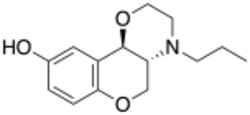
Chemistry:PD-128,907
From HandWiki
Short description: Chemical compound
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C14H19NO3 |
| Molar mass | 249.310 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
PD-128,907 is a drug used in scientific research which acts as a potent and selective agonist for the dopamine D2 and D3 receptors.[1] It is used for studying the role of these receptors in the brain, in roles such as inhibitory autoreceptors that act to limit further dopamine release,[2] as well as release of other neurotransmitters.[3] In animal studies, it has been shown to reduce toxicity from cocaine overdose.[4][5]
See also
References
- ↑ "Synthesis and dopamine agonist properties of (+-)-trans-3,4,4a,10b-tetrahydro-4-propyl-2H,5H-[1]benzopyrano [4,3-b]-1,4-oxazin-9-ol and its enantiomers". Journal of Medicinal Chemistry 33 (1): 445–50. January 1990. doi:10.1021/jm00163a068. PMID 1967318.
- ↑ "Alterations in dopamine release but not dopamine autoreceptor function in dopamine D3 receptor mutant mice". The Journal of Neuroscience 18 (6): 2231–8. March 1998. PMID 9482807.
- ↑ "Dopamine D3 receptors regulate GABAA receptor function through a phospho-dependent endocytosis mechanism in nucleus accumbens". The Journal of Neuroscience 26 (9): 2513–21. March 2006. doi:10.1523/JNEUROSCI.4712-05.2006. PMID 16510729.
- ↑ "Protection against cocaine toxicity in mice by the dopamine D3/D2 agonist R-(+)-trans-3,4a,10b-tetrahydro-4-propyl-2H,5H-[1]benzopyrano[4,3-b]-1,4-oxazin-9-ol [(+)-PD 128,907]". The Journal of Pharmacology and Experimental Therapeutics 308 (3): 957–64. March 2004. doi:10.1124/jpet.103.059980. PMID 14711932.
- ↑ "The dopamine D3/D2 agonist (+)-PD-128,907 [(R-(+)-trans-3,4a,10b-tetrahydro-4-propyl-2H,5H-[1]benzopyrano[4,3-b]-1,4-oxazin-9-ol)] protects against acute and cocaine-kindled seizures in mice: further evidence for the involvement of D3 receptors". The Journal of Pharmacology and Experimental Therapeutics 326 (3): 930–8. September 2008. doi:10.1124/jpet.108.139212. PMID 18566292.
 |
