Chemistry:Ecadotril
| |
| Names | |
|---|---|
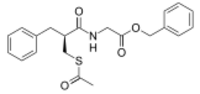
| Preferred IUPAC name
Benzyl [(2S)-3-(acetylsulfanyl)-2-benzylpropanamido]acetate | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C21H23NO4S | |
| Molar mass | 385.48 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ecadotril is a neutral endopeptidase inhibitor ((NEP[1]) EC 3.4.24.11[2]) and determined by the presence of peptidase family M13 as a neutral endopeptidase inhibited by phosphoramidon. Ecadotril is the (S)-enantiomer of racecadotril. NEP-like enzymes include the endothelin-converting enzymes.[3] The peptidase M13 family believed to activate or inactivate oligopeptide (pro)-hormones such as opioid peptides,[3] neprilysin[3] is another member of this group, in the case of the metallopeptidases and aspartic, the nucleophiles clan or family for example MA, is an activated water molecule.[1] The peptidase domain for members of this family also contains a bacterial member and resembles that of thermolysin the predicted active site residues for members of this family and thermolysin occur in the motif HEXXH.[4] Thermolysin complexed with the inhibitor (S)-thiorphan are isomeric thiol-containing inhibitors of endopeptidase EC 24-11[5] (also called "enkephalinase").
See also
References
- ↑ 1.0 1.1 "Substitution of potential metal-coordinating amino acid residues in the zinc-binding site of endopeptidase-24.11.". FEBS Lett. 324 (2): 196–200. June 1993. doi:10.1016/0014-5793(93)81392-D. PMID 8099556.
- ↑ "Molecular cloning and amino acid sequence of rat enkephalinase". Biochemical and Biophysical Research Communications 144 (1): 59–66. April 1987. doi:10.1016/S0006-291X(87)80475-8. PMID 3555489.
- ↑ 3.0 3.1 3.2 "The neprilysin (NEP) family of zinc metalloendopeptidases: Genomics and function.". BioEssays 23 (3): 261–9. March 2001. doi:10.1002/1521-1878(200103)23:3<261::AID-BIES1036>3.0.CO;2-K. PMID 11223883.
- ↑ "A family of membrane-embedded metalloproteases involved in regulated proteolysis of membrane-associated transcription factors.". Proc Natl Acad Sci USA 96 (26): 14765–14770. December 1999. doi:10.1073/pnas.96.26.14765. PMID 10611287.
- ↑ S. L. Roderick; M. C. Fournie-Zaluski; B. P. Roques; B. W. Matthews (February 1989). "Thiorphan and retro-thiorphan display equivalent interactions when bound to crystalline thermolysin". Biochemistry 28 (4): 1493–7. doi:10.1021/bi00430a011. PMID 2719912.
 |
