Chemistry:Iodyl fluoride
| |
| Names | |
|---|---|
IUPAC name
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
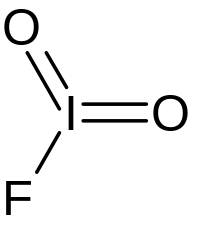
| IO 2F | |
| Molar mass | 177.901 g·mol−1 |
| Appearance | colorless crystals |
| Density | 4.982 g/cm3 |
| Melting point | 200 °C (392 °F; 473 K) |
| Reacts with water | |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Iodyl fluoride is an inorganic compound of iodine, fluorine, and oxygen with the chemical formula IO
2F. It is in the form of colorless crystals. Iodyl fluoride features iodine in the oxidation state of +5. The compound was initially synthesized in 1951.[1]
Synthesis
Iodyl fluoride can be decomposed by iodosyl trifluoride heated to 110 °C (230 °F) in nitrogen. Since this reaction is reversible, the reaction requires constant removal of iodine pentafluoride.[1]
- 2 IOF
3 ⇌ IO
2F + IF
5
It can also be obtained by dissolving iodine pentoxide, I
2O
5, in anhydrous hydrogen fluoride.[2]
- I
2O
5 + HF → IO
2F + HIO
3
Physical properties
Iodyl fluoride forms colorless crystals of orthorhombic system.[3] Reacts with water.[4]
Chemical properties
Iodyl fluoride is stable in dry air, but slowly hydrolyzes to iodic and hydrofluoric acids in moisture.[1]
- IO
2F + H
2O → HIO
3 + HF
The compound reacts with strong fluorinating agents such as bromine trifluoride and selenium tetrafluoride to form iodine pentafluoride. Iodyl fluoride can be reduced to elemental iodine by pure hydrogen peroxide.[5][6]
References
- ↑ 1.0 1.1 1.2 Aynsley, E. E.; Nichols, R.; Robinson, P. L. (1 January 1953). "126. Reactions of iodine pentafluoride with inorganic substances. Iodine oxytrifluoride and iodyl fluoride" (in en). Journal of the Chemical Society: 623–626. doi:10.1039/JR9530000623. ISSN 0368-1769. https://pubs.rsc.org/en/content/articlelanding/1953/JR/jr9530000623. Retrieved 24 May 2023.
- ↑ Wiberg, Egon; Wiberg, Nils (2001) (in en). Inorganic Chemistry. Academic Press. p. 468. ISBN 978-0-12-352651-9. https://books.google.com/books?id=Mtth5g59dEIC&dq=Iodosyl+trifluoride&pg=PA468. Retrieved 24 May 2023.
- ↑ Minkwitz, Rolf; Berkei, Michael; Ludwig, Ralf (1 December 2001). "Crystal Structure of IO2F" (in en). Inorganic Chemistry 40 (25): 6493–6495. doi:10.1021/ic0105462. ISSN 0020-1669. PMID 11720506. https://pubs.acs.org/doi/10.1021/ic0105462. Retrieved 24 May 2023.
- ↑ Haynes, William M. (4 June 2014) (in en). CRC Handbook of Chemistry and Physics. CRC Press. p. 4-67. ISBN 978-1-4822-0868-9. https://books.google.com/books?id=bNDMBQAAQBAJ&dq=Iodosyl+trifluoride&pg=SA4-PA67. Retrieved 24 May 2023.
- ↑ Schmeisser, M.; Brändle, K. (1 January 1963). "Oxides and Oxyfluorides of the Halogens" (in en). Advances in Inorganic Chemistry and Radiochemistry (Academic Press) 5: 41–89. doi:10.1016/S0065-2792(08)60152-1. ISBN 9780120236053. https://www.sciencedirect.com/science/article/abs/pii/S0065279208601521. Retrieved 24 May 2023.
- ↑ (in en) Advances in Inorganic Chemistry and Radiochemistry. Academic Press. 1 January 1963. ISBN 978-0-08-057854-5. https://books.google.com/books?id=pRXIwIV-hB8C&dq=%22Iodyl+fluoride%22&pg=PA83. Retrieved 24 May 2023.
 |
