Chemistry:A-84,543
From HandWiki
Short description: Chemical compound
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C11H16N2O |
| Molar mass | 192.257 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
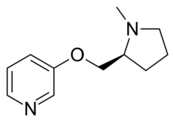
A-84543 is a drug developed by Abbott, which acts as an agonist at neural nicotinic acetylcholine receptors with high selectivity for the α4β2 subtype. It is widely used in scientific research into the structure and function of this receptor subtype and has been the lead compound for the development of a large family of related derivatives.[1][2][3][4]
References
- ↑ "Novel 3-Pyridyl ethers with subnanomolar affinity for central neuronal nicotinic acetylcholine receptors". Journal of Medicinal Chemistry 39 (4): 817–25. February 1996. doi:10.1021/jm9506884. PMID 8632405.
- ↑ "Synthesis and structure-activity relationships of pyridine-modified analogs of 3-[2-((S)-pyrrolidinyl)methoxy]pyridine, A-84543, a potent nicotinic acetylcholine receptor agonist". Bioorganic & Medicinal Chemistry Letters 8 (3): 249–54. February 1998. doi:10.1016/S0960-894X(98)00019-5. PMID 9871663.
- ↑ "Novel pyridyl ring C5 substituted analogues of epibatidine and 3-(1-methyl-2(S)-pyrrolidinylmethoxy)pyridine (A-84543) as highly selective agents for neuronal nicotinic acetylcholine receptors containing beta2 subunits". Journal of Medicinal Chemistry 48 (6): 1721–4. March 2005. doi:10.1021/jm0492406. PMID 15771418.
- ↑ "Synthesis of azabicyclo[2.2.n]alkane systems as analogues of 3-[1-methyl-2-(S)-pyrrolidinyl- methoxy]pyridine (A-84543)". The Journal of Organic Chemistry 72 (8): 3112–5. April 2007. doi:10.1021/jo0700732. PMID 17371077.
 |
