Chemistry:Xenon octafluoride
From HandWiki
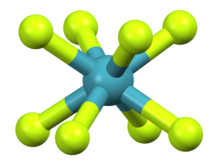 Approximate geometry predicted computationally
| |
| Names | |
|---|---|
| IUPAC name
Octafluoroxenon
| |
| Other names
Xenon(VIII) fluoride
| |
| Identifiers | |
3D model (JSmol)
|
|
| |
| Properties | |
| XeF 8 | |
| Related compounds | |
Related compounds
|
Osmium octafluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Xenon octafluoride is a chemical compound of xenon and fluorine with the chemical formula XeF
8. This is still a hypothetical compound.[1][2] XeF
8 is reported to be unstable even under pressures reaching 200 GPa.[3]
History
The compound was initially predicted in 1933 by Linus Pauling—among other noble gas compounds but which, unlike other xenon fluorides, could probably never be synthesized.[4][5] This appears to be due to the steric hindrance of the fluorine atoms around the xenon atom. However, scientists continue to try to synthesize it.[6]
Potential synthesis
The formation of xenon octafluoride has been calculated to be endothermic:[7]
- Xe + 4 F
2 → XeF
8
References
- ↑ Frlec, Boris; Holloway, John H.; Slivnik, Jože; Šmalc, Andrej; Volavšek, Bogdan; Zemljič, Anton (1 August 1970). "An examination of the possibility of the existence of xenon octafluoride" (in en). Journal of Inorganic and Nuclear Chemistry 32 (8): 2521–2527. doi:10.1016/0022-1902(70)80296-2. ISSN 0022-1902. https://www.sciencedirect.com/science/article/abs/pii/0022190270802962. Retrieved 31 March 2023.
- ↑ Housecroft, Catherine E.; Sharpe, A. G. (2008) (in en). Index. Pearson Prentice Hall. p. 1097. ISBN 978-0-13-175553-6. https://books.google.com/books?id=3sy4ZAP4EGAC&dq=xenon+octafluoride&pg=PA1097. Retrieved 31 March 2023.
- ↑ Luo, Dongbao; Lv, Jian; Peng, Feng; Wang, Yanchao; Yang, Guochun; Rahm, Martin; Ma, Yanming (2019). "A hypervalent and cubically coordinated molecular phase of IF 8 predicted at high pressure". Chemical Science 10 (8): 2543–2550. doi:10.1039/c8sc04635b. PMID 30881685. PMC 6385887. https://pubs.rsc.org/en/content/articlepdf/2019/sc/c8sc04635b. Retrieved 31 March 2023.
- ↑ Weinstock, Bernard; Weaver, E. Eugene; Knop, Charles P. (December 1, 1966). "The Xenon-Fluorine System". Inorg. Chem. 66 (5): 2189. doi:10.1021/ic50046a026. https://pubs.acs.org/doi/10.1021/ic50046a026#. Retrieved 31 March 2023.
- ↑ Pauling, Linus (May 1933). "The Formulas of Antimonic Acid and the Antimonates" (in en). Journal of the American Chemical Society 55 (5): 1895–1900. doi:10.1021/ja01332a016. ISSN 0002-7863. https://pubs.acs.org/doi/abs/10.1021/ja01332a016. Retrieved 31 March 2023.
- ↑ Cotton, F. Albert (17 September 2009) (in en). Progress in Inorganic Chemistry, Volume 6. John Wiley & Sons. p. 249. ISBN 978-0-470-16657-4. https://books.google.com/books?id=IhkQIoEGL24C&dq=xenon+octafluoride&pg=PA249. Retrieved 31 March 2023.
- ↑ Holleman, A. F.; Wiberg, Egon; Wiberg, Nils (2001) (in en). Inorganic Chemistry. Academic Press. p. 394. ISBN 978-0-12-352651-9. https://books.google.com/books?id=Mtth5g59dEIC&dq=xenon+octafluoride&pg=PA394. Retrieved 31 March 2023.
 |

