Chemistry:Bromo-DragonFLY
Bromo-DragonFLY, also known as DOB-DragonFLY (DOB-DFLY), is a psychedelic drug of the phenethylamine, DOx, and FLY families.[1][2] It is taken orally.[1][3] The drug has a delayed onset of up to 6 hours and a very long duration of up to 1 to 3 days.[1][3]
Side effects of Bromo-DragonFLY include nausea and vomiting, headache, tachycardia, hypertension, anxiety, panic attacks, pupil dilation, convulsions and vasoconstriction, among others.[1] The drug acts as an agonist of the serotonin 5-HT2 receptors, including the serotonin 5-HT2A, 5-HT2B, and 5-HT2C receptors.[1][4][5] It is also a potent monoamine oxidase A (MAO-A) inhibitor.[6]
Bromo-DragonFLY was first described in the scientific literature by David E. Nichols and colleagues in 1998.[1][4] It was encountered as a novel designer drug in 2006.[7] The drug has been associated with an unusually high degree of toxicity and numerous hospitalizations and fatalities.[1] It has become a controlled substance in various countries in the world.[1]
Use and effects
Bromo-DragonFLY was not included nor mentioned in Alexander Shulgin's 1991 book PiHKAL (Phenethylamines I Have Known and Loved) as it had not yet been discovered.[8] However, he subsequently described it in his 2011 book The Shulgin Index, Volume One: Psychedelic Phenethylamines and Related Compounds.[9] The dose range of Bromo-DragonFLY is not precisely known, but typical doses are in the range of 100 to 1,000 μg orally.[1][3][10][11] However, a death has been reported at approximately 700 μg Bromo-DragonFLY.[11] Its onset can be delayed by up to 6 hours and its duration is in the range of 12 to 24 hours for many users, but can be up to 2 to 3 days.[3][10][1] The drug's effects include profound hallucinations and visual distortions, sound alterations, a sense of connection or belonging with other realities, a sense of peace and well-being, emotional stimulation, and meeting with entities.[1]
Side effects
The side effects of Bromo-DragonFLY have been described.[1]
Overdose and toxicity

The toxicity of Bromo-DragonFLY appears to be fairly high for humans, with reports of at least five deaths believed to have resulted from Bromo-DragonFLY in Norway,[12] Sweden,[13][14] Denmark,[15][16] Finland[17] and the United States.
Laboratory testing has confirmed that in October 2009, a batch of Bromo-DragonFLY was distributed, mislabeled as the related compound 2C-B-FLY, which is around 20 times less potent than BDF by weight. This mistake is believed to have contributed to several lethal overdoses and additional hospitalizations. The batch implicated in these deaths also contained significant synthesis impurities, which may have contributed to the toxicity.[18]
Vasoconstrictive action resulting from severe overdose of Bromo-DragonFLY is known to have caused tissue necrosis of the extremities in at least one case. In September 2007, a 35-year-old Swedish male required amputation of the front part of his feet and several fingers on one hand after taking a massive (but unknown) overdose; reportedly, the compound acted as a long-acting efficacious vasoconstrictor, leading to necrosis and gangrene which became apparent several weeks after the overdose occurred. Treatment was of limited efficacy in this case, although tolazoline is reportedly an effective treatment where available.[19][20]
Overdose-associated disturbing experiences and health problems have been described. One case in 2008 in England involved inhalation of vomit, causing nearly fatal asphyxia.[21] Seizures have also been reported.[22]
On October 3, 2009, a 22-year-old male from Copenhagen died after ingesting Bromo-DragonFLY. His friend described the trip saying, "It was like being dragged to hell and back again. Many times. It is the most evil [thing] I've ever tried. It lasted an eternity."[23]
On May 7, 2011, in the United States, two young adults died after overdosing on Bromo-DragonFLY, which they thought was 2C-E, and several others were hospitalized during the same incident. Because they took a dosage appropriate for 2C-E, those who took the drug received, in some cases, 100 times the normal dose. Both deaths followed seizures, vomiting blood, and terrifying hallucinations.[24]
Interactions
Pharmacology
Pharmacodynamics
| Target | Affinity (Ki, nM) |
|---|---|
| 5-HT1A | 234 |
| 5-HT1B | 1,050 |
| 5-HT1D | 347 |
| 5-HT1E | 1,620 |
| 5-HT1F | ND |
| 5-HT2A | 0.04–1.86 (Ki) 0.141–4.79 (EC50) 80–120% (Emax) |
| 5-HT2B | 0.126–0.19 (Ki) 0.0851–0.457 (EC50) 75–105% (Emax) |
| 5-HT2C | 0.02–1.48 (Ki) 0.0617–4.90 (EC50) 63–104% (Emax) |
| 5-HT3 | >10,000 |
| 5-HT4 | ND |
| 5-HT5A | >10,000 |
| 5-HT6 | 339 |
| 5-HT7 | 148 |
| α1A–α1D | >10,000 |
| α2A | 398 |
| α2B | 550 |
| α2C | 955 |
| β1 | 1,100 |
| β2 | 2,140 |
| β3 | >10,000 |
| D1 | 955 |
| D2 | 2,240 |
| D3 | 417 |
| D4, D5 | >10,000 |
| H1–H4 | >10,000 |
| M1–M5 | >10,000 |
| TAAR1 | ND |
| I1 | ND |
| σ1 | 427 |
| σ2 | 2,340 |
| SERT | 4,070 (Ki) ND (IC50) |
| NET | >10,000 (Ki) ND (IC50) |
| DAT | >10,000 (Ki) ND (IC50) |
| MAO-A | 352 (Ki) 540 (IC50) |
| MAO-B | IA (IC50) |
| Notes: The smaller the value, the more avidly the drug binds to the site. All proteins are human unless otherwise specified. Refs: [25][5][6][26][27][28][4] | |
Bromo-DragonFLY has very high affinity for the serotonin 5-HT2 receptors, including for the serotonin 5-HT2A receptor (Ki = 0.04 nM), serotonin 5-HT2B receptor (Ki = 0.19 nM), and serotonin 5-HT2C receptor (Ki = 0.02 nM).[4][1] It is a highly potent high-efficacy partial agonist to full agonist of these three receptors.[5][25] Bromo-DragonFLY is also a potent monoamine oxidase A (MAO-A) inhibitor, which may contribute to its risks.[6] The comprehensive receptor interactions of Bromo-DragonFLY have been reported.[25]
Bromo-DragonFLY produces the head-twitch response, a behavioral proxy of psychedelic effects, in rodents.[29][5] It is among the most potent phenethylamine in terms of this effect, with similar potency as 25I-NBOMe.[29][5] Bromo-DragonFLY was only slightly less potent than LSD in this assay.[29] The drug also substitutes for LSD and DOI in rodent drug discrimination tests.[4][26] Once again, it was among the most potent phenethylamines tested.[26] Bromo-DragonFLY was slightly more potent than LSD in substituting for LSD in rodents, but was slightly less potent than LSD in substituting for DOI in rodents.[4][26][29] It was the first known phenethylamine to surpass LSD in potency in this assay.[30] In partial contrast to the preceding animal findings however, Bromo-DragonFLY is several-fold less potent as a psychedelic in humans than LSD but is similar in potency to 25I-NBOMe.[29]
Pharmacokinetics
The in-vitro and in-silico pharmacokinetics of Bromo-DragonFLY have been studied.[6][31]
Chemistry
Synthesis
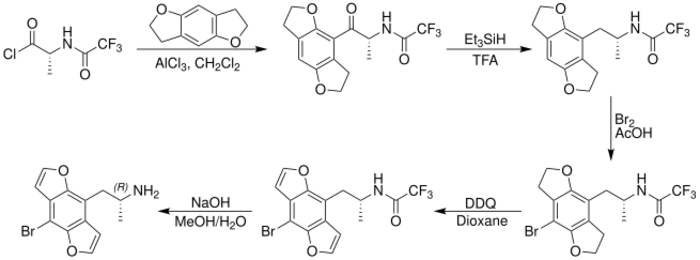
The first chemical synthesis of racemic Bromo-DragonFLY was reported by David E. Nichols in 1998 and was an expansion upon earlier research into the tetrahydrobenzodifuran analogue of DOB.[4] The 1998 synthesis of racemic Bromo-DragonFLY starts from hydroquinone, which is dialkylated with 1-bromo-2-chloroethane, brominated, and treated with n-butyllithium to yield the tetrahydrobenzodifuran ring system. After formylation of the ring system, the nitropropene derivative was obtained by condensation with nitroethane under ammonium acetate catalysis. The nitropropene derivative was then reduced with lithium aluminium hydride to yield the amine intermediate, which was protected with trifluoroacetic anhydride. Following para-bromination with elemental bromine and oxidation of the tetrahydrobenzodifuran ring system with DDQ, the trifluoroacetyl protecting group of the amine was removed to give Bromo-DragonFLY as a racemic mixture of the R and S enantiomers.
In 2001, David E. Nichols reported an enantiospecific synthesis of Bromo-DragonFLY which allowed the individual R and S enantiomers to be studied.[32] Further research determined that (R)-(-)-Bromo-DragonFLY possessed greater binding affinity at the 5-HT2A and 5-HT2C receptors than (S)-(-)-Bromo-DragonFLY. To synthesize the more active R enantiomer, a derivative of D-alanine was reacted with 2,3,6,7-tetrahydrobenzodifuran in a Friedel–Crafts acylation, yielding an intermediate containing a β-keto moiety which was removed by treatment with triethylsilane in trifluoroacetic acid. After para-bromination and oxidation of the ring system with DDQ, the amine was deprotected yielding (R)-(-)-Bromo-DragonFLY.
Analogues
Analogues of Bromo-DragonFLY (DOB-DFLY) include DOB, DOB-FLY, DOB-2-DRAGONFLY-5-BUTTERFLY, DOB-5-hemiFLY, 2C-B-FLY, 2C-B-DragonFLY, and 2CBFly-NBOMe, among others.[9][33]
History
Society and culture
Legal status
Internationally Bromo DragonFLY is an Unscheduled drug because is not into the Convention on Psychotropic substances of 1971 however still could be controlled for the analogue laws in some countries or for the sale of toxic substances for human consumption.
Australia
As of 9 September 2011, Bromo-DragonFLY was added to Schedule 2 of the Queensland Drugs Misuse Regulation 1987.[34]
Nationally, the drug is listed under Schedule 9 (Prohibited) of the Poisons Standard. Accordingly, the drug is prohibited in all states and territories.[35]
Canada
As of Oct 12, 2016, Bromo-DragonFLY is listed in Schedule III of the Canadian Controlled Drugs and Substances Act: "2C-phenethylamines and their salts, derivatives, isomers and salts of derivatives and isomers", a broad definition which corresponds to anything with a 2,5-dimethoxyphenethylamine core, including (but not limited to) the 2C family (including e.g. βk-2C-B), the DOx chemical class, the TMA family, Aleph aka DOT, NBOMe, the 25x-NBx series, and of course, Bromo-DragonFLY itself.[36][37]
Denmark
On December 3, 2007, the drug was banned in Denmark.[38] The substance has been declared illegal by health minister Jakob Axel Nielsen, following recommendations from the Danish Health Ministry. It is currently classified as a dangerous narcotic and therefore its possession, manufacture, importation, supply or usage is strictly prohibited. Anyone involved in such activities can face legal action.[39]
Finland
As of 12 March 2012, Bromo-DragonFLY is an illegal designer drug.[40]
Norway
Bromo-DragonFLY is currently on the Norwegian narcotics list.[41][42]
Poland
Romania
The chemical compound has been added as an illegal substance under the Law 143/2000 on February 10, 2010.[43]
Sweden
Sveriges riksdag added Bromo-Dragonfly to schedule IV ("substances, plant materials and fungi that hasn't any or without nothing medical use") as narcotics in Sweden as of Jan 3, 2008, published by Medical Products Agency in their regulation LVFS 2007:14 listed as Bromo-Dragonfly, brombensodifuranyl-isopropylamin.[44] Bromo-DragonFLY was first classified as "health hazard" by Sveriges riksdags health ministry Statens folkhälsoinstitut under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of Jul 15, 2007, in their regulation SFS 2007:600 listed as brombensodifuranylisopropylamin (Bromo-Dragonfly), making it illegal to sell, purchase, buy, retail or possess.[45]
United Kingdom
Bromo-DragonFLY is widely reported by the media as being a Class A drug.[21] However, as of 2014, it remains unclear to what extent it is covered by the UK phenylethylamine catch-all clause, with commentators noting both the structural similarities[46] and differences[47] to the phenylethylamine class. If the prosecution could demonstrate structural similarity in court, it would be considered a Class A substance[48] but as a benzodifuran it is significantly different to this class. It is not explicitly named in the misuse of drugs act.[49] It would be covered by the UK Psychoactive Substances Act 2016 but only if it is sold or traded for human consumption.
United States
Bromo-DragonFLY is unscheduled at federal level in the United States,[50] but could possibly be prosecuted under the Federal Analogue Act if it is sold for human consumption due to its similarities with 2C-B and DOB. Bromo-DragonFLY is listed as a Schedule I substance in Oklahoma.[51]
See also
- FLY (psychedelics)
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 "Designer drugs on the internet: a phenomenon out-of-control? the emergence of hallucinogenic drug Bromo-Dragonfly". Curr Clin Pharmacol 6 (2): 125–129. May 2011. doi:10.2174/157488411796151129. PMID 21592070.
- ↑ "Erowid Bromo-Dragonfly Vault". https://www.erowid.org/chemicals/bromo_dragonfly/bromo_dragonfly.shtml.
- ↑ 3.0 3.1 3.2 3.3 Cite error: Invalid
<ref>tag; no text was provided for refs namedMallaroniMasonVinckenbosch2022 - ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 4.7 "A novel (benzodifuranyl)aminoalkane with extremely potent activity at the 5-HT2A receptor". Journal of Medicinal Chemistry 41 (26): 5148–5149. December 1998. doi:10.1021/jm9803525. PMID 9857084.
- ↑ 5.0 5.1 5.2 5.3 5.4 "Identification of 5-HT2A receptor signaling pathways associated with psychedelic potential". Nature Communications 14 (1). December 2023. doi:10.1038/s41467-023-44016-1. PMID 38102107. Bibcode: 2023NatCo..14.8221W.
- ↑ 6.0 6.1 6.2 6.3 "Bromo-dragonfly, a psychoactive benzodifuran, is resistant to hepatic metabolism and potently inhibits monoamine oxidase A". Toxicology Letters 295: 397–407. October 2018. doi:10.1016/j.toxlet.2018.07.018. PMID 30036687.
- ↑ "EMCDDA–Europol 2006 Annual Report on the implementation of Council Decision 2005/387/JHA". European Monitoring Centre for Drugs and Drug Addiction & European Union Agency for Law Enforcement Cooperation. https://isomerdesign.com/bitnest/external/EMCDDA/New-Drugs-In-Europe-2006.
- ↑ Shulgin, Alexander; Shulgin, Ann (September 1991). PiHKAL: A Chemical Love Story. Berkeley, California: Transform Press. ISBN 0-9630096-0-5. OCLC 25627628. http://www.erowid.org/library/books_online/pihkal/pihkal.shtml.
- ↑ 9.0 9.1 The Shulgin Index, Volume One: Psychedelic Phenethylamines and Related Compounds. 1. Berkeley: Transform Press. 2011. ISBN 978-0-9630096-3-0.
- ↑ 10.0 10.1 "Clinical toxicology of newer recreational drugs". Clin Toxicol (Phila) 49 (8): 705–719. October 2011. doi:10.3109/15563650.2011.615318. PMID 21970769. "According to user websites, a typical BromodragonFLY dose is 0.2–1 mg with an onset of action of up to 6 h and duration of action of 2 or 3 days (Psychonaut web mapping 2010).66".
- ↑ 11.0 11.1 "A fatal poisoning involving Bromo-Dragonfly". Forensic Science International 183 (1): 91–96. 10 January 2009. doi:10.1016/j.forsciint.2008.11.001. PMID 19091499. https://www.sciencedirect.com/science/article/abs/pii/S0379073808004295.
- ↑ "Erowid Bromo-Dragonfly Vault: Reported GHB-Related Death Involved Bromo-Dragonfly". http://www.erowid.org/chemicals/bromo_dragonfly/bromo_dragonfly_death2.shtml.
- ↑ Kajsa Hallberg (2007-04-03). "Man i 20-årsåldern dog av drogen Dragonfly" (in sv). expressen.se (AB Kvällstidningen Expressen). http://www.expressen.se/Nyheter/1.622826.
- ↑ Ritzau (2008-08-24). "Nyt stof har slået dansker ihjel" (in da). jp.dk. http://jp.dk/indland/aar/article1418250.ece.
- ↑ Andreasen MF; Telving R; Birkler RI; Schumacher B; Johannsen M (January 2009). "A fatal poisoning involving Bromo-Dragonfly". Forensic Science International 183 (1–3): 91–6. doi:10.1016/j.forsciint.2008.11.001. PMID 19091499.
- ↑ "Kassisurman päätekijälle 11,5 vuoden vankeustuomio taposta" (in fi). Helsingin Sanomat. 2012-12-14. http://www.hs.fi/kotimaa/Kassisurman+p%C3%A4%C3%A4tekij%C3%A4lle+115+vuoden+vankeustuomio+taposta/a1305551347404.
- ↑ "Erowid 2C-B-Fly Vault: Death Report". http://www.erowid.org/chemicals/2cb_fly/2cb_fly_death1.shtml.
- ↑ Bromo-dragonfly – livsfarlig missbruksdrog
- ↑ "[Bromo-dragon fly--life-threatening drug. Can cause tissue necrosis as demonstrated by the first described case]". Läkartidningen 105 (16): 1199–1200. 2008. PMID 18522262.
- ↑ 21.0 21.1 "England | Surrey | 'I nearly died from taking £5 hit'". BBC News. 2008-03-27. http://news.bbc.co.uk/1/hi/england/surrey/7315020.stm.
- ↑ "Delayed onset of seizures and toxicity associated with recreational use of Bromo-dragonFLY". Journal of Medical Toxicology 5 (4): 226–9. December 2009. doi:10.1007/bf03178273. PMID 19876858.
- ↑ "| Danish man died after trip on Chinese drug". Jp.dk. 13 November 2009. http://jp.dk/indland/kbh/article1884968.ece.
- ↑ "Second Victim Dies After Taking Designer Drug In Konawa". http://www.newson6.com/story/14641463/second-victim-dies-after-taking-designer-drug-in-konawa.
- ↑ 25.0 25.1 25.2 "The polypharmacology of psychedelics reveals multiple targets for potential therapeutics". Neuron 113 (19): 3129–3142.e9. July 2025. doi:10.1016/j.neuron.2025.06.012. PMID 40683247. https://www.cell.com/cms/10.1016/j.neuron.2025.06.012/attachment/fb707b72-8fd1-4d4f-96f5-7fcf419b185d/mmc11.pdf.
- ↑ 26.0 26.1 26.2 26.3 Parker M (1998). Studies of perceptiotropic phenethylamines: Determinants of affinity for the 5-HT2A receptor (PhD. Thesis). Purdue University. Archived from the original on 2012-04-25. Retrieved 2011-12-16.
- ↑ "In vitro structure-activity relationship determination of 30 psychedelic new psychoactive substances by means of β-arrestin 2 recruitment to the serotonin 2A receptor". Arch Toxicol 94 (10): 3449–3460. October 2020. doi:10.1007/s00204-020-02836-w. PMID 32627074.
- ↑ "In vitro characterization of new psychoactive substances at the μ-opioid, CB1, 5HT1A, and 5-HT2A receptors-On-target receptor potency and efficacy, and off-target effects". Forensic Sci Int 317. December 2020. doi:10.1016/j.forsciint.2020.110553. PMID 33160102.
- ↑ 29.0 29.1 29.2 29.3 29.4 "Correlation between the potency of hallucinogens in the mouse head-twitch response assay and their behavioral and subjective effects in other species". Neuropharmacology 167. May 2020. doi:10.1016/j.neuropharm.2019.107933. PMID 31917152. PMC 9191653. https://isomerdesign.com/bitnest/external/10.1016/j.neuropharm.2019.107933.
- ↑ "Structure-activity relationships of phenylalkylamines as agonist ligands for 5-HT(2A) receptors". ChemMedChem 3 (9): 1299–1309. September 2008. doi:10.1002/cmdc.200800133. PMID 18666267. https://isomerdesign.com/bitnest/external/10.1002/cmdc.200800133.
- ↑ "ADME of Bromo-DragonFLY as an example of a new psychoactive substance (NPS) - application of in Silico methods for prediction: absorption, distribution, metabolism and excretion". Sci Rep 15 (1): 22949. July 2025. doi:10.1038/s41598-025-06453-4. PMID 40594888.
- ↑ 32.0 32.1 "C-(4,5,6-trimethoxyindan-1-yl)methanamine: a mescaline analogue designed using a homology model of the 5-HT2A receptor". Journal of Medicinal Chemistry 49 (14): 4269–4274. July 2006. doi:10.1021/jm060272y. PMID 16821786.
- ↑ (in de) Phenethylamine: von der Struktur zur Funktion. Nachtschatten-Science (1 ed.). Solothurn: Nachtschatten-Verlag. 2013. ISBN 978-3-03788-700-4. OCLC 858805226. https://books.google.com/books?id=-Us1kgEACAAJ. Retrieved 30 December 2025.
- ↑ "Queensland Drugs Misuse Regulation 1987". http://www.legislation.qld.gov.au/LEGISLTN/CURRENT/D/DrugsMisuseR87.pdf.
- ↑ Poisons Standard October 2015 "Poisons Standard October 2015". 30 September 2015. https://www.comlaw.gov.au/Details/F2015L01534.
- ↑ http://www.gazette.gc.ca/rp-pr/p2/2016/2016-05-04/html/sor-dors73-eng.php
- ↑ "Controlled Drugs and Substances Act". https://laws-lois.justice.gc.ca/eng/acts/c-38.8/FullText.html.
- ↑ "Amendment of Executive Order on Euphoriant Substances". Danish Medicines Agency. 2007. http://www.dkma.dk/da?sc_itemid=%7BF2733320-09EF-4477-AA70-0725C65FD9FD%7D&path=/1024/visUKLSArtikel.asp?artikelID=12407.
- ↑ "Erowid Bromo-Dragonfly Vault: Legal Status". http://www.erowid.org/chemicals/bromo_dragonfly/bromo_dragonfly_law.shtml#denmark.
- ↑ Design drugs Script error: The function "in_lang" does not exist.
- ↑ "List of narcotic drugs according to Norwegian law". http://www.lovdata.no/for/sf/ho/to-19780630-0008-001.html.
- ↑ "Statens Legemiddelverk about derivates and Bromo-DragonFLY". http://www.legemiddelverket.no/templates/InterPage____57471.aspx.
- ↑ "Modified Romanian law 143/2000 on January 10, 2010.". http://www.legestart.ro/Ordonanta-de-urgenta-6-2010-modificarea-completarea-Legii-143-2000-prevenirea-combaterea-traficului-consumului-ilicit-droguri-completarea-Legii-339-2005-regimul-juridic-plantelor-(MzQ3MzAw).htm.
- ↑ "Läkemedelsverkets föreskrifter - LVFS och HSLF-FS | Läkemedelsverket". http://www.lakemedelsverket.se/upload/lvfs/LVFS_2007-14.pdf.
- ↑ "Förordning om ändring i förordningen (1999:58) om förbud mot vissa hälsofarliga varor". Svensk författningssamling. http://www.notisum.se/rnp/sls/sfs/20070600.pdf.
- ↑ Psychonaut Webmapping Research Group. "Bromo-Dragonfly Report". Institute of Psychiatry, London. http://www.psychonautproject.eu/documents/reports/Bromodragonfly.pdf.
- ↑ "Ask Erowid : ID 3103: Does Bromo-Dragonfly fall under the UK ban on phenethylamines?". http://www.erowid.org/ask/ask.php?ID=3103.
- ↑ Advisory Council on the Misuse of Drugs. "Consideration of the Novel Psychoactive Substances ('Legal Highs')". UK Home Office. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/119139/acmdnps2011.pdf.
- ↑ UK Home Office. "UK Misuse of Drugs Act 1971 > 1971 c. 38 > SCHEDULE 2". http://www.legislation.gov.uk/ukpga/1971/38/schedule/2.
- ↑ Orange Book: List of Controlled Substances and Regulated Chemicals (January 2026), United States: U.S. Department of Justice: Drug Enforcement Administration (DEA): Diversion Control Division, January 2026, https://www.deadiversion.usdoj.gov/schedules/orangebook/orangebook.pdf
- ↑ "Man sentenced in 2011 designer drug deaths of two Oklahoma college students". https://www.oklahoman.com/story/news/crime/2012/08/13/man-sentenced-in-2011-designer-drug-deaths-of-two-oklahoma-college-students/61052749007/.
External links
- DOB-dragonFLY (Bromo-dragonFLY) - Isomer Design
- Bromo-DragonFLY - PsychonautWiki
- Bromo-Dragonfly - Erowid
- The Big & Dandy Bromo-Dragonfly/DOB-Dragonfly Thread - Bluelight
- Bromo-DragonFLY - TripSit
- The Story of Bromo-DragonFLY - Alien Insect on Drugs (Andrew Gallimore) - Substack
 |