Chemistry:Nomifensine
Nomifensine, formerly sold under the brand names Merital and Alival, is a norepinephrine–dopamine reuptake inhibitor (NDRI) drug[1] that was developed in the 1960s by Hoechst AG (now Sanofi-Aventis),[2] who then test marketed it in the United States.
Nomifensine was considered an effective antidepressant that lacked sedative effects. It did not interact significantly with alcohol and lacked anticholinergic effects. No withdrawal symptoms were seen after 6 months treatment. The drug was, however, considered not suitable for agitated patients as it presumably made agitation worse.[3][4] In January 1986 the drug was withdrawn by its manufacturers for safety reasons.[5]
Some case reports in the 1980s suggested that there was potential for psychological dependence on nomifensine, typically in patients with a history of stimulant addiction, or when the drug was used in very high doses (400–600 mg per day).[6]
In a 1989 study it was investigated for use in treating attention deficit hyperactivity disorder (ADHD) in adults and was proven to be effective.[7] In a 1977 study it was not proven of benefit in advanced parkinsonism, except for depression associated with the parkinsonism.[8]
Medical uses
Nomifensine was investigated for use as an antidepressant in the 1970s, and was found to be a useful antidepressant at doses of 50–225 mg per day, both motivating and anxiolytic.
Adverse effects
During treatment with nomifensine there were relatively few adverse effects, mainly renal failure, paranoid symptoms, drowsiness or insomnia, headache, and dry mouth. Side effects affecting the cardiovascular system included tachycardia and palpitations, but nomifensine was significantly less cardiotoxic than the standard tricyclic antidepressants.[9]
Withdrawal from market
Due to a risk of haemolytic anaemia, the U.S. Food and Drug Administration (FDA) withdrew approval for nomifensine on March 20, 1992. Nomifensine was subsequently withdrawn from the Canadian and UK markets as well.[10] Some deaths were linked to immunohaemolytic anemia caused by this compound, although the mechanism remained unclear.[11]
Synthesis
Nomifensine was a progenitor to Gastrophenzine.[12] See also: Isatin derivatives.[13]
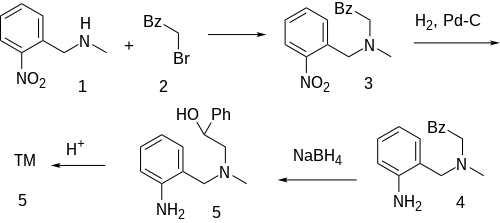
The alkylation between N-methyl-2-nitrobenzylamine [56222-08-3] (1) and phenacyl bromide (2) gives CID:15326127 (3). Catalytic hydrogenation over Raney Nickel reduces the nitro group to give CID:15113381 (4). The reduction of the ketone group with sodium borohydride to alcohol gives [65514-97-8] (5). Acid catalysed ring closure completes the formation of nomifensine (6).
Research
Motivational disorders
Nomifensine has been found to reverse tetrabenazine-induced motivational deficits in animals.[20] It shares these pro-motivational effects with other NDRIs like bupropion and methylphenidate and with selective dopamine reuptake inhibitors like modafinil and its analogues.[21][22][20] Conversely, selective norepinephrine reuptake inhibitors like desipramine and atomoxetine and selective serotonin reuptake inhibitors like fluoxetine and citalopram have not shown pro-motivational effects in animals.[21][22][20][23]
Wakefulness
Nomifensine shows wakefulness-promoting effects in animals and might be useful in the treatment of narcolepsy.[24][25][26][27]
See also
- Amineptine
- Diclofensine
- Perafensine
- Tesofensine
- The combination of Clobazam / nomifensine is called Psyton [75963-47-2].[28]
References
- ↑ "Nomifensine: A review of its pharmacological properties and therapeutic efficacy in depressive illness". Drugs 18 (1): 1–24. July 1979. doi:10.2165/00003495-197918010-00001. PMID 477572.
- ↑ Ehrhart G, Schmitt K, Hoffmann I, Ott H, "4-Phenyl-8-Amino Tetrahydroisoquinolines", US patent 3577424, issued 1971-05-04, assigned to Farbwerke Hoechst
- ↑ "A review of controlled studies with nomifensine, performed outside the UK". British Journal of Clinical Pharmacology 4 (Suppl 2): 237S–241S. 1977. doi:10.1111/j.1365-2125.1977.tb05759.x. PMID 334230.
- ↑ "An overview of side effects and long-term experience with nomifensine from United States clinical trials". The Journal of Clinical Psychiatry 45 (4 Pt 2): 96–101. April 1984. PMID 6370985.
- ↑ "CSM Update: Withdrawal of nomifensine". British Medical Journal 293 (6538): 41. July 1986. doi:10.1136/bmj.293.6538.41. PMID 20742679.
- ↑ "Nomifensine and psychological dependence--a case report". Pharmacopsychiatry 19 (5): 386–8. September 1986. doi:10.1055/s-2007-1017275. PMID 3774872.
- ↑ "Nomifensine maleate in adult attention deficit disorder". The Journal of Nervous and Mental Disease 177 (5): 296–9. May 1989. doi:10.1097/00005053-198905000-00008. PMID 2651559.
- ↑ "Nomifensine in Parkinson's disease". British Journal of Clinical Pharmacology 4 (Suppl 2): 187S–190S. 1977. doi:10.1111/j.1365-2125.1977.tb05751.x. PMID 334223.
- ↑ "A profile of nomifensine". British Journal of Clinical Pharmacology 4 (Suppl 2): 243S–248S. 1977. doi:10.1111/j.1365-2125.1977.tb05760.x. PMID 911653.
- ↑ "Nomifensine DB04821". Drugbank.ca. http://www.drugbank.ca/drugs/DB04821.
- ↑ "[Hematologic toxicity of antidepressive agents]" (in fr). L'Encéphale 14 (4): 307–18. 1988. PMID 3058454.
- ↑ 12.0 12.1 "Synthesis and pharmacological evaluation of some new tetrahydroisoquinoline derivatives inhibiting dopamine uptake and/or possessing a dopaminomimetic property". Journal of Medicinal Chemistry 29 (7): 1189–95. July 1986. doi:10.1021/jm00157a012. PMID 3806569.
- ↑ Boltze KH, Davies MA, Junge B, Schuurman T, Traber J, "Pyridoindole derivatives, compositions and use", DE patent 3333994, issued 14 January 1986
- ↑ "[8-amino-4-phenyl-1,2,3,4-tetrahydroisoquinolines, a new group of antidepressive psycholeptic drugs]". Arzneimittel-Forschung 21 (7): 1045. July 1971. PMID 5109496.
- ↑ "Synthesis of racemic (+) and (-) N-[methyl-11C]nomifensine, a ligand for evaluation of monoamine re-uptake sites by use of positron emission tomography". International Journal of Radiation Applications and Instrumentation, Part A 40 (2): 171–6. 1989. doi:10.1016/0883-2889(89)90194-9. PMID 2541106.
- ↑ "Verbesserte Synthese von 8-Amino-2-methyl-4-phenyl-1, 2, 3, 4-Tetrahydroisochinolin.". Journal für Praktische Chemie 331 (5): m 731–735. 1989. doi:10.1002/prac.19893310505.
- ↑ "A New Synthesis of 1, 2, 3, 4-Tetrahydro-2-methyl-4-phenylisoquinolines.". Synthesis 1990 (3): 253–255. 1990. doi:10.1055/s-1990-26846.
- ↑ "4-Phenyl tetrahydroisoquinolines as dual norepinephrine and dopamine reuptake inhibitors". Bioorganic & Medicinal Chemistry Letters 22 (23): 7219–22. December 2012. doi:10.1016/j.bmcl.2012.09.050. PMID 23084899.
- ↑ "Resolution, absolute stereochemistry, and enantioselective activity of nomifensine and hexahydro-1H-indeno[1,2-b]pyridines". Journal of Medicinal Chemistry 30 (5): 798–804. May 1987. doi:10.1021/jm00388a009. PMID 3572969.
- ↑ 20.0 20.1 20.2 "The Role of Dopamine in Effort- Based Decisions: Insights from Bupropion, Nomifensine, and Atomoxetine". 2023. https://archives.lib.uconn.edu/node/3764607.
- ↑ 21.0 21.1 "The Neurobiology of Activational Aspects of Motivation: Exertion of Effort, Effort-Based Decision Making, and the Role of Dopamine". Annu Rev Psychol 75: 1–32. January 2024. doi:10.1146/annurev-psych-020223-012208. PMID 37788571.
- ↑ 22.0 22.1 "The Psychopharmacology of Effort-Related Decision Making: Dopamine, Adenosine, and Insights into the Neurochemistry of Motivation". Pharmacol Rev 70 (4): 747–762. October 2018. doi:10.1124/pr.117.015107. PMID 30209181.
- ↑ "Not All Antidepressants Are Created Equal: Differential Effects of Monoamine Uptake Inhibitors on Effort-Related Choice Behavior". Neuropsychopharmacology 41 (3): 686–694. February 2016. doi:10.1038/npp.2015.188. PMID 26105139.
- ↑ "Modes of Action of Drugs Related to Narcolepsy: Pharmacology of Wake-Promoting Compounds and Anticataplectics". Narcolepsy. Cham: Springer International Publishing. 2016. pp. 307–329. doi:10.1007/978-3-319-23739-8_22. ISBN 978-3-319-23738-1.
- ↑ "Narcolepsy: pathophysiology and pharmacology". J Clin Psychiatry 68 (Suppl 13): 9–15. 2007. PMID 18078360.
- ↑ "A practical guide to the therapy of narcolepsy and hypersomnia syndromes". Neurotherapeutics 9 (4): 739–752. October 2012. doi:10.1007/s13311-012-0150-9. PMID 23065655.
- ↑ "Increased dopaminergic transmission mediates the wake-promoting effects of CNS stimulants". Sleep Res Online 1 (1): 49–61. 1998. PMID 11382857.
- ↑ Jellinger K, Koeppen D, Rössner M. Langzeitbehandlung depressiver Syndrome mit Psyton [Long-term treatment of depressive syndromes with Psyton (author's transl)]. Wien Med Wochenschr. 1982 Apr 30;132(8):183-8. German. PMID 6125057.
Template:Wakefulness-promoting agents
 |
