Chemistry:Deltorphin I
From HandWiki
| |
| Names | |
|---|---|
| IUPAC names
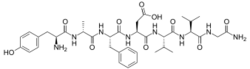
(3S)-3-[(2S)-2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl)propanamido]propanamido]-3-phenylpropanamido]-3-{[(1S)-1-{[(1S)-1-[(carbamoylmethyl)carbamoyl]-2-methylpropyl]carbamoyl}-2-methylpropyl]carbamoyl}propanoic acid
or L-tyrosyl-D-alanyl-L-phenylalanyl-L-α-aspartyl-L-valyl-L-valylglycinamide | |
| Other names
[D-Ala2]Deltorphin I; Deltorphin C
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| C37H52N8O10 | |
| Molar mass | 768.856 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Tracking categories (test):
Deltorphin I, also known as [D-Ala2]deltorphin I or deltorphin C, is a naturally occurring, exogenous opioid heptapeptide and hence, exorphin, with the amino acid sequence Tyr-D-Ala-Phe-Asp-Val-Val-Gly-NH2.[1][2] While not known to be endogenous to humans or other mammals, deltorphin I, along with the other deltorphins and the dermorphins, is produced naturally in the skin of species of Phyllomedusa,[1] a genus of frogs native to South and Central America. Deltorphin possesses very high affinity and selectivity as an agonist for the δ-opioid receptor,[1][2] and on account of its unusually high blood-brain-barrier penetration rate,[3] produces centrally-mediated analgesic effects in animals even when administered peripherally.[4]
See also
References
- ↑ 1.0 1.1 1.2 "Deltorphins: a family of naturally occurring peptides with high affinity and selectivity for delta opioid binding sites". Proceedings of the National Academy of Sciences of the United States of America 86 (13): 5188–92. July 1989. doi:10.1073/pnas.86.13.5188. PMID 2544892. Bibcode: 1989PNAS...86.5188E.
- ↑ 2.0 2.1 "New features of the delta opioid receptor: conformational properties of deltorphin I analogues". Biochemical and Biophysical Research Communications 169 (2): 617–22. June 1990. doi:10.1016/0006-291X(90)90375-W. PMID 2162669.
- ↑ "Deltorphin transport across the blood–brain barrier". Proceedings of the National Academy of Sciences of the United States of America 94 (17): 9469–74. August 1997. doi:10.1073/pnas.94.17.9469. PMID 9256506. Bibcode: 1997PNAS...94.9469F.
- ↑ "Structure-activity relationships of a series of [D-Ala2deltorphin I and II analogues; in vitro blood–brain barrier permeability and stability"]. The Journal of Pharmacology and Experimental Therapeutics 281 (2): 817–25. May 1997. PMID 9152390. http://jpet.aspetjournals.org/cgi/pmidlookup?view=long&pmid=9152390.
 |
