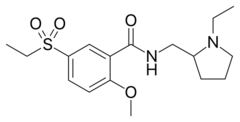
Chemistry:Sultopride
From HandWiki
Short description: Antipsychotic medication
 | |
| Clinical data | |
|---|---|
| Trade names | Barnetil, Barnotil, Topral |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, IM |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 3–5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C17H26N2O4S |
| Molar mass | 354.47 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Sultopride (trade names Barnetil, Barnotil, Topral) is an atypical antipsychotic of the benzamide chemical class used in Europe, Japan , and Hong Kong for the treatment of schizophrenia.[1][2][3] It was launched by Sanofi-Aventis in 1976.[1] Sultopride acts as a selective D2 and D3 receptor antagonist.[4] It has also been shown to have clinically relevant affinity for the GHB receptor as well, a property it shares in common with amisulpride and sulpiride.[5]
Pharmacology
| Site | Ki | Species | Ref |
|---|---|---|---|
| D2 | 1.6 | Human | [6] |
| D3 | 3.8 | Human | [6] |
References
- ↑ 1.0 1.1 Antidepressants, Antipsychotics, Anxiolytics: From Chemistry and Pharmacology to Clinical Application. Weinheim: Wiley-VCH. 2007. ISBN 978-3-527-31058-6. https://books.google.com/books?id=yXD4QA-Y_Z0C&pg=PA537.
- ↑ Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. ISBN 3-88763-075-0. https://books.google.com/books?id=5GpcTQD_L2oC&dq=sultopride&pg=PA983.
- ↑ European Drug Index (4th ed.). Boca Raton: CRC Press. 1998. ISBN 3-7692-2114-1. https://books.google.com/books?id=HiSdvzs2pPAC&pg=PA136.
- ↑ "Intrinsic efficacy of antipsychotics at human D2, D3, and D4 dopamine receptors: identification of the clozapine metabolite N-desmethylclozapine as a D2/D3 partial agonist". The Journal of Pharmacology and Experimental Therapeutics 315 (3): 1278–1287. December 2005. doi:10.1124/jpet.105.092155. PMID 16135699.
- ↑ "Displacement of [3H] gamma-hydroxybutyrate binding by benzamide neuroleptics and prochlorperazine but not by other antipsychotics". European Journal of Pharmacology 256 (2): 211–214. April 1994. doi:10.1016/0014-2999(94)90248-8. PMID 7914168.
- ↑ 6.0 6.1 "Intrinsic efficacy of antipsychotics at human D2, D3, and D4 dopamine receptors: identification of the clozapine metabolite N-desmethylclozapine as a D2/D3 partial agonist". The Journal of Pharmacology and Experimental Therapeutics 315 (3): 1278–1287. December 2005. doi:10.1124/jpet.105.092155. PMID 16135699.
 |
