Chemistry:Tolcapone
Tolcapone, sold under the brand name Tasmar, is a medication used to treat Parkinson's disease (PD). It is a selective, potent and reversible nitrocatechol-type inhibitor of the enzyme catechol-O-methyltransferase (COMT).[1] It has demonstrated significant liver toxicity, which has led to suspension of marketing authorisations in a number of countries.
Tolcapone appears to be peripherally selective, but can still cross into the brain in significant amounts and has been found to inhibit COMT centrally as well[clarification needed].[2][3] In comparison with entacapone, another nitrocatechol COMT inhibitor, tolcapone has a longer half life (2.9 hours vs. 0.8 hours) and can better penetrate into the brain, acting both in the central nervous system and in the periphery.[4] However, entacapone is less toxic for the liver.
Medical uses
Tolcapone is used in the treatment of Parkinson's disease as an adjunct to levodopa/carbidopa or levodopa/benserazide medications. Levodopa is a prodrug for dopamine, which reduces Parkinson symptoms; carbidopa and benserazide are aromatic L-amino acid decarboxylase (AADC) inhibitors.[5]
Without administration of tolcapone, the beneficial effects of levodopa tend to wear off more quickly, resulting in motor fluctuations.[6]
Contraindications
Combining tolcapone with non-selective monoamine oxidase inhibitors such as phenelzine or tranylcypromine is contraindicated.[7] Tolcapone is also contraindicated for people with liver diseases or increased liver enzymes.[8]
Side effects
Tolcapone has demonstrated significant liver toxicity (hepatotoxicity)[9] that limits the drug's utility. Entacapone is an alternative, largely since it has a more favorable toxicity profile.
The hepatotoxicity can be related to elevated levels of transaminases, but studies have shown that minimal risk exists for those without preexisting liver conditions when their enzyme levels were being monitored. No clear mechanism is implicated, but it has been hypothesized that it has something to do with abnormal mitochondrial respiration due to the uncoupling of oxidative phosphorylation.[10]
Other side effects regard the increase in dopaminergic activity, including digestive symptoms.[8] Treatment with tolcapone runs the risk of eliciting or prolonging dyskinesia; this can be counteracted by decreasing the dose of levodopa. This occurs because the administration of tolcapone results in the accumulation of the biological methyl donor S-adenosyl-L-methionine (SAM) in the striatum that induces Parkinson symptoms.[10]
Digestive symptoms include nausea and diarrhea; further dopaminergic side effects include orthostatic hypotension, dry mouth, sweating and dizziness. Tolcapone causes more severe diarrhea than entacapone; this was the most common reason for therapy termination in studies.[7] Urine discoloration comes from yellow tolcapone metabolites being excreted in the urine and is harmless.[8][10]
Interactions
While increase of dopamine levels is a desired interaction, tolcapone can theoretically also increase the levels of other drugs metabolised by COMT, such as the AADC inhibitors carbidopa and benserazide, as well as methyldopa, dobutamine, apomorphine, epinephrine (adrenaline), and isoprenaline. In studies, a slight interaction with benzerazide was seen, but not with carbidopa. Other interactions with this group of drugs have not been studied. A related type of theoretical interactions is with drugs that increase catecholamine concentrations, such as monoamine oxidase (MAO) inhibitors and norepinephrine reuptake inhibitors; these also showed only slight effects in practice. Combination with non-selective MAO inhibitors might be dangerous.[7][8]
Due to its affinity to the liver enzyme CYP2C9, interactions with drugs being metabolised by this enzyme are also possible, but unlikely. No interaction with tolbutamide, a 2C9 substrate, was observed in studies.[8]
Pharmacology
Mechanism of action

Tolcapone selectively and reversibly[7] binds to the catalytic site of COMT in both the periphery and the central nervous system (CNS) with greater affinity than any of the three catecholamines, including levodopa.[12] It thereby prevents the 3-O-methylation of levodopa by COMT in the periphery, which produces 3-O-methyldopa, a major metabolite that competes with levodopa to cross the blood–brain barrier. More of the levodopa that is administered reaches the CNS. Additionally, levodopa that has already reached the CNS, after being converted to dopamine, will not be degraded as quickly when tolcapone inhibits COMT activity. Thus, tolcapone improves the bioavailability and reduces the clearance of levodopa and subsequently dopamine from the CNS.[13]

The strength of the binding affinity of tolcapone, represented by the inhibition constant Ki (2.5 nM), can be thought of as the dissociation constant for enzyme and inhibitor complex kinetics. Maximum catalytic activity denotes the efficacy of tolcapone (Vmax = 58.4 pmol/min·mg).[14]
Pharmacokinetics
Tolcapone is quickly absorbed from the gut to about 85%. It has an absolute bioavailability of 65%, which is only slightly decreased when taken with food. The substance reaches highest blood plasma concentrations after about two hours. When in the bloodstream, it is almost completely (>99.9%) bound to plasma proteins, primarily albumin. The main inactivation step is glucuronidation; other processes are methylation by COMT, hydroxylation by CYP3A4 and CYP2A6 with subsequent oxidation to a carboxylic acid, and possibly a minor path with reduction to an amine with subsequent acetylation.[7][8]
The half-life of tolcapone is two to three hours, the volume of distribution (Vd) being 0.3 L/kg (21 L in an average 70 kg person).[6] 60% of the metabolites are excreted via the urine and 40% via the feces. Only 0.5% of the drug are excreted in unchanged form via the urine.[7][8]

99% of tolcapone is in monoanionic form in the body because the physiological pH is 7.4. Tolcapone penetrates the blood–brain barrier much better than two other nitrocatechols, nitecapone and entacapone, because it has higher lipophilicity due to its R-substituent. Partition coefficients quantify the ability of the molecule to cross the blood–brain barrier. LogPIdce= 0.2, –1.4, –0.4 for tolcapone, nitecapone and entacopone respectively. Partition coefficients in this case were measured in 1,2-dichloroethane/H2O solution which caused molecules to be in ionized form. There is no current explanation for how these charged molecules permeate the blood–brain barrier.[15]
Tolcapone has been said to enter the central nervous system only minimally and hence would appear to be peripherally selective.[2] However, tolcapone is more lipophilic than other COMT inhibitors like entacapone and may be more likely to cross the blood–brain barrier (BBB).[3] A imaging study in humans found that tolcapone also inhibited COMT in the brain to a significant degree.[3]
Chemistry
Tolcapone is an intensely yellow, odorless, bitter tasting, non-hygroscopic, crystalline compound with a relative molecular mass of 273.25 g/mol. It melts at 143 to 146 °C (289 to 295 °F), is practically insoluble in water and acids but soluble in 0.1 M aqueous sodium hydroxide solution. The pKa values are 4.5 and 10.6 for the two phenyl groups; and the maximum absorption is at 268 nm (in 0.1 M hydrochloric acid / ethanol).[7] Its chemical name is 3,4-dihydroxy-4'-methyl-5-nitrobenzophenone.
Synthesis
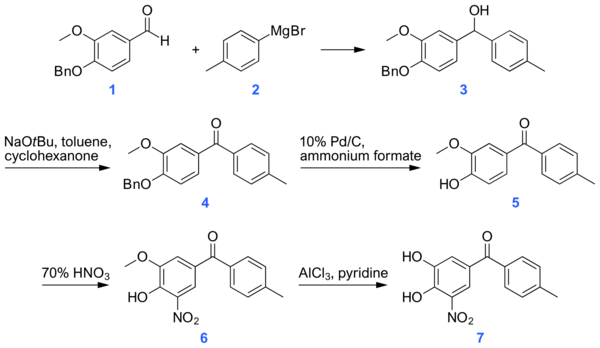
A synthesis of tolcapone proposed in 2008, begins with a Grignard reaction between 4-Benzyloxy-3-methoxybenzaldehyde [16][17] and p-tolyl magnesium bromide. The alcohol thus produced is then converted to a ketone using sodium t-butoxide. The benzyl protecting group is removed by palladium-catalyzed hydrogenation in the presence of ammonium formate. A nitro group is introduced at the 5-position adjacent to the hydroxyl group unmasked in the cleavage of the benzyl ether. The synthesis ends with cleavage of the methoxy group using aluminum chloride to yield the product alcohol.[18]
History
Tolcapone was introduced into the European market in August 1997, and subsequently into the United States market in March 1998. Liver toxicity was reported in four people who were administered tolcapone, three people died due to complications. Consequentially, the marketing authorization of tolcapone was suspended from December 1998 until August 2004 when it was lifted. In November 1998, the company that manufactured tolcapone voluntarily[19] removed the drug from the market. The authorization was then renewed in August 2009.[20]
As a result of reported complications, the US Food and Drug Administration (FDA) issued a black box warning for tolcapone and label revisions that aimed to regulate the monitoring of those prescribed tolcapone for Parkinson's disease in November 1998.[21] A number of other countries withdrew tolcapone from the market; Australia in February 1999, Bulgaria in April 1999, Iceland in November 1998, Lithuania in December 1998.[19]
Research
Transthyretin amyloidosis
Because of preliminary data suggesting the drug may have activity, the U.S. FDA in 2013 granted tolcapone "orphan drug status" in studies aiming at the treatment of transthyretin familial amyloidosis (ATTR).[22] However, as of 2015[update] tolcapone was not FDA approved for the treatment of this disease.[23]
Psychiatric disorders
In animal studies, tolcapone shows antidepressant- and anti-anhedonia-like effects, stimulates exploratory behavior, and enhances the locomotor hyperactivity induced by psychostimulants like amphetamine and nomifensine.[24][25][26] Tolcapone has been clinically studied in the treatment of certain psychiatric disorders such as obsessive–compulsive disorder (OCD) and for the negative symptoms of schizophrenia.[27][28][29][30] There is also interest in brain-penetrant COMT inhibitors like tolcapone for the treatment of disorders of diminished motivation like apathy.[31]
References
- ↑ "COMT inhibition with tolcapone in the treatment algorithm of patients with Parkinson's disease (PD): relevance for motor and non-motor features". Neuropsychiatric Disease and Treatment 4 (1): 1–9. February 2008. doi:10.2147/NDT.S2404. PMID 18728767.
- ↑ 2.0 2.1 "Tolcapone: a review of its use in the management of Parkinson's disease". CNS Drugs 19 (2): 165–184. 2005. doi:10.2165/00023210-200519020-00006. PMID 15697329. "The efficacy of tolcapone as an adjunct to levodopa in patients with Parkinson's disease has primarily been attributed to its ability to inhibit peripheral it is thought that tolcapone enters the CNS to a minimal extent only.[16] However, results [17] of a study in patients with Parkinson's disease, as well as results of animal studies,[18-21] suggest that tolcapone also has central activity.".
- ↑ 3.0 3.1 3.2 "Opicapone for the treatment of Parkinson's disease: A review of a new licensed medicine". Mov Disord 33 (10): 1528–1539. October 2018. doi:10.1002/mds.27475. PMID 30264443. "The possible central effect of tolcapone, which is more lipophilic than entacapone and may be more likely to cross the blood-brain barrier (BBB), has been under debate for many years.26 An (18)F-dopa positron emission tomography study showed its effect as a central COMT inhibitior.27 However, this effect was not translated into clinical benefit in L-dopa-naive patients, in whom tolcapone alone or added to selegiline was compared with placebo.28 Tolcapone is more efficacious than entacapone, as it reduces total OFF time by an average of 98 minutes daily.".
- ↑ "Pharmacokinetics and pharmacodynamics of entacapone and tolcapone after acute and repeated administration: a comparative study in the rat". The Journal of Pharmacology and Experimental Therapeutics 304 (2): 498–506. February 2003. doi:10.1124/jpet.102.042846. PMID 12538800.
- ↑ "Pharmacokinetic-pharmacodynamic interaction between the COMT inhibitor tolcapone and single-dose levodopa". British Journal of Clinical Pharmacology 40 (3): 253–62. September 1995. doi:10.1111/j.1365-2125.1995.tb05781.x. PMID 8527287.
- ↑ 6.0 6.1 "Pharmacokinetics and pharmacodynamics after oral and intravenous administration of tolcapone, a novel adjunct to Parkinson's disease therapy". European Journal of Clinical Pharmacology 54 (5): 443–7. July 1998. doi:10.1007/s002280050490. PMID 9754991.
- ↑ 7.0 7.1 7.2 7.3 7.4 7.5 7.6 (in German) Arzneistoff-Profile. 10 (13 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. 1998. ISBN 978-3-7741-9846-3.
- ↑ 8.0 8.1 8.2 8.3 8.4 8.5 8.6 8.7 "Tasmar: EPAR – Product Information". European Medicines Agency. 3 August 2016. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000132/WC500034731.pdf.
- ↑ "Tolcapone: an efficacy and safety review (2007)". Clinical Neuropharmacology 30 (5): 287–94. 2007. doi:10.1097/wnf.0b013e318038d2b6. PMID 17909307.
- ↑ 10.0 10.1 10.2 "Tolcapone: review of its pharmacology and use as adjunctive therapy in patients with Parkinson's disease". Clinical Interventions in Aging 4: 109–13. 2009. doi:10.2147/CIA.S3787. PMID 19503773.
- ↑ "Catechol-O-methyltransferase in complex with substituted 3'-deoxyribose bisubstrate inhibitors". Acta Crystallographica. Section D, Biological Crystallography 68 (Pt 3): 253–60. March 2012. doi:10.1107/S0907444912001138. PMID 22349227.
- ↑ "Clinical pharmacokinetic and pharmacodynamic properties of drugs used in the treatment of Parkinson's disease". Clinical Pharmacokinetics 41 (4): 261–309. 2002. doi:10.2165/00003088-200241040-00003. PMID 11978145.
- ↑ "Metabolism and excretion of tolcapone, a novel inhibitor of catechol-O-methyltransferase". British Journal of Clinical Pharmacology 48 (4): 513–20. October 1999. doi:10.1046/j.1365-2125.1999.00036.x. PMID 10583021.
- ↑ "The role of physicochemical properties of entacapone and tolcapone on their efficacy during local intrastriatal administration". European Journal of Pharmaceutical Sciences 24 (5): 503–11. April 2005. doi:10.1016/j.ejps.2005.01.005. PMID 15784340.
- ↑ "The Lipophilicity Behavior of Three Catechol-O-methyltransferase (COMT) Inhibitors and Simple Analogues". Helvetica Chimica Acta 89 (1): 144–152. 2006. doi:10.1002/hlca.200690007. Bibcode: 2006HChAc..89..144N.
- ↑ "3-Benzyloxy-4-methoxybenzaldehyde". PubChem. U.S. National Library of Medicine. https://pubchem.ncbi.nlm.nih.gov/compound/3-Benzyloxy-4-methoxybenzaldehyde.
- ↑ "Convenient Synthesis of Tolcapone, a Selective Catechol-O-methyltransferase Inhibitor". Synthetic Communications 38 (5): 810–815. 2008. doi:10.1080/00397910701821077. https://www.tandfonline.com/doi/abs/10.1080/00397910701821077?journalCode=lsyc20.
- ↑ 18.0 18.1 "Convenient Synthesis of Tolcapone, a Selective Catechol-O-methyltransferase Inhibitor". Synthetic Communications 38 (5): 810–815. 2008. doi:10.1080/00397910701821077.
- ↑ 19.0 19.1 "2001 Pharmaceuticals: Restrictions in Use and Availability". World Health Organization. http://apps.who.int/medicinedocs/en/d/Js2203e/2.59.html.
- ↑ "Tasmar tolcapone EPAR summary for the public". European Medicine Agency. http://www.emea.europa.eu/docs/en_GB/document_library/EPAR_-_Summary_for_the_public/human/000132/WC500034729.pdf.
- ↑ Press Office, U.S. Department of Health and Human Services. "FDA Talk Paper". New Warnings for Parkinson's Drug, Tasmar. Food and Drug Administration. http://scienceblog.com/community/older/archives/M/1/fda0533.htm.
- ↑ "Tolcapone". 1 January 2013. https://www.accessdata.fda.gov/scripts/opdlisting/oopd/detailedIndex.cfm?cfgridkey=413913.
- ↑ "SOM0226, a repositioned compound for the treatment of TTR amyloidosis". Orphanet J Rare Dis 10 (Suppl 1). 2015. doi:10.1186/1750-1172-10-s1-p9.
- ↑ "Tolcapone, a selective catechol-O-methyltransferase inhibitor for treatment of Parkinson's disease". Pharmacotherapy 19 (1): 6–20. January 1999. doi:10.1592/phco.19.1.6.30516. PMID 9917075. "It also enhances locomotor hyperactivity induced by amphetamine and nomifensine and stereotypy induced by amphetamine, and stimulates exploratory activity in the open field test in rats and mice.14 Tolcapone potentiates levodopa antagonism of haloperidol-induced catalepsy in MPP+-lesioned mice (murine model of Parkinson’s disease) and potentiates and prolongs levodopa-induced circling behavior in rats with 6-hydroxydopamine-induced nigrostriatal pathway lesions (another animal model of Parkinson’s disease).23, 24 [...] The effect of tolcapone on animal models of depression was evaluated in two studies. In rats with chronic mild stress-induced anhedonia, tolcapone 10 or 30 mg/kg twice/day by intraperitoneal injection prevented the stress-induced anhedonic state compared with vehicle-treated controls.28 Another rat study using the forced swimming test and learned helplessness paradigm, found no significant antidepressant activity of the agent.29 The relevance of these findings to the management of depression in humans with both parkinsonian and nonparkinsonian disease is unknown.".
- ↑ "Behavioural and neurochemical effects of Ro 40-7592, a new COMT inhibitor with a potential therapeutic activity in Parkinson's disease". J Neural Transm Park Dis Dement Sect 2 (2): 101–112. 1990. doi:10.1007/BF02260898. PMID 1977408.
- ↑ "POSTER COMMUNICATIONS: 49P. BIA 3-202 does not potentiate locomotor hyperactivity during increased dopaminergic stimulation". British Journal of Pharmacology (Wiley) 131 (Suppl): 38P–129P. October 2000. "Tolcapone administered 6 h before amphetamine challenge was found to significantly increase locomotor activity in rats treated with 0.5 and 2.0 mg kg-1 amphetamine. In rats given 4.0 mg kg-1 amphetamine, tolcapone produced a marked decrease in locomotor activity and increased two-fold the duration of the stereotyped behaviour.".
- ↑ "A systematic review of the cognitive effects of the COMT inhibitor, tolcapone, in adult humans". CNS Spectr 29 (3): 166–175. June 2024. doi:10.1017/S1092852924000130. PMID 38487834.
- ↑ "Tolcapone in obsessive-compulsive disorder: a randomized double-blind placebo-controlled crossover trial". Int Clin Psychopharmacol 36 (5): 225–229. September 2021. doi:10.1097/YIC.0000000000000368. PMID 34310432.
- ↑ "Treatment of cognitive deficits associated with schizophrenia: potential role of catechol-O-methyltransferase inhibitors". CNS Drugs 21 (7): 535–557. 2007. doi:10.2165/00023210-200721070-00002. PMID 17579498.
- ↑ "ACNP 63rd Annual Meeting: Poster Abstracts P1-P304: P158. Tolcapone for Negative Symptoms of Schizophrenia Spectrum Disorders, A Pilot Study". Neuropsychopharmacology 49 (S1): 65–235. 2024. doi:10.1038/s41386-024-02011-0. ISSN 0893-133X. PMC 11627186. https://www.nature.com/articles/s41386-024-02011-0. Retrieved 19 January 2026.
- ↑ "Apathy and Motivation: Biological Basis and Drug Treatment". Annu Rev Pharmacol Toxicol 64: 313–338. January 2024. doi:10.1146/annurev-pharmtox-022423-014645. PMID 37585659.
External links
- "Tolcapone". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/tolcapone.
 |
