Chemistry:Chlornaltrexamine
From HandWiki
Short description: Chemical compound
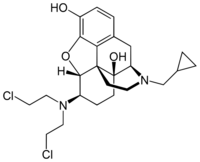
| |
| Names | |
|---|---|
| IUPAC name
6β-[Bis(2-chloroethyl)amino]-17-(cyclopropylmethyl)-4,5α-epoxymorphinan-3,14-diol
| |
| Systematic IUPAC name
(4R,4aS,7R,7aR,12bS)-7-[Bis(2-chloroethyl)amino]-3-(cyclopropylmethyl)-2,3,4,4a,5,6,7,7a-octahydro-1H-4,12-methano[1]benzofuro[3,2-e]isoquinoline-4a,9-diol | |
| Other names
α-Chlornaltrexamine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C24H32Cl2N2O3 | |
| Molar mass | 467.43 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Chlornaltrexamine is an irreversible mixed agonist–antagonist for μ-opioid receptors, which forms a covalent bond to the binding site. It is 22 times more potent than morphine. Its alkylating group is a bis(chloroalkyl)amino-residue similar to that of the nitrogen mustards.[1][2][3][4][5][6]
See also
- Chloroxymorphamine, an irreversible full agonist
- Naloxazone, an irreversible μ-opioid antagonist
- Oxymorphazone, an irreversible full agonist
References
- ↑ "6β-[N,N-Bis(2-chloroethyl)amino]-17-(cyclopropylmethyl)-4,5α-epoxy-3,14-dihydroxymorphinan(chlornaltrexamine) a potent opioid receptor alkylating agent with ultralong narcotic antagonist actitivty". J. Med. Chem. 21 (7): 598–9. July 1978. doi:10.1021/jm00205a002. PMID 209185.
- ↑ "Synthesis and pharmacologic characterization of an alkylating analogue (chlornaltrexamine) of naltrexone with ultralong-lasting narcotic antagonist properties". J. Med. Chem. 22 (2): 168–73. February 1979. doi:10.1021/jm00188a008. PMID 218009.
- ↑ "Chloroxymorphamine, and opioid receptor site-directed alkylating agent having narcotic agonist activity". Science 204 (4390): 316–8. April 1979. doi:10.1126/science.86208. PMID 86208. Bibcode: 1979Sci...204..316C.
- ↑ "Pharmacological studies with an alkylating narcotic agonist, chloroxymorphamine, and antagonist, chlornaltrexamine". J. Pharmacol. Exp. Ther. 213 (3): 539–44. June 1980. PMID 6162947. http://jpet.aspetjournals.org/cgi/pmidlookup?view=long&pmid=6162947.
- ↑ "Isolation of selective 3H-chlornaltrexamine-bound complexes, possible opioid receptor components in brains of mice". Life Sci. 27 (22): 2063–9. December 1980. doi:10.1016/0024-3205(80)90485-3. PMID 6259471.
- ↑ "Alkylation of opioid receptor subtypes by α-chlornaltrexamine produces concurrent irreversible agonistic and irreversible antagonistic activities.". J. Med. Chem. 26 (4): 503–6. 1983. doi:10.1021/jm00358a009. PMID 6300401.
