quietly-tests-heartburn-remedy-against-coronavirus.
- ↑ "COVID-19: Famotidine, Histamine, Mast Cells, and Mechanisms". Frontiers in Pharmacology 12: 633680. 2021. doi:10.3389/fphar.2021.633680. PMID 33833683.
- ↑ 38.0 38.1 "No evidence of clinical efficacy of famotidine for the treatment of COVID-19: a systematic review and meta-analysis". J Infect 86 (2): 154–225. February 2023. doi:10.1016/j.jinf.2022.11.022. PMID 36462586.
- ↑ 39.0 39.1 "Famotidine: A potential mitigator of mast cell activation in post-COVID-19 cognitive impairment". J Psychosom Res 172: 111425. September 2023. doi:10.1016/j.jpsychores.2023.111425. PMID 37399740.
- ↑ 40.0 40.1 "Famotidine activates the vagus nerve inflammatory reflex to attenuate cytokine storm". Mol Med 28 (1): 57. May 2022. doi:10.1186/s10020-022-00483-8. PMID 35578169.
- ↑ "Antihistamines improve cardiovascular manifestations and other symptoms of long-COVID attributed to mast cell activation". Front Cardiovasc Med 10: 1202696. 2023. doi:10.3389/fcvm.2023.1202696. PMID 37529714.
- ↑ "Clinical update on COVID-19 for the emergency and critical care clinician: Medical management". The American Journal of Emergency Medicine 56: 158–170. June 2022. doi:10.1016/j.ajem.2022.03.036. PMID 35397357.
- ↑ Andrade, Chittaranjan (2013). "Famotidine Augmentation in Schizophrenia: Hope or Hype?". The Journal of Clinical Psychiatry 74 (9): e855–e858. doi:10.4088/JCP.13f08707. PMID 24107771. https://www.psychiatrist.com/jcp/famotidine-augmentation-schizophrenia-hope-hype/. Retrieved 11 January 2024.
- ↑ "Famotidine". http://www.petmd.com/pet-medication/famotidine.
External links
 |
