Chemistry:1-Phenylethylamine
From HandWiki
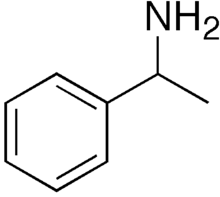
| |
| Names | |
|---|---|
| Preferred IUPAC name
1-Phenylethan-1-amine | |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII |
|
| |
| |
| Properties | |
| C8H11N | |
| Molar mass | 121.183 g·mol−1 |
| Density | 0.94 g/mL |
| Boiling point | 187 °C (369 °F; 460 K) |
| Hazards | |
| Main hazards | Corrosive |
| Related compounds | |
Related stereoisomers
|
(R)-(+)- (CAS [3886-69-9]) (S)-(−)- (CAS [2627-86-3]) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
1-Phenylethylamine is the organic compound with the formula C6H5CH(NH2)CH3. This primary amine is a colorless liquid is often used in chiral resolutions. Like benzylamine, it is relatively basic and forms stable ammonium salts and imines.
Preparation and optical resolution
1-Phenylethylamine may be prepared by the reductive amination of acetophenone:[1]
- C
6H
5C(O)CH
3 + NH
3 + H
2 → C
6H
5CH(NH
2)CH
3 + H
2O
The Leuckart reaction, using ammonium formate, is another method for this transformation.[2]
L-malic acid is used to resolve 1-Phenylethylamine, a versatile resolving agent in its own right. The dextrorotatory enantiomer crystallizes with the malate, leaving the levorotatory form in solution.[3]
See also
- 2-Phenylethylamine
References
- ↑ John C. Robinson, Jr. and H. R. Snyder (1943). "α-Phenylethylamine". Organic Syntheses 23: 68. doi:10.15227/orgsyn.023.0068.
- ↑ Mann, F. G.; Saunders, B. C. (1960). Practical Organic Chemistry, 4th Ed.. London: Longman. pp. 223–224. ISBN 9780582444072. https://www.scribd.com/doc/46973684/Practical-Organic-Chemistry-Frederick-George-Mann.
- ↑ A. W. Ingersoll (1937). "d- and l-α-Phenylethylamine". Organic Syntheses 17: 80. doi:10.15227/orgsyn.017.0080.
 |
