Chemistry:Dynorphin B
| |
| Names | |
|---|---|
| Other names
Dynorphin B-13; Rimorphin
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C74H115N21O17 | |
| Molar mass | 1570.8354 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
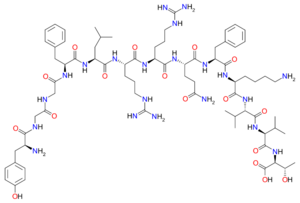
Dynorphin B, also known as rimorphin,[2] is a form of dynorphin and an endogenous opioid peptide with the amino acid sequence Tyr-Gly-Gly-Phe-Leu-Arg-Arg-Gln-Phe-Lys-Val-Val-Thr (YGGFLRRQFKVVT). Dynorphin B is generated as a proteolytic cleavage product of leumorphin, which in turn is a cleavage product of preproenkephalin B (prodynorphin). Dynorphin B has an identical N-terminal sequence, but different C-terminal sequence to dynorphin A. In an alanine scan of the non-glycine residues of dynorphin B, it was discovered that Tyr1 and Phe4 residues are critical for both opioid receptor affinity and κ-opioid receptor agonist potency, Arg6 and Arg7 promote κ-opioid affinity and Lys10 contributes to the opioid receptor affinity.[3]
Inducers of dynorphin B
Cannabinoid CP55,940 and △9-tetrahydrocannabinol (△9-THC) can induce the release of dynorphin B, which in return acts as an agonist of κ-opioid receptors, resulting in the production of antinociception.[4] Similarly, Tyr-D-Arg-Phe-Sar (TAPS) is capable of promoting a release of dynorphin B through the simulation of μ1-opioid receptors, causing a production of antinociception.[5] The antinociceptive effect produced by dynorphin B allows for spinal analgesia.
See also
References
- ↑ Dynorphin B - Compound Summary, PubChem.
- ↑ Paul V. Malven (12 January 1993). Mammalian Neuroendocrinology. CRC Press. p. 70. ISBN 978-0-8493-8757-9. https://books.google.com/books?id=nZoRPQa_qTkC&pg=PA70. Retrieved 22 April 2012.
- ↑ Joshi, Anand A.; Murray, Thomas F.; Aldrich, Jane V. (September 2017). "Alanine scan of the opioid peptide dynorphin B amide" (in en). Biopolymers 108 (5). doi:10.1002/bip.23026. PMID 28464209.
- ↑ Houser, Susan J; Eads, Micah; Embrey, James P; Welch, Sandra P (February 2000). "Dynorphin B and spinal analgesia: induction of antinociception by the cannabinoids CP55,940, Δ9-THC and anandamide1Published on the World Wide Web on 18 January 2000.1" (in en). Brain Research 857 (1–2): 337–342. doi:10.1016/S0006-8993(00)01981-8. PMID 10700588. https://linkinghub.elsevier.com/retrieve/pii/S0006899300019818.
- ↑ Mizoguchi, Hirokazu; Ito, Kanenori; Watanabe, Hiroyuki; Watanabe, Chizuko; Katsuyama, Sou; Fujimura, Tsutomu; Sakurada, Tsukasa; Sakurada, Shinobu (November 2006). "Contribution of spinal μ1-opioid receptors and dynorphin B to the antinociception induced by Tyr-d-Arg-Phe-Sar" (in en). Peptides 27 (11): 2786–2793. doi:10.1016/j.peptides.2006.07.006. PMID 16919848. https://linkinghub.elsevier.com/retrieve/pii/S0196978106003172.
 |
