Chemistry:Esketamine
Esketamine, sold under the brand names Spravato (for depression) and Ketanest (for anesthesia) among others,[1][2] is the S(+) enantiomer of ketamine.[3][4] It is a dissociative medication used as a general anesthetic and as an antidepressant. Esketamine is the active enantiomer of ketamine in terms of NMDA receptor antagonism and is more potent than racemic ketamine.[5] However, racemic ketamine produces larger and more sustained antidepressant effects than esketamine.[6]
As an anesthetic, esketamine is indicated for high-risk patients or as a supplement to incomplete regional anesthesia. As an antidepressant, it is specifically used as both a monotherapy and combination therapy for treatment-resistant depression (TRD) as well as major depressive disorder (MDD) with co-occurring suicidal ideation or behavior.[1][7] Its efficacy as combination therapy for TRD is modest and similar to that of atypical antipsychotics; evidence for its efficacy as a monotherapy is very limited.[8][9] Antisuicidal efficacy remains unproven.[7] Esketamine is not used by infusion into a vein for depression as it is only FDA-approved in the form of a nasal spray under direct medical supervision for this indication (the parent compound ketamine is most often administered intravenously).[1][3]
Adverse effects of esketamine include dissociation, dizziness, sedation, nausea, vomiting, vertigo, numbness, anxiety, lethargy, increased blood pressure, and feelings of drunkenness.[1] Less often, esketamine can cause bladder problems.[1][10] Esketamine acts primarily as a NMDA receptor antagonist.[3][4] Esketamine has faster clearance and stronger dopamine inhibition than arketamine, contributing to its dissociative and psychotomimetic effects.
In the form of racemic ketamine, esketamine was first synthesized in 1962 and introduced for medical use as an anesthetic in 1970.[11] Enantiopure esketamine was introduced for medical use as an anesthetic in 1997 and as an antidepressant in 2019.[3][1][12] It is used as an anesthetic in the European Union and as an antidepressant in the United States and Canada.[12][13][14] Due to misuse liability as a dissociative, esketamine is a controlled substance.[11][1]
Medical uses
Anesthesia
Esketamine is used for similar indications as ketamine.[3] Such uses include induction of anesthesia in high-risk patients such as those with circulatory shock, severe bronchospasm, or as a supplement to regional anesthesia with incomplete nerve blocks.[3]
Depression
In the US, esketamine (brand name Spravato) is a nasal spray indicated, as monotherapy, or in conjunction with an oral antidepressant as a therapy for treatment-resistant depression (TRD) as well as major depressive disorder (MDD) associated with suicidal ideation or behavior in adults.[1] In the clinical trials that led to approval of esketamine, treatment-resistant depression was defined as major depressive disorder with inadequate response to at least two different conventional antidepressants.[1] The nasal spray formulation of esketamine used for depression delivers two sprays containing a total of 28 mg esketamine and doses of 56 mg (2 devices) to 84 mg (3 devices) are used.[1]
Esketamine has modest short-term efficacy on TRD (similar in efficacy to atypical antipsychotics); there is limited long-term safety data available and some concerning signals regarding adverse events and abuse potential.[8] Due to concerns about sedation, dissociation, and misuse, esketamine is available for treatment of depression only from certified providers through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called Spravato REMS.[1]
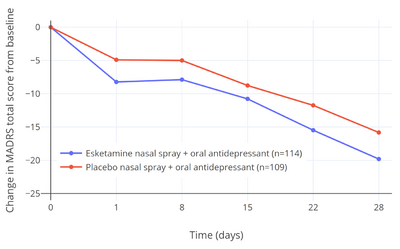

Expectations were initially very high for ketamine and esketamine for treatment of depression based on early small-scale clinical studies, with discovery of the rapid and ostensibly robust antidepressant effects of ketamine described by some authors as "the most important advance in the field of psychiatry in the past half century".[20][21][22] According to a 2018 review, ketamine showed more than double the antidepressant effect size over placebo of conventional antidepressants in the treatment of depression based on the preliminary evidence available at the time (Cohen's d = 1.3–1.7 for ketamine, Cohen's d = 0.8 for midazolam (active placebo), and Cohen's d = 0.53–0.81 for conventional antidepressants).[20] However, the efficacy of ketamine/esketamine for depression declined dramatically as studies became larger and more methodologically rigorous.[23][24]
In February 2019, an outside panel of experts recommended in a 14–2 vote that the FDA approve the nasal spray version of esketamine for treatment-resistant depression, provided that it be given in a clinical setting, with people remaining on site for at least two hours after.[25][26] The reasoning for this requirement is that trial participants temporarily experienced sedation, visual disturbances, trouble speaking, confusion, numbness, and feelings of dizziness immediately after.[27] The approval of esketamine for treatment-resistant depression by the FDA was controversial due to limited and mixed evidence of efficacy and safety.[26][17][16][28]
In January 2020, esketamine was rejected by the National Health Service (NHS) of Great Britain.[29] The NHS questioned the benefits of the medication for depression and claimed that it was too expensive.[29] People already using esketamine were allowed to complete treatment if their doctors considered this necessary.[29]
Esketamine is approved in the United Stares for and shows promise as a rapid-acting monotherapy for treatment-resistant depression, but evidence is currently limited to a single trial.[9] It is an effective and generally safe long-term treatment for adults with treatment-resistant depression, feasible in outpatient settings; optimal oral antidepressant combinations and predictive biomarkers need further research.[30]
Spravato debuted at a cost of treatment of US$32,400 per year when it launched in the United States in March 2019.[31] The Institute for Clinical and Economic Review (ICER), which evaluates cost effectiveness of drugs analogously to the National Institute for Health and Care Excellence (NICE) in the United Kingdom, declined to recommend esketamine for depression due to its steep cost and modest efficacy, deeming it not sufficiently cost-effective.[31][32]
Esketamine is the second drug to be approved for treatment-resistant depression by the FDA, following olanzapine/fluoxetine (Symbyax) in 2009.[28][33] Other agents, like the atypical antipsychotics aripiprazole (Abilify) and quetiapine (Seroquel), have been approved for use in the adjunctive therapy of major depressive disorder in people with a partial response to treatment.[28]
In a meta-analysis conducted internally by the FDA during its evaluation of esketamine for treatment-resistant depression, the FDA reported a standardized mean difference (SMD) of esketamine for treatment-resistant depression of 0.28 using the three phase III short-term efficacy trials conducted by Janssen.[28] This was similar to an SMD of 0.26 for olanzapine/fluoxetine for treatment-resistant depression and lower than SMDs of 0.35 for aripiprazole and 0.40 for quetiapine as adjuncts for major depressive disorder.[28] These drugs are less expensive than esketamine and may serve as more affordable alternatives to it for depression with similar effectiveness.[28] Both rTMS and intranasal esketamine are more effective than starting a new antidepressant for treatment-resistant depression, with rTMS potentially offering slightly greater or comparable symptom reduction compared to esketamine.[34]
Racemic ketamine produces larger and more sustained antidepressant effects than esketamine, with higher doses generally more effective; both ketamine and esketamine have similar dropout rates.[6]
Preliminary research suggests that arketamine, the R(−) enantiomer of ketamine, may also have its own independent antidepressant effects and may contribute to the antidepressant efficacy of racemic ketamine, but more research likewise is needed to evaluate this possibility.[35][36]
Adverse effects
The most common adverse effects of esketamine for depression (≥5% incidence) include dissociation, dizziness, sedation, nausea, vomiting, vertigo, numbness, anxiety, lethargy, increased blood pressure, and feelings of drunkenness.[1] Long-term abuse of ketamine has been associated with bladder disease.[1][10]
Pharmacology
Pharmacodynamics
Esketamine is approximately twice as potent an anesthetic as racemic ketamine.[37]
The possibility that arketamine may be more effective than esketamine has been suggested by some researchers.[38][39][40]
Esketamine inhibits dopamine transporters eight times more than arketamine.[41] This increases dopamine activity in the brain. At doses causing the same intensity of effects, esketamine is generally considered to be more pleasant by patients.[42][43] Patients also generally recover mental function more quickly after being treated with pure esketamine, which may be a result of the fact that it is cleared from their system more quickly.[37][44] However, this observation contrasts with findings that arketamine exhibits antidepressant effects without causing psychotomimetic side effects.[45]
Unlike arketamine, esketamine does not bind significantly to sigma receptors. Esketamine increases glucose metabolism in the frontal cortex, while arketamine decreases glucose metabolism in the brain. This difference may be responsible for the fact that esketamine generally has a more dissociative or hallucinogenic effect while arketamine is reportedly more relaxing.[44] However, another study found no difference between racemic ketamine and esketamine on the patient's level of vigilance.[42] Interpretation of this finding is complicated by the fact that racemic ketamine is 50% esketamine.[46]
Pharmacokinetics
Esketamine is eliminated from the human body more quickly than arketamine (R(–)-ketamine) or racemic ketamine, although arketamine slows the elimination of esketamine.[47] The half-life of esketamine was found to be approximately 5 hours.[48] When administered intranasally, esketamine's bioavailability is approximately 30–50%.[48]
Chemistry
Esketamine is the (S)-enantiomer of ketamine, which is a racemic mixture of esketamine and arketamine ((R)-ketamine).[49]
History
Esketamine was introduced for medical use as an anesthetic in Germany in 1997, and was subsequently marketed in other countries.[3][13] In addition to its anesthetic effects, the medication showed properties of being a rapid-acting antidepressant, and was subsequently investigated for use as such.[50][51] Esketamine received a breakthrough designation from the FDA for treatment-resistant depression (TRD) in 2013 and major depressive disorder (MDD) with accompanying suicidal ideation in 2016.[51][52] In November 2017, it completed phase III clinical trials for treatment-resistant depression in the United States.[50][51] Johnson & Johnson filed a Food and Drug Administration (FDA) New Drug Application (NDA) for approval on 4 September 2018;[53] the application was endorsed by an FDA advisory panel on 12 February 2019, and on 5 March 2019, the FDA approved esketamine, in conjunction with an oral antidepressant, for the treatment of depression in adults.[12] In August 2020, it was approved by the U.S. Food and Drug Administration (FDA) with the added indication for the short-term treatment of suicidal thoughts.[54]
Since the 1980s, closely associated ketamine has been used as a club drug also known as "Special K" for its trip-inducing side effects.[55][56]
Society and culture

Names
Esketamine is the generic name of the drug and its INN and BAN, while esketamine hydrochloride is its BANM.[13] It is also known as S(+)-ketamine, (S)-ketamine, or (–)-ketamine ((-)[+] ketamine), as well as by its developmental code name JNJ-54135419.[13][51]
Esketamine is sold under the brand name Spravato for use as an antidepressant and the brand names Eskesia, Ketanest, Ketanest S, Ketanest-S, Keta-S for use as an anesthetic (veterinary), among others.[13]
Controversy
The British critical psychiatrist Joanna Moncrieff has critiqued the use and study of esketamine and ketamine as well as related drugs like psychedelics for treatment of psychiatric disorders, highlighting concerns including excessive hype around these drugs, questionable biologically based theories of benefit, blurred lines between medical and recreational use, flawed clinical trial findings, financial conflicts of interest, strong expectancy effects and large placebo responses, small and short-term benefits over placebo, and their potential for difficult experiences and adverse effects, among others.[57]
Legal status
Esketamine is a Schedule III controlled substance in the United States.[1]
Esketamine is a controlled drug In The United Arab Emirates due to its potential for abuse, its use is only under strict medical supervision which is only available on government hospitals in the country, and its use only approved for treatment-resistant depression registered under the trademark Spravato.[58]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 "Spravato- esketamine hydrochloride solution". 6 August 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=d81a6a79-a74a-44b7-822c-0dfa3036eaed.
- ↑ "Esketamin - Anwendung, Wirkung, Nebenwirkungen | Gelbe Liste" (in de). https://www.gelbe-liste.de/wirkstoffe/Esketamin_45078.
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 "[The clinical use of S-(+)-ketamine--a determination of its place]" (in de). Anästhesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie 33 (12): 764–70. December 1998. doi:10.1055/s-2007-994851. PMID 9893910.
- ↑ 4.0 4.1 "Ketamine: A tale of two enantiomers". J Psychopharmacol 35 (2): 109–123. February 2021. doi:10.1177/0269881120959644. PMID 33155503.
- ↑ "Ketamine: teaching an old drug new tricks". Anesthesia and Analgesia 87 (5): 1186–1193. November 1998. doi:10.1213/00000539-199811000-00039. PMID 9806706.
- ↑ 6.0 6.1 Nikolin, Stevan; Rodgers, Anthony; Schwaab, Andreas; Bahji, Anees; Zarate, Carlos; Vazquez, Gustavo; Loo, Colleen (2023-08-01). "Ketamine for the treatment of major depression: a systematic review and meta-analysis" (in English). eClinicalMedicine 62. doi:10.1016/j.eclinm.2023.102127. ISSN 2589-5370. PMID 37593223.
- ↑ 7.0 7.1 "Synthesizing the Evidence for Ketamine and Esketamine in Treatment-Resistant Depression: An International Expert Opinion on the Available Evidence and Implementation". Am J Psychiatry 178 (5): 383–399. May 2021. doi:10.1176/appi.ajp.2020.20081251. PMID 33726522. "A legitimate criticism, as it relates to interpreting the effect sizes reported with single or repeat-dose ketamine in treatment-resistant depression, is the possibility that nonspecific effects such as functional unblinding (e.g., by patients experiencing dissociation or euphoric responses) and expectancymayinadvertentlyinflate the efficacy of ketamine (51, 52). [...] Given the absence of an adequately designed head-to-head trial, the relative efficacies of intranasal esketamine and intravenous racemic ketamine are not known (65). [...] A recent meta-analysis comparing intranasal and intravenous ketamine formulations was unable to identify a significant difference between formulations as well as routes of delivery in efficacy at 24 hours, 7 days, and 28 days (17). A separate meta-analysis concluded that intravenous ketamine may be superior in efficacy and have lower dropout rates (66). However, it is difficult to draw definitive conclusions from these analyses given the heterogeneity across component studies.".
- ↑ 8.0 8.1 Fountoulakis, Konstantinos N.; Saitis, Athanasios; Schatzberg, Alan F. (2025). "Esketamine Treatment for Depression in Adults: A PRISMA Systematic Review and Meta-Analysis". American Journal of Psychiatry 182 (3): 259–275. doi:10.1176/appi.ajp.20240515. https://psychiatryonline.org/doi/abs/10.1176/appi.ajp.20240515.
- ↑ 9.0 9.1 Bhavya, null; Shetty, Srijan; Sadasivam, Balakrishnan (2025). "A New Era for Esketamine in Managing Treatment-Resistant Depression: A Systematic Review of Its Use From Adjunct to First-Line Therapy". Cureus 17 (9). doi:10.7759/cureus.91829. ISSN 2168-8184. PMID 41185718.
- ↑ 10.0 10.1 "Ketamine-induced urological toxicity: potential mechanisms and translation for adults with mood disorders receiving ketamine treatment". Psychopharmacology (Berl) 238 (4): 917–926. April 2021. doi:10.1007/s00213-021-05767-1. PMID 33484298.
- ↑ 11.0 11.1 "Classics in Chemical Neuroscience: Ketamine". ACS Chem Neurosci 8 (6): 1122–1134. June 2017. doi:10.1021/acschemneuro.7b00074. PMID 28418641.
- ↑ 12.0 12.1 12.2 "FDA approves new nasal spray medication for treatment-resistant depression; available only at a certified doctor's office or clinic". U.S. Food and Drug Administration (FDA) (Press release). Archived from the original on 23 July 2021. Retrieved 6 March 2019.
- ↑ 13.0 13.1 13.2 13.3 13.4 "Esketamine". Drugs.com. 25 October 2022. https://www.drugs.com/international/esketamine.html.
- ↑ "The Canadian Network for Mood and Anxiety Treatments (CANMAT) Task Force Recommendations for the Use of Racemic Ketamine in Adults with Major Depressive Disorder: Recommandations Du Groupe De Travail Du Réseau Canadien Pour Les Traitements De L'humeur Et De L'anxiété (Canmat) Concernant L'utilisation De La Kétamine Racémique Chez Les Adultes Souffrant De Trouble Dépressif Majeur". Can J Psychiatry 66 (2): 113–125. November 2020. doi:10.1177/0706743720970860. PMID 33174760.
- ↑ "SPRAVATO™ Clinical Studies | Touchstone TMS". 13 January 2020. https://touchstonetms.com/spravato-clinical-studies/.
- ↑ 16.0 16.1 "Are we repeating mistakes of the past? A review of the evidence for esketamine". Br J Psychiatry 219 (5): 614–617. May 2020. doi:10.1192/bjp.2020.89. PMID 32456714.
- ↑ 17.0 17.1 "Esketamine for treatment resistant depression: a trick of smoke and mirrors?". Epidemiol Psychiatr Sci 29. December 2019. doi:10.1017/S2045796019000751. PMID 31841104.
- ↑ "Efficacy and Safety of Intranasal Esketamine in Treatment-Resistant Depression in Adults: A Systematic Review". Cureus 13 (8). August 2021. doi:10.7759/cureus.17352. PMID 34447651.
- ↑ 19.0 19.1 "Esketamine Nasal Spray for the Rapid Reduction of Depressive Symptoms in Major Depressive Disorder With Acute Suicidal Ideation or Behavior". J Clin Psychopharmacol 41 (5): 516–524. 2021. doi:10.1097/JCP.0000000000001465. PMID 34412104.
- ↑ 20.0 20.1 "Antidepressant Efficacy and Tolerability of Ketamine and Esketamine: A Critical Review". CNS Drugs 32 (5): 411–420. May 2018. doi:10.1007/s40263-018-0519-3. PMID 29736744. "In brief, these studies (Table 1) have globally assessed responses to a single dose of intravenous ketamine in 166 patients with TDR with multiple treatment failures, including electroconvulsive therapy (ECT). The findings provide evidence of improvement in depressive symptoms within hours, with a response rate > 60% in the first 4.5 and 24 h, and > 40% after 7 days, with a big effect size in comparison with placebo (Cohen's d 1.3–1.7) or active placebo (midazolam, d = 0.8). These figures, though preliminary, contrast with the average effect size of conventional antidepressants (Cohen's d 0.53–0.81 in patients with intense symptoms) [32] and their response latency (about 4–7 weeks) [1].".
- ↑ "Esketamine/ketamine for treatment-resistant depression". Braz J Psychiatry 42 (6): 579–580. 2020. doi:10.1590/1516-4446-2020-0996. PMID 32401866. "Some authors have described the discovery of rapid and robust antidepressant effects of the N-methyl-D-aspartate (NMDA) receptor antagonist ketamine as the most important advance in the field of psychiatry in the past half century.".
- ↑ "Ketamine treatment for depression: opportunities for clinical innovation and ethical foresight". Lancet Psychiatry 4 (5): 419–426. May 2017. doi:10.1016/S2215-0366(17)30102-5. PMID 28395988. https://discovery.ucl.ac.uk/id/eprint/1552865/1/Singh_Ketamine_treatment_depression_AAM.pdf. "Ketamine has been hailed as the most important advance in the treatment of depression of the past 50 years.1".
- ↑ "Consistently Modest Antidepressant Effects in Clinical Trials: the Role of Regulatory Requirements". Psychopharmacol Bull 51 (3): 79–108. June 2021. doi:10.64719/pb.4412. PMID 34421147. "Even drugs with novel mechanisms of action such as the esketamine nasal spray show the same effect size and look nearly identical to other antidepressants when evaluated in the regulatory context (42% symptom reduction with placebo, 54% with drug, effect size 0.29). However, it must be taken under consideration that this trial was unique from the others in that it was an adjunctive study of esketamine nasal spray in treatment resistant patients. It is worth noting that two shortterm trials conducted for regulatory approval of esketamine but not included in the label did not reach statistical significance (P = 0.058 and P = 0.088).28 Independent analysis of these esketamine trial data submitted to the FDA show that despite expectations from smallscale preliminary studies, esketamine performs modestly in patients with treatment resistant depression in the context of large, regulatory trials.29 These authors also raised concerns about the potential lack of specificity of drug effects and the risk of side effects demonstrated in these trials. [...] False negatives are well-known risks of small sized studies. However, it is equally important to note that if we do not enroll adequate sample sizes we will continue run the serious risk of getting an inflated false positive resulting in an overestimate of treatment effects that is not replicable (as was the case with many of the earlier regulatory trials, which tended to have small sample sizes).25 This is especially pertinent for early pilot studies of investigational antidepressants (phase I and II trials), which are not always subject to the same regulatory statutes of later stage trials. This phenomenon is illustrated by the dramatic decline of treatment effect sizes seen with esketamine over the course of development (from small pilot studies to large regulatory trials). Although regulatory agencies allow for more lenient methods for exploratory purposes, this method may yield misleading conclusions because these small trials are invariably under-powered. Specifically, these exploratory trials may end up with an erroneously low placebo response and thus a falsely inflated estimate of effect size.46 This possibility is under appreciated by many investigators but should be strongly considered given the persistence of modest effect sizes in regulatory trials of antidepressants.".
- ↑ "Safety and effectiveness of NMDA receptor antagonists for depression: A multidisciplinary review". Pharmacotherapy 42 (7): 567–579. July 2022. doi:10.1002/phar.2707. PMID 35665948. "The promising results seen in the small, single-infusion, single-center trials of racemic ketamine were generally not replicated in the larger, multi-site trials of esketamine nasal spray. The esketamine trials were also subject to FDA site inspections, data integrity checks, and other forms of independent scrutiny.".
- ↑ "First Big Depression Advance Since Prozac Nears FDA Approval.". Bloomberg News. 12 February 2019. https://www.bloomberg.com/news/articles/2019-02-12/first-big-depression-advance-since-prozac-nears-fda-approval.
- ↑ 26.0 26.1 "Why a ketamine-like drug is being used to treat depression". 6 March 2019. https://www.vox.com/2019/3/6/18253041/ketamine-johnson-johnson-spravato.
- ↑ Psychopharmacologic Drugs Advisory Committee (PDAC) and Drug Safety and Risk Management (DSaRM) Advisory Committee (12 February 2019). "FDA Briefing Document". Food and Drug Administration. https://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/PsychopharmacologicDrugsAdvisoryCommittee/UCM630970.pdf. "Meeting, February 12, 2019. Agenda Topic: The committees will discuss the efficacy, safety, and risk-benefit profile of New Drug Application (NDA) 211243, esketamine 28 mg single-use nasal spray device, submitted by Janssen Pharmaceutica, for the treatment of treatment-resistant depression."
- ↑ 28.0 28.1 28.2 28.3 28.4 28.5 "Esketamine for treatment-resistant depression: seven concerns about efficacy and FDA approval". Lancet Psychiatry 6 (12): 977–979. December 2019. doi:10.1016/S2215-0366(19)30394-3. PMID 31680014.
- ↑ 29.0 29.1 29.2 "Anti-depressant spray not recommended on NHS". 28 January 2020. https://www.bbc.com/news/health-51279176.
- ↑ Price, Maxwell Z.; Price, Richard L. (2024-12-01). "Benefits and risks of esketamine nasal spray continuation in treatment-resistant depression". Biomarkers in Neuropsychiatry 11. doi:10.1016/j.bionps.2024.100104. ISSN 2666-1446. https://www.sciencedirect.com/science/article/pii/S2666144624000224.
- ↑ 31.0 31.1 "J&J scores Spravato trial win in high-risk depression. Will doctors and payers buy in?". 10 September 2019. https://www.fiercepharma.com/pharma/j-j-scores-spravato-trial-win-high-risk-depression-will-doctors-and-payers-buy. "Pricing, though, may still be an issue. In early May, the Institute for Clinical and Economic Review (ICER) declined to recommend Spravato for use at its steep list price of $32,400 per year. The U.S. cost watchdog said J&J would need to cut the sticker price between 25% and 52% to be considered cost-effective."
- ↑ "1 Recommendations | Esketamine nasal spray for treatment-resistant depression | Guidance | NICE". 14 December 2022. https://www.nice.org.uk/guidance/ta854/chapter/1-Recommendations.
- ↑ "Intranasal esketamine: From origins to future implications in treatment-resistant depression". J Psychiatr Res 137: 29–35. May 2021. doi:10.1016/j.jpsychires.2021.02.020. PMID 33647726.
- ↑ Kaster, Tyler S.; Dai, Yi; Vila-Rodriguez, Fidel; Downar, Jonathan; Daskalakis, Zafiris J.; Blumberger, Daniel M.; Rhee, Taeho G. (2025-12-01). "Efficacy of intranasal esketamine versus rTMS for treatment-resistant depression: analysis of individual participant data from two clinical trials" (in English). eClinicalMedicine 90. doi:10.1016/j.eclinm.2025.103609. ISSN 2589-5370. PMID 41245530.
- ↑ "Molecular mechanisms of the rapid-acting and long-lasting antidepressant actions of (R)-ketamine". Biochem Pharmacol 177. July 2020. doi:10.1016/j.bcp.2020.113935. PMID 32224141.
- ↑ "Molecular mechanisms underlying the antidepressant actions of arketamine: beyond the NMDA receptor". Mol Psychiatry 27 (1): 559–573. May 2021. doi:10.1038/s41380-021-01121-1. PMID 33963284.
- ↑ 37.0 37.1 "[The clinical use of S-(+)-ketamine--a determination of its place]" (in de). Anästhesiologie, Intensivmedizin, Notfallmedizin, Schmerztherapie 33 (12): 764–70. December 1998. doi:10.1055/s-2007-994851. PMID 9893910.
- ↑ "Ketamine enantiomers in the rapid and sustained antidepressant effects". Therapeutic Advances in Psychopharmacology 6 (3): 185–92. June 2016. doi:10.1177/2045125316631267. PMID 27354907.
- ↑ "Ketamine's antidepressant action: beyond NMDA receptor inhibition". Expert Opinion on Therapeutic Targets 20 (11): 1389–1392. November 2016. doi:10.1080/14728222.2016.1238899. PMID 27646666.
- ↑ "Comparison of R-ketamine and rapastinel antidepressant effects in the social defeat stress model of depression". Psychopharmacology 233 (19–20): 3647–57. October 2016. doi:10.1007/s00213-016-4399-2. PMID 27488193.
- ↑ "Ketamine stereoselectively inhibits rat dopamine transporter". Neuroscience Letters 274 (2): 131–4. October 1999. doi:10.1016/s0304-3940(99)00688-6. PMID 10553955.
- ↑ 42.0 42.1 "[Ketamine racemate or S-(+)-ketamine and midazolam. The effect on vigilance, efficacy and subjective findings]" (in de). Der Anaesthesist 41 (10): 610–8. October 1992. PMID 1443509.
- ↑ "[Psychometric changes as well as analgesic action and cardiovascular adverse effects of ketamine racemate versus s-(+)-ketamine in subanesthetic doses]" (in de). Der Anaesthesist 43 (Suppl 2): S68-75. November 1994. PMID 7840417.
- ↑ 44.0 44.1 "Differential psychopathology and patterns of cerebral glucose utilisation produced by (S)- and (R)-ketamine in healthy volunteers using positron emission tomography (PET)". European Neuropsychopharmacology 7 (1): 25–38. February 1997. doi:10.1016/s0924-977x(96)00042-9. PMID 9088882.
- ↑ "R-ketamine: a rapid-onset and sustained antidepressant without psychotomimetic side effects". Translational Psychiatry 5 (9): e632. September 2015. doi:10.1038/tp.2015.136. PMID 26327690.
- ↑ "The Nuances of Ketamine's Neurochemistry" (in en-US). 15 February 2021. https://psychedelicreview.com/the-nuances-of-ketamines-neurochemistry/.
- ↑ "Stereoselective pharmacokinetics of ketamine: R(-)-ketamine inhibits the elimination of S(+)-ketamine". Clinical Pharmacology and Therapeutics 70 (5): 431–438. November 2001. doi:10.1016/S0009-9236(01)06321-4. PMID 11719729.
- ↑ 48.0 48.1 "Synthesizing the Evidence for Ketamine and Esketamine in Treatment-Resistant Depression: An International Expert Opinion on the Available Evidence and Implementation". The American Journal of Psychiatry 178 (5): 383–399. May 2021. doi:10.1176/appi.ajp.2020.20081251. PMID 33726522.
- ↑ "Ketamine and Esketamine in Clinical Trials: FDA-Approved and Emerging Indications, Trial Trends With Putative Mechanistic Explanations". Clin Pharmacol Ther 117 (2): 374–386. February 2025. doi:10.1002/cpt.3478. PMID 39428602.
- ↑ 50.0 50.1 "Beyond serotonin: newer antidepressants in the future". Expert Review of Neurotherapeutics 17 (8): 777–790. August 2017. doi:10.1080/14737175.2017.1341310. PMID 28598698.
- ↑ 51.0 51.1 51.2 51.3 "Esketamine - Johnson & Johnson - AdisInsight". http://adisinsight.springer.com/drugs/800037644.
- ↑ "Ketamine and Beyond: Investigations into the Potential of Glutamatergic Agents to Treat Depression". Drugs 77 (4): 381–401. March 2017. doi:10.1007/s40265-017-0702-8. PMID 28194724.
- ↑ "Janssen Submits Esketamine Nasal Spray New Drug Application to U.S. FDA for Treatment-Resistant Depression". Janssen Pharmaceuticals, Inc.. https://www.janssen.com/janssen-submits-esketamine-nasal-spray-new-drug-application-us-fda-treatment-resistant-depression.
- ↑ "FDA Approves A Nasal Spray To Treat Patients Who Are Suicidal". NPR.org. 4 August 2020. https://www.npr.org/2020/08/04/899060885/fda-approves-a-nasal-spray-to-treat-patients-who-are-suicidal.
- ↑ "A Paradigm Shift for Depression Treatment". Discover (Kalmbach Media). January 2020.
- ↑ "The FDA Approved a Ketamine-Like Nasal Spray for Hard-to-Treat Depression". Vice. 7 March 2019. https://www.vice.com/en/article/what-is-esketamine-fda-approves-nasal-spray-for-depression/. Retrieved 11 February 2020.
- ↑ Moncrieff, Joanna (16 January 2025). "Alternative Approaches: The Good, the Bad and the Worrying: Psychedelics for Depression". Chemically Imbalanced: The Making and Unmaking of the Serotonin Myth. Flint. ISBN 978-1-80399-680-6. https://books.google.com/books?id=e0MkEQAAQBAJ. Retrieved 16 October 2025.
- ↑ "'Breakthrough' nasal spray to treat depression coming to UAE". https://www.thenationalnews.com/uae/breakthrough-nasal-spray-to-treat-depression-coming-to-uae-1.928221?outputType=amp.
 |
